Professional Documents
Culture Documents
Amoniglycosid
Amoniglycosid
Uploaded by
kimanh phạmCopyright:
Available Formats
You might also like
- The AminoglycosidesDocument8 pagesThe AminoglycosidesVangenique Nieves AgrudaNo ratings yet
- Risk Management PlanDocument4 pagesRisk Management PlanHannah BalucaNo ratings yet
- DP On AglDocument12 pagesDP On AglDeepikaNo ratings yet
- Protein Synthesis Inhibitors - Second SessionalDocument10 pagesProtein Synthesis Inhibitors - Second Sessionaljhanavi rajeshNo ratings yet
- Protein Systhesis InhibitorsDocument31 pagesProtein Systhesis InhibitorsCabaddu AngieNo ratings yet
- Aminoglycoside Antibiotics: Mechanism of ActionDocument9 pagesAminoglycoside Antibiotics: Mechanism of Actionprabhakaran payamNo ratings yet
- Chapter 45 - Aminoglycosides SpectinomycinDocument6 pagesChapter 45 - Aminoglycosides SpectinomycinrheaasarenNo ratings yet
- AminoglycosidesDocument20 pagesAminoglycosidesPrasad SangishettyNo ratings yet
- AminoglycosideDocument22 pagesAminoglycosideRabi ShahNo ratings yet
- (BACTE-LEC) 8. Antimicrobial Susceptibility TestingDocument6 pages(BACTE-LEC) 8. Antimicrobial Susceptibility TestingCarlo CataagNo ratings yet
- Aminoglycoside: Systemic AminoglycosidesDocument47 pagesAminoglycoside: Systemic AminoglycosidesPawan PatelNo ratings yet
- Protein Synthesis InhibitorsDocument35 pagesProtein Synthesis InhibitorsHarsha MaheshwariNo ratings yet
- Aminoglycosides (17.07.2017)Document44 pagesAminoglycosides (17.07.2017)Habibul Kowser (Rishat)No ratings yet
- Streptomycin PDFDocument7 pagesStreptomycin PDFHdjdNo ratings yet
- Antibiotics GuideDocument33 pagesAntibiotics Guidej7qs46h8wrNo ratings yet
- Antibiotics Chart For MicrobiologyDocument5 pagesAntibiotics Chart For MicrobiologyTrevorNo ratings yet
- Pathophysiology Assignment 5Document5 pagesPathophysiology Assignment 5venkatasaiyuvarajNo ratings yet
- Inhibitors of Protein SynthesisDocument2 pagesInhibitors of Protein Synthesiselsayed barhomeNo ratings yet
- Protein Synthesis Inhibitors: Tasneem SmeratDocument78 pagesProtein Synthesis Inhibitors: Tasneem Smeratansam hirbaweNo ratings yet
- AMINOGLYCOSIDESDocument45 pagesAMINOGLYCOSIDESAbdullah EmadNo ratings yet
- Amino Glyc o SidesDocument27 pagesAmino Glyc o SidesAyesha AkbarNo ratings yet
- Table: Selected Antibacterial Antibiotics Antibiotics THAT . Mechanism of Action Names of Drugs Notes and ProblemsDocument4 pagesTable: Selected Antibacterial Antibiotics Antibiotics THAT . Mechanism of Action Names of Drugs Notes and ProblemsTJNo ratings yet
- 46 Aminoglycosides-2023Document16 pages46 Aminoglycosides-2023deo okiaNo ratings yet
- HandoutDocument17 pagesHandoutAlmalyn TimbayanNo ratings yet
- QuinolonDocument10 pagesQuinolonkimanh phạmNo ratings yet
- Aminoglikosida Farkol 2 2023Document20 pagesAminoglikosida Farkol 2 2023ghazialghifariNo ratings yet
- Aminoglycosides: Amlan GangulyDocument14 pagesAminoglycosides: Amlan GangulyAli Akand AsifNo ratings yet
- Aminoglycosides, Macrolides, Chloramphenicol, LincomycinsDocument7 pagesAminoglycosides, Macrolides, Chloramphenicol, LincomycinsMello DiaxNo ratings yet
- ANTIBIOTIKA REVIEW-2021 enDocument45 pagesANTIBIOTIKA REVIEW-2021 enBeby CantikqNo ratings yet
- P'cology of Amin & Spect (2022-P)Document42 pagesP'cology of Amin & Spect (2022-P)Tewodros TeshomeNo ratings yet
- Aminoglycosides.: Chapter No. 02 Week # 07Document2 pagesAminoglycosides.: Chapter No. 02 Week # 07Alee Iz HarNo ratings yet
- AntifungalagentsDocument52 pagesAntifungalagentsspriyansh202No ratings yet
- Inhibitor of Bacterial Protein SynthesisDocument83 pagesInhibitor of Bacterial Protein SynthesisNdayisaba CorneilleNo ratings yet
- Aminoglycoside: From Wikipedia, The Free EncyclopediaDocument11 pagesAminoglycoside: From Wikipedia, The Free Encyclopediaawajahat100% (3)
- ANTITUBERCULOUSDocument11 pagesANTITUBERCULOUSScribdTranslationsNo ratings yet
- MicrobialsDocument5 pagesMicrobialsMARIEMIL FOLLOSONo ratings yet
- CH 10 Microbiology Lecture PresentationDocument56 pagesCH 10 Microbiology Lecture PresentationRiriNo ratings yet
- Bacteriostatic Agents: Drugs Which Bind To The 50s Ribosomal UnitDocument3 pagesBacteriostatic Agents: Drugs Which Bind To The 50s Ribosomal UnitJoshua Trinidad100% (1)
- Amr SeminarDocument48 pagesAmr SeminarSwijalNo ratings yet
- Antibióticos en NefrotoxicidadDocument15 pagesAntibióticos en NefrotoxicidadHilma BermudezNo ratings yet
- Ch12 Part1Document15 pagesCh12 Part1lasanders601No ratings yet
- Antibacterial Class 03Document80 pagesAntibacterial Class 03tsegaab yosephNo ratings yet
- Department of Pharmacology: Prof. Dr. Asya RehmanDocument15 pagesDepartment of Pharmacology: Prof. Dr. Asya RehmanGareth BaleNo ratings yet
- Antibiotic Susceptibility TestingDocument46 pagesAntibiotic Susceptibility Testingditta antNo ratings yet
- AntimikrobaDocument50 pagesAntimikrobaarisita firmanNo ratings yet
- H01033 Hoá Dư C 1 Lecture 4 AminoglycosidesDocument27 pagesH01033 Hoá Dư C 1 Lecture 4 AminoglycosidesTor NgânNo ratings yet
- 6 - Lectins and PolypeptidesDocument6 pages6 - Lectins and PolypeptidesJonafe JuntillaNo ratings yet
- Notes MacrolidesDocument6 pagesNotes MacrolidesAlessa Marie BadonNo ratings yet
- TuberculosisDocument2 pagesTuberculosisElle ReyesNo ratings yet
- Pharmacology of AntimicrobialsDocument31 pagesPharmacology of AntimicrobialsJosette LeyvaNo ratings yet
- Antimicrobal Drugs #Dental 1Document30 pagesAntimicrobal Drugs #Dental 1ggNo ratings yet
- Antimicrobial Agents and Vaccinations - UpdatedDocument62 pagesAntimicrobial Agents and Vaccinations - Updatedjohn tsuiNo ratings yet
- Aminoglycosides: Official Reprint From Uptodate ©2021 Uptodate, Inc. And/Or Its Affiliates. All Rights ReservedDocument17 pagesAminoglycosides: Official Reprint From Uptodate ©2021 Uptodate, Inc. And/Or Its Affiliates. All Rights ReservedVanessa GomesNo ratings yet
