Professional Documents
Culture Documents
Rezista SPEC
Rezista SPEC
Uploaded by
EnriqueReyesOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Rezista SPEC
Rezista SPEC
Uploaded by
EnriqueReyesCopyright:
Available Formats
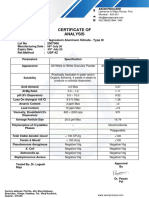
Specification Sheet
REZISTA® Starch
Routine Tests Specifications Test Numbers
Moisture 10.0 - 13.0% 46550
Foreign Matter 10 ppm (max.) 32555
Color 10 (max.) 22568
Ash 0.6% (max.) 09560
Anaerobic Mesophiles 3.9 tubes max. 10584
Aerobic Mesophiles 499 max./g 10580
pH (uncooked) 4.5 - 6.0 60520
Non-waxy Starch 7% (max.) 93820
Total Bacteria Count 20,000 max./g 10560
Mold 100 max./g 47010
Yeast 100 max./g 97010
Thermophilic Spore Counts
Total Thermophiles 150 max/10 g 16010
Flat Sours Spores 75 max/10 g 16011
Sulfide Spoilage Spores 5 max/10 g 16012
Gas Forming Anaerobic Spores 4 max/6 tubes 16013
Flavor Good 31060
Odor Good 52560
Brookfield Viscosity on Uncooked
Starch Slurry @ 77°F. 100 cps (max.) 88057
RVA Viscosity @ 50 ° C 850 - 1300 cps 90693
FDA Status
Complies with FDA Regulation 21CFR172.892 as Food Starch - Modified.
Recommended Storage Conditions
Store at ambient temperature and humidities. Storage in a cool, dry area will increase shelf life.
Shelf Life
Twenty-four months.
(Revised: 10/31/2008)
Tate & Lyle 2200 E. Eldorado Street Decatur, IL 62525 Phone: 217/423-4411 Fax: 217/421-2216 www.tateandlyle.com
The information contained in this bulletin should not be construed as recommending the use of our product in violation of any patent, or as warranties (expressed or
implied) of non infringement or its fitness for any particular purpose. Prospective purchasers are invited to conduct their own tests, studies and regulatory review to
determine the fitness of Tate & Lyle products for their particular purposes product claims or specific applications.
You might also like
- Epimastic 4100Document2 pagesEpimastic 4100Moin AltafNo ratings yet
- Certificate of Analysis: Address: No. 43, 6 Road Hi-Tech, Hi-Tech Zone, Xi'an, ChinaDocument1 pageCertificate of Analysis: Address: No. 43, 6 Road Hi-Tech, Hi-Tech Zone, Xi'an, ChinabluemyNo ratings yet
- BRAZIL SUGAR April 2021Document4 pagesBRAZIL SUGAR April 2021neaase kevin100% (1)
- Pumpkin Extract Powder Lot.K0422Document1 pagePumpkin Extract Powder Lot.K0422sudarat.phaNo ratings yet
- Rezista SPECDocument1 pageRezista SPECTran TuanNo ratings yet
- Cumicrete: Conventional Dense Castables (Calcined Clay Based)Document6 pagesCumicrete: Conventional Dense Castables (Calcined Clay Based)Devanathan ChinnasamyNo ratings yet
- Semola ExtraDocument1 pageSemola ExtraMihai NituNo ratings yet
- Whey Protein Concentrate 80 InstantDocument1 pageWhey Protein Concentrate 80 Instantkapil chopraNo ratings yet
- COA Sodium Alginate PureDocument1 pageCOA Sodium Alginate PurePankaj PatelNo ratings yet
- Propylene Glycol Pharma Grade FTDocument1 pagePropylene Glycol Pharma Grade FTDiego GuzmánNo ratings yet
- Lynn Dairy Lactose Specification 04-04-2023Document1 pageLynn Dairy Lactose Specification 04-04-2023Guadalupe GarcíaNo ratings yet
- Pro Self Flow Castables Cumiflow CumicastDocument2 pagesPro Self Flow Castables Cumiflow CumicastjulianoducaNo ratings yet
- TDS Skydrol LD 4 TDSDocument2 pagesTDS Skydrol LD 4 TDSsimbua72No ratings yet
- Eastman Skydrol PE-5 DSDocument2 pagesEastman Skydrol PE-5 DSavio321No ratings yet
- Bharat Petroleum Corpn. LTD.: Quality Assurance Laboratory: Irugur (A Govt. of India Undertaking)Document1 pageBharat Petroleum Corpn. LTD.: Quality Assurance Laboratory: Irugur (A Govt. of India Undertaking)madhuNo ratings yet
- Recent Advances in Volarization and Utilization of Fruits WasteDocument2 pagesRecent Advances in Volarization and Utilization of Fruits WasteSalman Nazir WattuNo ratings yet
- Certificate of AnalysisDocument1 pageCertificate of AnalysisbluemyNo ratings yet