- Tonghop ProDocument16 pagesTonghop Prokimanh phạmNo ratings yet
- General Characteristics of Antimicrobial Drug1Document5 pagesGeneral Characteristics of Antimicrobial Drug1vishuop061No ratings yet
- Antimicrobial AgentsDocument2 pagesAntimicrobial AgentsFahima RahmanNo ratings yet
- Haematology-Oncology McqsDocument80 pagesHaematology-Oncology McqsKarim MohamedNo ratings yet
- AminoglycosidesDocument1 pageAminoglycosidesmariam ibrahimNo ratings yet
- Antibiotic 1Document13 pagesAntibiotic 1ريدان عياشNo ratings yet
- 8 - AntibioticsDocument8 pages8 - AntibioticsArshad AbbasNo ratings yet
- 7 Natural Remedies For Epstein Barr VirusDocument40 pages7 Natural Remedies For Epstein Barr VirusalbinutaNo ratings yet
- Rai Work Experience SheetDocument2 pagesRai Work Experience Sheetmark langcayNo ratings yet
- Concepts of ProsthoDocument22 pagesConcepts of ProsthoKirti SharmaNo ratings yet
- 50 Items HADocument7 pages50 Items HAToni Marie Buenconsejo PunzalanNo ratings yet
- Tristill - Brochure - March 2017 New A4 Three FoldDocument2 pagesTristill - Brochure - March 2017 New A4 Three FoldLesley HollardNo ratings yet
- Case Study On PneumoniaDocument12 pagesCase Study On PneumoniaRintu JenaNo ratings yet
- The Structure and Function of The Cardiovascular SystemDocument28 pagesThe Structure and Function of The Cardiovascular SystemShamaeNogaNo ratings yet
- The Dorsal Skinfold Chamber: A Versatile Tool For Preclinical Research in Tissue Engineering and Regenerative MedicineDocument14 pagesThe Dorsal Skinfold Chamber: A Versatile Tool For Preclinical Research in Tissue Engineering and Regenerative MedicineTanvir DaphedarNo ratings yet
- Extracts of Justicia Wynaadensis LeavesDocument9 pagesExtracts of Justicia Wynaadensis LeavesPawan KumarNo ratings yet
- SBL 4 - Xerostomia - FINALDocument16 pagesSBL 4 - Xerostomia - FINALCara ChanNo ratings yet
- HCD2Document2 pagesHCD2fwijaya888No ratings yet
- Nursing Practice II EdgeworthDocument9 pagesNursing Practice II EdgeworthRichard Ines Valino100% (1)
- Communicable DiseaseDocument38 pagesCommunicable DiseaseDrShrikant JahagirdarNo ratings yet
- AF SeedingDocument6 pagesAF SeedingdonkeyendutNo ratings yet
- Chemical Product and Company Information: Material Safety Data SheetDocument5 pagesChemical Product and Company Information: Material Safety Data Sheetkirandas_mullasseryNo ratings yet
- Narrative Report - NSTP 2nd Sem Chapter 3Document1 pageNarrative Report - NSTP 2nd Sem Chapter 3Clint Kove YtemNo ratings yet
- Who Killed Christopher Goodman? by Allan Wolf Chapter SamplerDocument21 pagesWho Killed Christopher Goodman? by Allan Wolf Chapter SamplerCandlewick Press50% (2)
- Daftar PustakaDocument3 pagesDaftar PustakamemeeeyyyNo ratings yet
- Sample Academic Essay in APA Style (6 Edition)Document3 pagesSample Academic Essay in APA Style (6 Edition)Papa YawNo ratings yet
- N410 SyllabusDocument20 pagesN410 SyllabusTwobucktinNo ratings yet
- When To See A DoctorDocument2 pagesWhen To See A DoctorPooja ChapdiNo ratings yet
- نموذج انقاذ حياة الجديدDocument2 pagesنموذج انقاذ حياة الجديدraad_alghamdi_10% (1)
- HIV Transmission Through Breastfeeding: A Review of Available EvidenceDocument33 pagesHIV Transmission Through Breastfeeding: A Review of Available EvidenceJessica RamosNo ratings yet
- Psychophysiological Disorders - Kanna KDocument16 pagesPsychophysiological Disorders - Kanna KKannan KrishnamurthyNo ratings yet
- Formatif NBSSDocument17 pagesFormatif NBSSCYNTHIA ARISTANo ratings yet
- Escape From The Jade Colossus: by Bruce R. CordellDocument30 pagesEscape From The Jade Colossus: by Bruce R. CordellJuh Freitas38% (8)
- CGHS Enclosure 1Document77 pagesCGHS Enclosure 1bhupendrapawar279No ratings yet
- Paediatric Proton Therapy (Thomas 2019)Document9 pagesPaediatric Proton Therapy (Thomas 2019)Milda InayahNo ratings yet
- Surgical Hand Hygiene PosterDocument2 pagesSurgical Hand Hygiene PosterIrenne WibowoNo ratings yet
Amoniglycosid
Amoniglycosid
Uploaded by
kimanh phạmOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Amoniglycosid
Amoniglycosid
Uploaded by
kimanh phạmCopyright:
Available Formats
58
Chapter
MECHANISM OF ACTION
ANTIMICROBIAL ACTIVITY
Aminoglycosides
Conan MacDougall
■■ Cystic Fibrosis
■■ Topical Applications
ADVERSE EFFECTS OF AMINOGLYCOSIDES
RESISTANCE TO THE AMINOGLYCOSIDES ■■ Ototoxicity
■■ Nephrotoxicity
ADME ■■ Neuromuscular Blockade
■■ Other Adverse Effects
DOSING AND MONITORING
PHARMACOLOGICAL PROPERTIES OF INDIVIDUAL
THERAPEUTIC USES OF AMINOGLYCOSIDES AMINOGLYCOSIDES
■■ Urinary Tract Infections ■■ Gentamicin
■■ Pneumonia ■■ Tobramycin
■■ Meningitis ■■ Amikacin
■■ Peritonitis ■■ Netilmicin
■■ Bacterial Endocarditis ■■ Streptomycin
■■ Sepsis ■■ Neomycin
■■ Tularemia ■■ Paromomycin
■■ Plague ■■ Kanamycin
■■ Mycobacterial Infections
Aminoglycosides contain amino sugars linked to an aminocyclitol
ORIGINS
ring by glycosidic bonds (Figure 58–1). They are polycations, and their
Aminoglycosides are natural products or semisynthetic derivatives of polarity is responsible in part for pharmacokinetic properties shared by
compounds produced by a variety of soil actinomycetes. Strepto- all members of the group. For example, none is absorbed adequately after
mycin was first isolated from a strain of Streptomyces griseus. Gen- oral administration, inadequate concentrations are found in CSF, and all
tamicin and netilmicin are derived from species of the actinomycete are excreted relatively rapidly by the normal kidney. All members of the
Micromonospora. The difference in spelling (-micin) compared with group share the same spectrum of toxicity, most notably nephrotoxicity
the other aminoglycoside antibiotics (-mycin) reflects this difference and ototoxicity, which can involve the auditory and vestibular functions
in origin. Tobramycin is one of several components of an aminogly- of the eighth cranial nerve.
coside complex known as “nebramycin” that is produced by Streptomyces
tenebrarius. It is most similar in antimicrobial activity and toxicity
to gentamicin. In contrast to the other aminoglycosides, amikacin, a Mechanism of Action
derivative of kanamycin, and netilmicin, a derivative of sisomicin, are The aminoglycoside antibiotics are rapidly bactericidal. Bacterial killing
semisynthetic products. is concentration dependent: the higher the concentration, the greater the
rate of bacterial killing. The ratio of the peak concentration to the organ-
ism’s MIC is thus a key predictor of aminoglycoside efficacy. The inhibi-
tory activity of aminoglycosides persists after the serum concentration has
Aminoglycosides are natural products or semisynthetic derivatives of fallen below the MIC, a phenomenon known as the postantibiotic effect.
compounds produced by a variety of soil actinomycetes. Amikacin, a These properties probably account for the efficacy of high-dose, extended-
derivative of kanamycin, and netilmicin, a derivative of sisomicin, are interval dosing regimens.
semisynthetic products. Aminoglycosides diffuse through aqueous channels formed by porin
Aminoglycosides (gentamicin, tobramycin, amikacin, netilmicin, proteins in the outer membrane of gram-negative bacteria to enter the
kanamycin, streptomycin, paromomycin, and neomycin) are used primarily periplasmic space. Transport of aminoglycosides across the cytoplasmic
to treat infections caused by aerobic gram-negative bacteria. Streptomy- (inner) membrane depends on a transmembrane electrical gradient cou-
cin and amikacin are important agents for the treatment of mycobacte- pled to electron transport to drive permeation of these antibiotics. This
rial infections, and paromomycin is used orally for intestinal amebiasis. energy-dependent phase is rate limiting and can be blocked or inhibited
Aminoglycosides are bactericidal inhibitors of protein synthesis. Mutations by divalent cations (e.g., Ca2+ and Mg2+), hyperosmolarity, a reduction in
affecting proteins in the bacterial ribosome can confer marked resistance pH, and anaerobic conditions. Thus, the antimicrobial activity of amino-
to their action. Most commonly, resistance is due to aminoglycoside- glycosides is reduced markedly in the anaerobic environment of an abscess
metabolizing enzymes or impaired transport of drug into the cell; these mech- and in hyperosmolar acidic urine.
anisms may confer resistance to all aminoglycosides or only select agents. Once inside the cell, aminoglycosides bind to polysomes and interfere
Resistance genes are frequently acquired via plasmids or transposons. with protein synthesis by causing misreading and premature termination
Brunton_Ch58_p1039-p1048.indd 1039 08/09/17 5:58 PM
1040
Abbreviations AC ACI ACII ACIII
AC: acetylase H
AD: adenylase HC NH2 NH2
CHAPTER 58 AMINOGLYCOSIDES
ADME: absorption, distribution, metabolism, excretion NH2
O
CNS: central nervous system
CSF: cerebrospinal fluid O
OH
GI: gastrointestinal HO
IM: intramuscular NH2
IV: intravenous CH2OH
O
MIC: minimum inhibitory concentration AC
O
mRNA: messenger RNA
PO: by mouth NH2
HO
UTI: urinary tract infection
OH AD
Tobramycin
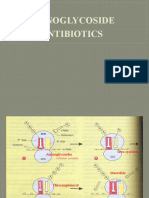
of mRNA translation (Figure 58–2). The primary intracellular site of
action of the aminoglycosides is the 30S ribosomal subunit. At least three AC Acetylase AD Adenylase
of these ribosomal proteins, and perhaps the 16S ribosomal RNA as well,
contribute to the streptomycin-binding site. Aminoglycosides interfere with Figure 58–1 Aminoglycoside structure and sites of activity of plasmid-mediated
the initiation of protein synthesis, leading to the accumulation of abnormal enzymes capable of inactivating aminoglycosides. Tobramycin is shown as a
initiation complexes; the drugs also can cause misreading of the mRNA representative; structural characteristics protect some aminoglycosides from
template and incorporation of incorrect amino acids into the growing the actions of some of these enzymes, explaining differences in spectrum of
polypeptide chains (Davis, 1988). The resulting aberrant proteins may be activity.
inserted into the cell membrane, leading to altered permeability and
further stimulation of aminoglycoside transport (Busse et al., 1992).
single agents to treat infections caused by gram-positive bacteria. How-
ever, in combination with a cell wall–active agent, such as a penicillin
Antimicrobial Activity or vancomycin, an aminoglycoside can produce a synergistic bacteri-
The antibacterial activity of gentamicin, tobramycin, and amikacin cidal effect in vitro. This effect has been most commonly employed for
is directed primarily against aerobic gram-negative bacilli (Mingeot- treatment of infections due to staphylococci, enterococci, viridans group
Leclercq et al., 1999). Kanamycin, like streptomycin, has a more limited streptococci, and Listeria. Clinically, the superiority of aminoglycoside
spectrum. The aerobic gram-negative bacilli vary in their susceptibility to combination regimens over cell-wall agents alone is not proven except in
the aminoglycosides (see Table 58–1). Gram-negative aerobic cocci such relatively few infections (discussed further in the chapter).
as Neisseria, Moraxella, and Haemophilus have varying susceptibilities. An
increasing number of gram-negative bacilli encountered in healthcare set-
tings (especially Klebsiella and Pseudomonas) display extensive resistance Resistance to the Aminoglycosides
to multiple classes of antibacterials; in these isolates, aminoglycosides may
Bacteria may be resistant to aminoglycosides through
be the only class of commonly used agents with in vitro activity.
Aminoglycosides have little activity against anaerobic microorganisms • inactivation of the drug by microbial enzymes;
or facultative bacteria under anaerobic conditions. Their action against • failure of the antibiotic to penetrate intracellularly; and
most gram-positive bacteria is limited, and they should not be used as • low affinity of the drug for the bacterial ribosome.