- WPC 80% InstantDocument1 pageWPC 80% InstantPAULA AGUILERANo ratings yet
- Specification - DC Fine and MediumDocument3 pagesSpecification - DC Fine and MediumNurhasanahNo ratings yet
- Exxonmobil Marine Fuel OilDocument1 pageExxonmobil Marine Fuel OilPrinceSadhotraNo ratings yet
- 18ee En590, Euro 5, Gost R52368 - 2005 - WW ..Document1 page18ee En590, Euro 5, Gost R52368 - 2005 - WW ..JZ HuangNo ratings yet
- Eastman Skydrol LD4 Fire Resistant Hydraulic Fluid DSDocument2 pagesEastman Skydrol LD4 Fire Resistant Hydraulic Fluid DSavio321100% (1)
- Amur Cork TreeCOADocument1 pageAmur Cork TreeCOAAngelo SantosNo ratings yet
- SDS Natve Tapioca Starch Food GradeDocument1 pageSDS Natve Tapioca Starch Food GradesinarkanakajayaNo ratings yet
- B20 Biodiesel Fuel and Technical Requirements PDFDocument35 pagesB20 Biodiesel Fuel and Technical Requirements PDFandistwn99No ratings yet
- Pertamina Biodiesel Technical Requirements PDFDocument35 pagesPertamina Biodiesel Technical Requirements PDFandistwn99No ratings yet
- Lubricity Pilot Test: Test Performed by Eduard Mora Barrancabermeja's Lab TechDocument1 pageLubricity Pilot Test: Test Performed by Eduard Mora Barrancabermeja's Lab TechLiliana Rey OsorioNo ratings yet
- Biodiesel Fuel and How To HandlingDocument35 pagesBiodiesel Fuel and How To Handlingandistwn99No ratings yet
- COA - MAS IC Gaffel-Convertido (1) 20222Document2 pagesCOA - MAS IC Gaffel-Convertido (1) 20222Alejandro RestrepoNo ratings yet
- COA LOT36517 Natural Regular SlicedDocument1 pageCOA LOT36517 Natural Regular Slicedgracielaserrano90No ratings yet
- 4 DocsDocument4 pages4 DocsYanina GutierrezNo ratings yet
- Whytheat KDocument1 pageWhytheat KShubham SharmaNo ratings yet
- COA of Bromelain 1200 GDUDocument1 pageCOA of Bromelain 1200 GDUEndah WulandariNo ratings yet
- Specification - DC FineDocument1 pageSpecification - DC FineBepdjNo ratings yet
- COA of Riboflavin 5 - Sodium Phosphate 23120803Document2 pagesCOA of Riboflavin 5 - Sodium Phosphate 23120803Dwi Satria PutraNo ratings yet
- Regulation in Petrol IndustryDocument10 pagesRegulation in Petrol Industryhanah87No ratings yet
- Hakkani Paper BoilerDocument1 pageHakkani Paper BoilerpacificoneassociatesNo ratings yet
- Typical Properties: Mil Marketing International Ltda. - Representante en Chile de Jyoti Ceramic Industries LTDDocument1 pageTypical Properties: Mil Marketing International Ltda. - Representante en Chile de Jyoti Ceramic Industries LTDFelipe Reyes GallardoNo ratings yet
- Epilux 800Document2 pagesEpilux 800anandNo ratings yet
- 04 PDFDocument24 pages04 PDFnamNo ratings yet
- General Properties: High Purity Insulating CastableDocument1 pageGeneral Properties: High Purity Insulating CastableDeepakNo ratings yet
- RobbiathaneDocument2 pagesRobbiathaneAnson FranklinNo ratings yet
- Epilux 5: Product DescriptionDocument2 pagesEpilux 5: Product DescriptionShyam ShrishNo ratings yet
- Continuous PretreatmentDocument5 pagesContinuous PretreatmentNghia Phan Trung100% (1)
- Ad Hess IvesDocument3 pagesAd Hess Ivesfarukh azeem100% (1)
- Overview Qa Manufacture: Disusun Oleh: Bara AfganDocument23 pagesOverview Qa Manufacture: Disusun Oleh: Bara AfganSindy AstaNo ratings yet
- 4000 TCDDocument18 pages4000 TCDNaeemSialNo ratings yet
- HSD BS Vi-Coa-Fb7005c - (516687)Document2 pagesHSD BS Vi-Coa-Fb7005c - (516687)vprabuNo ratings yet
- Phenoline 187 VOC: Selection & Specification DataDocument4 pagesPhenoline 187 VOC: Selection & Specification Datarajiv_quantumNo ratings yet
- Gasoline & Diesel SpecsDocument3 pagesGasoline & Diesel SpecsKaustubhNo ratings yet
- Racing Brake Fluid 660 Factory LineDocument2 pagesRacing Brake Fluid 660 Factory LineVelibor KaranovicNo ratings yet
- Guar Gum FCC 200 Mesh Edicol 60-70 GG6070Document1 pageGuar Gum FCC 200 Mesh Edicol 60-70 GG6070darkroomzcNo ratings yet