Blocks initiation
5′ 3′ of protein synthesis
Growing polypeptide
Blocks further
5′ 3′ translation and elicits
5′ 3′ premature termination
X
Incorporation of
aminoglycoside 5′ 3′ incorrect amino acid
Figure 58–2 Effects of aminoglycosides on protein synthesis. A. Aminoglycoside (represented by red circles) binds to the 30S ribosomal subunit and interferes
with initiation of protein synthesis by fixing the 30S-50S ribosomal complex at the start codon (AUG) of mRNA. As 30S-50S complexes downstream complete
translation of mRNA and detach; the abnormal initiation complexes, so-called streptomycin monosomes, accumulate, blocking further translation of the mes-
sage. Aminoglycoside binding to the 30S subunit also causes misreading of mRNA, leading to B, premature termination of translation with detachment of the
ribosomal complex and incompletely synthesized protein or C, incorporation of incorrect amino acids (indicated by the red X), resulting in the production of
abnormal or nonfunctional proteins.
Brunton_Ch58_p1039-p1048.indd 1040 08/09/17 5:58 PM
1041
TABLE 58–1 ■ SUSCEPTIBILITY TO AMINOGLYCOSIDES AND TYPICAL MINIMAL CONCENTRATIONS THAT WILL INHIBIT
90% (MIC90) OF CLINICAL ISOLATES FOR SEVERAL SPECIES
% SUSCEPTIBLE (MIC90 μg/mL)
SECTION VII CHEMOTHERAPY OF INFECTIOUS DISEASES
SPECIES GENTAMICIN TOBRAMYCIN AMIKACIN
Enterobacter spp. 97.0% (1) 96.0% (1) 100.0% (2)
Escherichia coli 88.2% (8) 86.3% (8) 99.0% (4)
Klebsiella pneumoniae 89.2% (8) 82.4% (32) 88.2% (32)
Pseudomonas aeruginosa 88.0% (16) 90.0% (4) 98.0% (16)
Serratia spp. 97.0% (1) 94.0% (4) 99.0% (4)
Acinetobacter baumanii 37.0% (>128) 51.0% (>128) 58.0% (>128)
Staphylococcus aureus 95.0% (0.5) 76.0% (>128) 96.0% (8)
Source: Data from Sader HS, et al. Arbekacin activity against contemporary clinical bacteria isolated from patients hospitalized with pneumonia. Antimicrob Agents Chemother,
2015, 59:3263–3270.
Clinically, drug inactivation is the most common mechanism for surfaces also may result in rapid absorption and unexpected toxicity
acquired microbial resistance. The genes encoding aminoglycoside- (i.e., neuromuscular blockade). Intoxication may occur when aminogly-
modifying enzymes are acquired primarily by conjugation and transfer of cosides are applied topically for long periods to large wounds, burns, or
resistance plasmids (see Chapter 52). These enzymes phosphorylate, ade- cutaneous ulcers, particularly if there is renal insufficiency.
nylate, or acetylate specific hydroxyl or amino groups (see Figure 58–1). All the aminoglycosides are absorbed rapidly from intramuscular sites
The ability of these enzymes to attack these groups in differing amino- of injection. Peak concentrations in plasma occur after 30–90 min. These
glycosides explains some of the variability in antimicrobial activity concentrations range from 4 to 12 μg/mL following a 1.5- to 2-mg/kg dose
across the class. Amikacin is a suitable substrate for only a few of these of gentamicin, tobramycin, or netilmicin and from 20 to 35 μg/mL follow-
inactivating enzymes; thus, strains that are resistant to multiple other ing a 7.5-mg/kg dose of amikacin or kanamycin. There is increasing use
aminoglycosides tend to be susceptible to amikacin, particularly amongst of aminoglycosides administered via inhalation, primarily for the man-
gram-negative bacilli. A significant percentage of clinical isolates of agement of patients with cystic fibrosis who have chronic P. aeruginosa
Enterococcus faecalis and Enterococcus faecium are highly resistant to all pulmonary infections (Geller et al., 2002). Amikacin and tobramycin solu-
aminoglycosides (Eliopoulos et al., 1984). Resistance to gentamicin indi- tions for injection have been used, as well as a commercial formulation of
cates cross-resistance to tobramycin, amikacin, kanamycin, and netilmi- tobramycin designed for inhalation.
cin because the inactivating enzyme is bifunctional and can modify all
these aminoglycosides. Owing to differences in the chemical structures Distribution
between streptomycin and other aminoglycosides, the most common Because of their polar nature, the aminoglycosides do not penetrate well
enzyme seen in enterococci does not modify streptomycin, which is inac- into most cells, the CNS, or the eye. Except for streptomycin, there is negli-
tivated by another enzyme. Consequently, gentamicin-resistant strains of gible binding of aminoglycosides to plasma albumin. The apparent volume
enterococci may be susceptible to streptomycin. Intrinsic resistance to of distribution of these drugs is 25% of lean body weight and approximates
aminoglycosides may be caused by failure of the drug to penetrate the the volume of extracellular fluid. The aminoglycosides distribute poorly
cytoplasmic (inner) membrane. Transport of aminoglycosides across the into adipose tissue, which must be considered when using weight-based
cytoplasmic membrane is an active process that depends on oxidative dosing regimens in obese patients.
metabolism. Strictly anaerobic bacteria thus are resistant to these drugs Concentrations of aminoglycosides in secretions and tissues are low
because they lack the necessary transport system. (Panidis et al., 2005). High concentrations are found only in the renal
Missense mutations in Escherichia coli that substitute a single amino cortex and the endolymph and perilymph of the inner ear; the high
acid in a crucial ribosomal protein may prevent binding of streptomycin. concentration in these sites likely contributes to the nephrotoxicity and
Although highly resistant to streptomycin, these strains are not wide- ototoxicity caused by these drugs. As a result of active hepatic secretion,
spread in nature. Similarly, 5% of strains of Pseudomonas aeruginosa concentrations in bile approach 30% of those found in plasma, but this
exhibit such ribosomal resistance to streptomycin. Because ribosomal represents a very minor excretory route for the aminoglycosides. Inflam-
resistance usually is specific for streptomycin, enterococci with ribosomal mation increases the penetration of aminoglycosides into peritoneal and
mutations typically remain sensitive to a combination of penicillin and pericardial cavities. Concentrations of aminoglycosides achieved in CSF
gentamicin in vitro. with parenteral administration usually are subtherapeutic (Kearney and
Aweeka, 1999). Treatment of meningitis with intravenous administration
is generally suboptimal. Intrathecal or intraventricular administration of
ADME aminoglycosides has been used to achieve therapeutic levels in the CNS
but the availability of extended-spectrum cephalosporins has generally
Absorption made this unnecessary.
The aminoglycosides are polar cations and therefore are poorly absorbed Administration of aminoglycosides to women late in pregnancy may
from the GI tract. Less than 1% of a dose is absorbed after either oral or result in accumulation of drug in fetal plasma and amniotic fluid. Strepto-
rectal administration. Nonetheless, long-term oral or rectal administra- mycin and tobramycin can cause hearing loss in children born to women
tion of aminoglycosides may result in accumulation to toxic concentra- who receive the drug during pregnancy. Insufficient data are available
tions in patients with renal impairment. Absorption of gentamicin from regarding the other aminoglycosides; therefore, these agents should be
the GI tract may be increased by GI disease (e.g., ulcers or inflammatory used with caution during pregnancy and only for strong clinical indications
bowel disease). Instillation of these drugs into body cavities with serosal in the absence of suitable alternatives.
Brunton_Ch58_p1039-p1048.indd 1041 08/09/17 5:58 PM
1042 Metabolism and Excretion 35
The aminoglycosides undergo minimal metabolism and are excreted 30
almost entirely by glomerular filtration, achieving urine concentrations
of 50–200 μg/mL. The half-lives of the aminoglycosides in plasma are 25
2–3 h in patients with normal renal function. Because the elimination
CHAPTER 58 AMINOGLYCOSIDES
CP (µg/mL)
of aminoglycosides depends almost entirely on the kidney, a linear rela- 20
tionship exists between the concentration of creatinine in plasma and
the t1/2 of all aminoglycosides in patients with moderately compromised 15
renal function. In anephric patients, the t1/2 varies from 20 to 40 times that
determined in normal individuals. Because the incidence of nephrotoxicity 10
and ototoxicity is likely related to the overall exposure to aminoglycosides, it
5
is critical to reduce the maintenance dose and dosing interval of these drugs Toxicity
in patients with impaired renal function. threshold 0
Although excretion of aminoglycosides is similar in adults and chil- 0 8 16 24
dren older than 6 months, half-lives of aminoglycosides may be prolonged Time (hours)
significantly in the newborn: 8–11 h in the first week of life in newborns
weighing less than 2 kg and about 5 h in those weighing more than 2 kg. Figure 58–3 Comparison of single-dose and divided-dose regimens for gen-
Thus, it is critically important to monitor plasma concentrations of amino- tamicin. In a hypothetical patient, a dose of gentamicin (5.1 mg/kg) is admin-
glycosides during treatment of neonates. Aminoglycoside clearances are istered intravenously as a single bolus (red line) or in three portions, a third
increased and half-lives are reduced in patients with cystic fibrosis (Mann of the dose every 8 h (purple line), such that the total drug administered is
et al., 1985). Larger doses of aminoglycosides may likewise be required in the same in the two cases. The threshold for toxicity (green dashed line) is the
burn patients because of more rapid drug clearance, possibly because of plasma concentration of 2 μg/mL, the maximum recommended for prolonged
drug loss through burn tissue. Aminoglycosides can be removed from the exposure. The single-dose regimen produces a higher plasma concentration
than the regimen given every 8 h; this higher peak provides efficacy that
body by either hemodialysis or peritoneal dialysis.
otherwise might be compromised due to prolonged subthreshold concentra-
Aminoglycosides can be inactivated by various penicillins in vitro and
tions later in the dosing interval or that is provided by the lower peak levels
thus should not be admixed in solution (Blair et al., 1982). Some reports
achieved with the regimen every 8 h. The once-daily regimen also provides a
indicate that this inactivation may occur in vivo in patients with end-stage
13-h period during which plasma concentrations are below the threshold for
renal failure, making monitoring of aminoglycoside plasma concentrations toxicity. The every-8-h regimen, by contrast, provides only three short (~1 h)
even more necessary in such patients. Amikacin appears to be the amino- periods in 24 h during which plasma concentrations are below the threshold
glycoside least affected by this interaction; penicillins with more nonrenal for toxicity. The single high-dose, extended interval is generally preferred for
elimination (such as piperacillin) may be less prone to cause this interaction. aminoglycosides, with a few exceptions (during pregnancy, in neonates, etc.),
as noted in the text.
Dosing and Monitoring
High-dose, extended-interval administration of aminoglycosides is the Steady-state trough concentrations should be less than 1–2 μg/mL for
preferred means of administering aminoglycosides for most indications gentamicin, netilmicin, and tobramycin and less than 10 μg/mL for ami-
and patient populations. Administering higher doses at extended intervals kacin and streptomycin. Peak level goals vary by indication and infection
(i.e., once daily) is likely to be at least equally efficacious and potentially severity, but range from 4 to 8 μg/mL with gentamicin, netilmicin, and
less toxic than administration of divided doses. This dosing strategy takes tobramycin and 20–35 μg/mL for amikacin. Monitoring of aminogly-
advantage of the concentration-dependent activity of aminoglycosides to coside plasma concentrations also is important when using an extend-
achieve maximal initial bacterial killing, and because of the postantibiotic ed-interval dosing regimen. For routine monitoring of extended-interval
effect of aminoglycosides, good therapeutic response can be attained even dosing, a single random concentration obtained 6 to 14 h after the start
when concentrations fall below inhibitory concentrations for a substan- of the infusion can be obtained and plotted against a standard nomo-
tial fraction of the dosing interval. High-dose, extended-interval dosing gram to determine if dosage adjustment is required (Barclay et al., 1999).
schemes for aminoglycosides may also reduce the characteristic oto- and However, the most accurate method for monitoring plasma levels for dose
nephrotoxicity of these drugs. This diminished toxicity is probably due adjustment is to measure the concentration in two plasma samples drawn
to a threshold effect from accumulation of drug in the inner ear or in the several hours apart (e.g., at 2 and 12 h after a dose). The clearance then
kidney. High-dose, extended-interval regimens, despite the higher peak can be calculated and the dose adjusted to achieve the desired target range.
concentration, provide a longer period when concentrations fall below the
threshold for toxicity than does a multiple-dose regimen (compare the two
dosage regimens shown in Figure 58–3). Therapeutic Uses of Aminoglycosides
Populations in which use of the high-dose/extended-interval dosing
Gentamicin, tobramycin, amikacin, and netilmicin can be used inter-
strategy is more controversial include pregnancy, neonates, and pediat-
changeably for the treatment of most of the infections mentioned in this
rics and as combination therapy for endocarditis (Contopoulos-Ioannidis
section. For most indications, gentamicin is preferred because of long
et al., 2004; Knoderer et al., 2003; Nestaas et al., 2005; Ward and Theiler,
experience with its use and its lower cost. Many different types of infec-
2008). In these infections, multiple daily doses (with a lower total daily
tions can be treated successfully with these aminoglycosides; however,
dose) may be preferred because data documenting equivalent safety and
owing to their toxicities, prolonged use should be restricted to the therapy
efficacy of extended-interval dosing are limited. Extended-interval dosing
of life-threatening infections and those for which a less-toxic agent is
is also usually avoided in patients with significant renal dysfunction (i.e.,
contraindicated or less effective.
creatinine clearance < 25 mL/min).