- Bioprex Labs: Certificate of AnalysisDocument1 pageBioprex Labs: Certificate of AnalysisThuy PhanNo ratings yet
- Contavan GalDocument3 pagesContavan Galraju_hosssenNo ratings yet
- EPD 100 Plascotuff Epoxy Coal TarDocument2 pagesEPD 100 Plascotuff Epoxy Coal TarAdhil RamsurupNo ratings yet
- Swancor Chempulse 907: Novolac Epoxy Vinyl Ester ResinDocument4 pagesSwancor Chempulse 907: Novolac Epoxy Vinyl Ester ResinEVLYNNo ratings yet
- 1402-00 Simazinc EP PrimerDocument2 pages1402-00 Simazinc EP PrimerMuhammad Mulya JatiNo ratings yet
- Pds Luxathane 5000 VocDocument2 pagesPds Luxathane 5000 Vocmuthukumar100% (1)
- (M-05900) METHYLENE BLUE (For Microscopy)Document1 page(M-05900) METHYLENE BLUE (For Microscopy)hamza manafNo ratings yet
- Guide 5 Talking About Past PerfectDocument5 pagesGuide 5 Talking About Past PerfectgeraldineNo ratings yet
- Arthur Narrative ReportsDocument5 pagesArthur Narrative ReportsArthur TalomaNo ratings yet
- Contractual Methods of Sampling PDFDocument12 pagesContractual Methods of Sampling PDFBarclayNo ratings yet
- Sylhet International School & College: STD - III Subject: English 2 PaperDocument6 pagesSylhet International School & College: STD - III Subject: English 2 PaperFarhanChowdhuryMehdiNo ratings yet
- Tong Hop Bai Tap Cau Hoi Duoi Co Dap AnDocument18 pagesTong Hop Bai Tap Cau Hoi Duoi Co Dap AnTùng Nguyễn BáNo ratings yet
- Speakout DVD Extra Starter Unit 04Document1 pageSpeakout DVD Extra Starter Unit 04lkzhtubcnhfwbbgjxnsNo ratings yet
- Super Size MeDocument9 pagesSuper Size MeKevin HuynhNo ratings yet
- Dental CariesDocument20 pagesDental Carieschantelle acopNo ratings yet
- Tradition and Modernity in The Domestic Urban Kitchen Design in UgandaDocument165 pagesTradition and Modernity in The Domestic Urban Kitchen Design in UgandaAurora NorvalNo ratings yet
- Childhood ObesityDocument13 pagesChildhood Obesityapi-315466994100% (1)
- Auxiliary Verbs: Abbreviated Verb Clause Tense Aspect Modality VoiceDocument6 pagesAuxiliary Verbs: Abbreviated Verb Clause Tense Aspect Modality VoiceSeby Yacob0% (1)
- Verbs Transitive Types ANSWERSDocument11 pagesVerbs Transitive Types ANSWERSzander_knighttNo ratings yet
- Coca Cola IndiaDocument5 pagesCoca Cola IndiaGoldberry26No ratings yet
- DuolingoDocument11 pagesDuolingoFranNo ratings yet
- Bu I 2Document8 pagesBu I 2k61.2211740096No ratings yet
- Coffee TrainingDocument109 pagesCoffee TrainingKeith Hendrix Fagyan100% (1)
- Smart FarmingDocument68 pagesSmart FarminghaikalNo ratings yet
- Laufer - Was Odoric of Pordenone Ever in TibetDocument8 pagesLaufer - Was Odoric of Pordenone Ever in TibetbrendancrummerNo ratings yet
- Aldrin JornalllllllllllllllllllllDocument29 pagesAldrin JornalllllllllllllllllllllgemarieNo ratings yet
- Decanters Disc Stack, Belt Presses, BreweriesDocument12 pagesDecanters Disc Stack, Belt Presses, BreweriesMurali Krishna IndanaNo ratings yet
- The DPS Ultimate Guide To Food PhotographyDocument40 pagesThe DPS Ultimate Guide To Food PhotographyBeatrice BudeaNo ratings yet
- MANAGEMENTDocument35 pagesMANAGEMENTJincy Babu100% (1)
- Powerpoint PBL 2Document13 pagesPowerpoint PBL 2tariqNo ratings yet
- Tic TacDocument3 pagesTic TacalexcusNo ratings yet
- ROSA Aflatoxin (Quantitative) Test - Prepared For CargillDocument4 pagesROSA Aflatoxin (Quantitative) Test - Prepared For CargillOctavian Mita100% (1)
- Session 8-Case Studies On CSR Policy and Conflicts PDFDocument23 pagesSession 8-Case Studies On CSR Policy and Conflicts PDFSamarth TuliNo ratings yet
- HaccpDocument67 pagesHaccpPaulo DantasNo ratings yet
- PemDocument37 pagesPemJOSLINNo ratings yet
- Mauritius DineInDocument4 pagesMauritius DineInDaimler AGNo ratings yet
- I. Lý thuyết: Đại từ nhân xưng Tính từ sở hữu Đại từ sở hữuDocument4 pagesI. Lý thuyết: Đại từ nhân xưng Tính từ sở hữu Đại từ sở hữuLÂM LINHNo ratings yet