Aminoglycosides frequently are used in combination with a cell wall–
Concentrations of aminoglycosides achieved in plasma after a given
active agent (β-lactam or glycopeptide) for the therapy of serious proven or
dose vary widely amongst patients, and therapeutic drug monitoring is
suspected bacterial infections. The three rationales for this approach are
standard practice (Bartal et al., 2003). For twice- or thrice-daily dosing
regimens, both peak and trough plasma concentrations are determined. • to expand the empiric spectrum of activity of the antimicrobial regimen
The peak concentration documents that the dose produces therapeutic • to provide synergistic bacterial killing
concentrations, while the trough concentration is used to avoid toxicity. • to prevent the emergence of resistance to the individual agents
Brunton_Ch58_p1039-p1048.indd 1042 08/09/17 5:58 PM
Combination therapy is used in infections such as healthcare- endocarditis. For this indication, the administration of gentamicin may 1043
associated pneumonia or sepsis, where multidrug-resistant gram-negative be given as a consolidated once-daily dose. In cases of enterococcal
organisms such as P. aeruginosa, Enterobacter, Klebsiella, and Serratia may endocarditis, concomitant administration of penicillin (or ampicillin)
be causative and the consequences of failing to provide initially active and gentamicin (given as divided doses) for 4–6 weeks is recommended
therapy are dire. The use of aminoglycosides to achieve synergistic bacte- as standard therapy. However, safer alternatives such as ampicillin/
SECTION VII CHEMOTHERAPY OF INFECTIOUS DISEASES
rial killing and improve clinical response is most well established for the ceftriaxone combinations or use of the aminoglycoside for only the first
treatment of endocarditis due to gram-positive organisms, most impor- 2–3 weeks, are gaining favor to limit the risk of toxicity due to prolonged
tantly Enterococcus (Le and Bayer, 2003). Clinical data do not support aminoglycoside administration (Olaison and Schadewitz, 2002). A 2-week
the use of combination therapy for synergistic killing of gram-negative regimen of gentamicin in combination with nafcillin is effective for the
organisms, with the possible exceptions of serious P. aeruginosa infections. treatment of selected cases of staphylococcal tricuspid native-valve endo-
Aminoglycosides (primarily streptomycin and amikacin) are occasionally carditis. For patients with native mitral or aortic valve staphylococcal
used in multidrug regimens for treatment of mycobacterial infections, in endocarditis, the risks of aminoglycoside administration likely outweigh
part because of the need to suppress the emergence of resistant subpopu- the benefits (Cosgrove et al., 2009).
lations during therapy; data do not strongly support this practice for other
bacteria (Bliziotis et al., 2005). Sepsis
Inclusion of an aminoglycoside in an empirical regimen is commonly
Urinary Tract Infections recommended for the febrile patient with neutropenia and for sepsis
Although the spectrum of activity and concentration in the urinary tract when P. aeruginosa is a potential pathogen. However, studies using potent
of aminoglycosides make them well-suited for treatment of urinary tract broad-spectrum β-lactams (e.g., carbapenems and antipseudomonal
infections, less-toxic alternatives are preferred for uncomplicated infec- cephalosporins) have demonstrated no benefit from adding an amino-
tions. However, as strains of E. coli have acquired resistance to β-lactams, glycoside to the regimen unless there is concern that an infection may
trimethoprim-sulfamethoxazole, and fluoroquinolones, use of aminogly- be caused by a multiple-drug-resistant organism (Paul et al., 2003). Thus,
cosides for urinary tract infections may increase. A single intramuscular local susceptibility patterns should be considered when weighing the risks
dose of gentamicin (5 mg/kg) has been effective in uncomplicated infec- and benefits of adjunctive aminoglycoside administration for empiric
tions of the lower urinary tract. A 10- to 14-day course of gentamicin or therapy in patients with sepsis.
tobramycin is an alternative for treatment of pyelonephritis if other agents
cannot be used. Tularemia
Streptomycin (or gentamicin) is the drug of choice for the treatment of
Pneumonia tularemia. Most cases respond to the administration of 1–2 g (15–25 mg/kg)
The organisms that cause community-acquired pneumonia are susceptible to streptomycin per day (in divided doses) for 10–14 days.
broad-spectrum β-lactam antibiotics, macrolides, or a fluoroquinolone, and
usually it is not necessary to add an aminoglycoside. Aminoglycosides are Plague
ineffective for the treatment of pneumonia due to anaerobes or Streptococcus
A 10-day treatment course of streptomycin or gentamicin is recommended
pneumoniae, which are common causes of community-acquired pneumo-
for severe forms of plague (Boulanger et al., 2004).
nia. In hospital-acquired pneumonia where aerobic multidrug-resistant
gram-negative bacilli are frequently causative pathogens, an aminoglycoside
in combination with a β-lactam antibiotic is recommended as standard
Mycobacterial Infections
empiric therapy to increase the likelihood that at least one agent is active Streptomycin is a second-line agent for the treatment of active tubercu-
against the infecting pathogen (American Thoracic Society, 2005). Once it losis, and streptomycin always should be used in combination with at
is established that the β-lactam is active against the causative agent, there is least one or two other drugs to which the causative strain is susceptible.
generally no benefit from continuing the aminoglycoside. Amikacin is another alternative agent for infections due to drug-resistant
Mycobacterium tuberculosis or to other nontuberculous mycobacteria
Meningitis (e.g., M. avium, M. abscessus, M. chelonae).
Availability of third-generation cephalosporins, especially cefotaxime and
ceftriaxone, has reduced the need for treatment with aminoglycosides in
Cystic Fibrosis
most cases of meningitis, except for infections caused by gram-negative Recurrent infections due to multidrug-resistant gram-negative bacilli,
organisms resistant to β-lactam antibiotics (e.g., species of Pseudomonas especially Pseudomonas species, are a hallmark of cystic fibrosis. Amino-
and Acinetobacter). If an aminoglycoside is necessary, direct instillation glycosides are frequently used as therapy during acute exacerbations of
into the CNS is more likely to achieve therapeutic levels than intravenous cystic fibrosis, for which higher-than-standard doses (e.g., 10 mg/kg of
administration. In adults, this can be achieved with 5 mg of a preservative- tobramycin) are frequently employed due to the unusual pharmacok-
free formulation of gentamicin (or equivalent dose of another aminogly- inetics observed in patients with cystic fibrosis. These agents may also
coside) administered intrathecally or intraventricularly once daily. be administered via inhalation between exacerbations to improve lung
function and reduce exacerbation frequency.
Peritonitis
Patients who develop peritonitis as a result of peritoneal dialysis may be Topical Applications
treated with aminoglycoside diluted into the dialysis fluid to a concentra- Aminoglycosides, especially neomycin and paromomycin, may be
tion of 4–8 mg/L for gentamicin, netilmicin, or tobramycin or 6–12 mg/L employed as topical agents in skin and mucous membrane infections.
for amikacin. Intravenous or intramuscular administration of drug is Oral administration of aminoglycosides may be employed as “bowel prep”
unnecessary because serum and peritoneal fluid will equilibrate rapidly. prior to surgical procedures or as “selective digestive decontamination” to
reduce the risk of ventilator-associated pneumonia.
Bacterial Endocarditis
“Synergistic” or low-dose gentamicin (3 mg/kg/d) in combination with a
penicillin or vancomycin has been recommended in certain circumstances
Adverse Effects of Aminoglycosides
for treatment of bacterial endocarditis due to certain gram-positive organ- All aminoglycosides have the potential to produce reversible and irre-
isms. Penicillin and gentamicin in combination are effective as a short- versible vestibular, cochlear, and renal toxicity and neuromuscular
course (i.e., 2-week) regimen for uncomplicated native-valve streptococcal blockade.
Brunton_Ch58_p1039-p1048.indd 1043 08/09/17 5:58 PM
1044 Ototoxicity than divided-dose approaches (see Figure 58–3). Neomycin, which con-
centrates to the greatest degree, is highly nephrotoxic in human beings
Vestibular and auditory dysfunction can follow the administration of
and should not be administered systemically. Streptomycin does not con-
any of the aminoglycosides (Guthrie, 2008). Aminoglycoside-induced
centrate in the renal cortex and is the least nephrotoxic. Drugs such as
ototoxicity may result in irreversible, bilateral, high-frequency hearing
amphotericin B, vancomycin, angiotensin-converting enzyme inhibitors,
loss or vestibular hypofunction. Degeneration of hair cells and neurons
CHAPTER 58 AMINOGLYCOSIDES
cisplatin, and cyclosporine may potentiate aminoglycoside-induced neph-
in the cochlea correlates with the loss of hearing. Accumulation within the
rotoxicity (Wood et al., 1986).
perilymph and endolymph occurs predominantly when aminoglycoside
concentrations in plasma are high. Diffusion back into the bloodstream
is slow; the half-lives of the aminoglycosides are five to six times longer
Neuromuscular Blockade
in the otic fluids than in plasma. Drugs such as ethacrynic acid and furo- Acute neuromuscular blockade and apnea have been attributed to the
semide potentiate the ototoxic effects of the aminoglycosides in animals, aminoglycosides; patients with myasthenia gravis are particularly sus-
but data from humans implicating furosemide are less convincing (Smith ceptible. The order of decreasing potency for blockade is neomycin,
and Lietman, 1983). kanamycin, amikacin, gentamicin, and tobramycin. In humans, neuro-
Streptomycin and gentamicin produce predominantly vestibular effects, muscular blockade generally has occurred after intrapleural or intrap-
whereas amikacin, kanamycin, and neomycin primarily affect auditory eritoneal instillation of large doses of an aminoglycoside; however, the
function; tobramycin affects both equally. The incidence of ototoxicity is reaction can follow intravenous, intramuscular, and even oral admin-
difficult to determine. Audiometric data suggest that the incidence could istration of these agents. Most episodes have occurred in association
be as high as 25% (Brummett and Morrison, 1990). The incidence of ves- with anesthesia or the administration of other neuromuscular blocking
tibular toxicity is particularly high in patients receiving streptomycin; agents. Neuromuscular blockade may be reversed by intravenous admin-
nearly 20% of individuals who received 500 mg twice daily for 4 weeks for istration of a Ca2+ salt.
enterococcal endocarditis developed clinically detectable irreversible ves- Aminoglycosides may inhibit prejunctional release of acetylcholine
tibular damage. Because the initial symptoms may be reversible, patients while also reducing postsynaptic sensitivity to the transmitter, but Ca2+
receiving high doses or prolonged courses of aminoglycosides should be can overcome this effect, and the intravenous administration of a calcium
monitored carefully for ototoxicity; however, deafness may occur several salt is the preferred treatment of this toxicity (Sarkar et al., 1992). Inhib-
weeks after therapy is discontinued. itors of acetylcholinesterase (e.g., edrophonium and neostigmine) also
A high-pitched tinnitus often is the first symptom of cochlear toxicity. have been used with varying degrees of success.
If the drug is not discontinued, auditory impairment may develop after a
few days. The tinnitus may persist for several days to 2 weeks after ther- Other Adverse Effects
apy is stopped. Because perception of sound in the high-frequency range In general, the aminoglycosides have little allergenic potential. Rare
(outside the conversational range) is lost first, the affected individual is not hypersensitivity reactions—including skin rashes, eosinophilia, fever,
always aware of the difficulty, and it will not be detected except by careful blood dyscrasias, angioedema, exfoliative dermatitis, stomatitis, and ana-
audiometric examination. If the hearing loss progresses, the lower sound phylactic shock—have been reported as cross-hypersensitivity amongst
ranges are affected. drugs in this class. Aminoglycosides appear to be less commonly associ-
Amongst patients experiencing vestibular toxicity, moderately intense ated with superinfection due to Clostridium difficile than other classes of
headache lasting 1–2 days may precede the onset of labyrinthine dysfunction. antibacterials.
This is followed immediately by an acute stage in which nausea, vomiting,
and difficulty with equilibrium develop and persist for 1–2 weeks. Prominent
symptoms include vertigo in the upright position, inability to perceive Pharmacological Properties of Individual
termination of movement (“mental past-pointing”), and difficulty in Aminoglycosides
sitting or standing without visual cues. The acute stage ends suddenly
and is followed by chronic labyrinthitis, in which the patient has difficulty Gentamicin
when attempting to walk or make sudden movements; ataxia is the most
Gentamicin is an important agent for the treatment of many serious
prominent feature. The chronic phase persists for about 2 months. Recovery
gram-negative bacillary infections. It is the aminoglycoside of first choice
from this phase may require 12–18 months, and most patients have some
because of its lower cost and reliable activity against all but the most
permanent residual damage. Early discontinuation of the drug may permit
resistant gram-negative aerobes. Gentamicin preparations are available
recovery before irreversible damage of the hair cells.
for parenteral, ophthalmic, and topical administration. The typical rec-
ommended intramuscular or intravenous dose of gentamicin sulfate when
Nephrotoxicity used for the treatment of known or suspected gram-negative organisms
Approximately 8%–26% of patients who receive an aminoglycoside for as a single agent or in combination therapy for adults with normal renal
several days develop mild renal impairment that is almost always revers- function is 5–7 mg/kg daily given over 30–60 min. For patients with
ible. The toxicity results from accumulation and retention of aminogly- renal dysfunction, the interval may be extended. For patients who are
coside in the proximal tubular cells. The initial manifestation of damage at not candidates for extended-interval dosing, a loading dose of 2 mg/kg
this site is excretion of enzymes of the renal tubular brush border followed and then 3–5 mg/kg per day, given as divided doses every 8–12 h, are
by mild proteinuria and the appearance of hyaline and granular casts. recommended. Dosages at the upper end of this range may be required to
The glomerular filtration rate is reduced after several additional days. The achieve therapeutic levels for trauma or burn patients, those with septic
nonoliguric phase of renal insufficiency is thought to be due to the effects shock, patients with cystic fibrosis, and others in whom drug clearance is
of aminoglycosides on the distal portion of the nephron with a reduced more rapid or volume of distribution is larger than normal.
sensitivity of the collecting duct epithelium to vasopressin. Although Several dosage schedules have been suggested for newborns and infants:
severe acute tubular necrosis may occur rarely, the most common sig- 3 mg/kg once daily for preterm newborns less than 35 weeks of gesta-
nificant finding is a mild rise in plasma creatinine. The impairment in tion; 4 mg/kg once daily for newborns more than 35 weeks of gestation;
renal function is almost always reversible because the proximal tubular 5 mg/kg daily in two divided doses for neonates with severe infections;
cells have the capacity to regenerate (Lietman and Smith, 1983). Toxicity and 2–2.5 mg/kg every 8 h for children up to 2 years of age. Peak plasma
correlates with the total amount of drug administered and with longer concentrations range from 4 to 10 mg/mL (dosing: 1.7 mg/kg every 8 h)
courses of therapy (de Jager and van Altena, 2002). High-dose, extend- and 16–24 mg/mL (extended-interval dosing: 5 mg/kg once daily).
ed-interval dosing approaches lead to less nephrotoxicity at the same It should be emphasized that the recommended doses of gentamicin
level of total drug exposure (as measured by the area under the curve) do not always yield desired concentrations. Periodic determinations of
Brunton_Ch58_p1039-p1048.indd 1044 08/09/17 5:58 PM
the plasma concentration of aminoglycosides are recommended strongly. Streptomycin 1045
Gentamicin is absorbed slowly when it is applied topically in an ointment
Streptomycin is used for the treatment of certain unusual infections, gen-
and somewhat more rapidly when it is applied as a cream. When the anti-
erally in combination with other antimicrobial agents. It generally is less
biotic is applied to large areas of denuded body surface, as may be the case
active than other members of the class against aerobic gram-negative rods.
in burn patients, plasma concentrations can reach 4 μg/mL, and 2%–5%
SECTION VII CHEMOTHERAPY OF INFECTIOUS DISEASES
The combination of penicillin G (bacteriostatic against enterococci) and
of the drug may appear in the urine.
streptomycin is effective as bactericidal therapy for enterococcal endo-
carditis, although gentamicin is generally preferred for its lesser toxicity.
Tobramycin Streptomycin should be used instead of gentamicin when the strain is
The antimicrobial activity, pharmacokinetic properties, and toxicity pro- resistant to the latter and has demonstrable susceptibility to streptomycin,
file of tobramycin are similar to those of gentamicin. Tobramycin may be which may occur because the enzymes that inactivate these two amino-
given intramuscularly, intravenously, or by inhalation. Tobramycin also is glycosides are different.
available in ophthalmic ointments and solutions. The superior activity of Streptomycin may be administered by deep intramuscular injec-
tobramycin against P. aeruginosa makes it the preferred aminoglycoside tion or intravenously. Intramuscular injection may be painful, with a
for treatment of serious infections known or suspected to be caused by this hot tender mass developing at the site of injection. The dose range of
organism, typically in combination with an antipseudomonal β-lactam streptomycin for most indications is 15–25 mg/kg daily or in divided
antibiotic. In contrast to gentamicin, tobramycin shows poor activity in doses twice daily. During initial therapy for tuberculosis, it is frequently
combination with penicillin against many strains of enterococci. Most administered as a 1000-mg single daily dose, resulting in peak serum
strains of E. faecium are highly resistant. Tobramycin is ineffective against concentrations of about 50–60 and 15–30 μg/mL, and trough concentra-
mycobacteria. Dosages and serum concentrations are identical to those tions of less than 1 and 5–10 μg/mL, respectively. The dosing frequency
for gentamicin. may be reduced to two or three times a week after the initial phase of
tuberculosis treatment.
Amikacin Streptomycin has been replaced by gentamicin for most indica-
The spectrum of antimicrobial activity of amikacin is the broad- tions because the toxicity of gentamicin is primarily renal and revers-
est of the group. Because of its resistance to many of the aminogly- ible, whereas that of streptomycin is vestibular and irreversible. The
coside-inactivating enzymes, amikacin has a special role for the initial administration of streptomycin may produce dysfunction of the optic
treatment of serious nosocomial gram-negative bacillary infections in nerve, including scotomas, presenting as enlargement of the blind spot.
hospitals where resistance to gentamicin and tobramycin has become a Amongst the less-common toxic reactions to streptomycin is peripheral
significant problem. Amikacin is active against most strains of Serratia, neuritis.
Proteus, and P. aeruginosa as well as most strains of Klebsiella, Enterobac-
ter, and E. coli that are resistant to gentamicin and tobramycin. Most resis- Neomycin
tance to amikacin is found amongst strains of Acinetobacter, Providencia, Neomycin is a broad-spectrum antibiotic. Susceptible microorgan-
and Flavobacterium and strains of Pseudomonas other than P. aeruginosa; isms usually are inhibited by concentrations of 10 μg/mL or less.
these all are unusual pathogens. Amikacin is less active than gentamicin Gram-negative species that are highly sensitive are E. coli, Enterobacter
against enterococci and should not be used for this organism. Amikacin aerogenes, Klebsiella pneumoniae, and Proteus vulgaris. Gram-positive
is not active against the majority of gram-positive anaerobic bacteria. It microorganisms that are inhibited include S. aureus and E. faecalis.
is active against M. tuberculosis, including streptomycin-resistant strains Mycobacterium tuberculosis also is sensitive to neomycin. Strains of
and atypical mycobacteria. P. aeruginosa are resistant to neomycin. Neomycin sulfate is available
The recommended dose of amikacin is 15 mg/kg/d as a single daily for topical and oral administration. Neomycin currently is available
dose or divided into two or three equal portions, which must be reduced in many brands of creams, ointments, and other products alone and
for patients with renal failure. The drug is absorbed rapidly after intra- in combination with polymyxin, bacitracin, other antibiotics, and a
muscular injection, and peak concentrations in plasma approximate variety of corticosteroids.
20 μg/mL after injection of 7.5 mg/kg. The concentration 12 h after a Neomycin is used widely for topical application in a variety of infec-
7.5-mg/kg dose is 5–10 μg/mL. A 15-mg/kg once-daily dose produces tions of the skin and mucous membranes. The oral administration of
peak concentrations of 50–60 μg/mL and a trough of less than 1 μg/mL. neomycin (usually in combination with erythromycin base) has been
For treatment of mycobacterial infections, thrice-weekly dosing sched- employed primarily for “preparation” of the bowel for surgery. Orally
ules are used, with doses up to 25 mg/kg (Peloquin et al., 2004). As with administered neomycin is poorly absorbed from the GI tract—about
the other aminoglycosides, amikacin causes ototoxicity, hearing loss, and 97% of an oral dose of neomycin is not absorbed and is eliminated
nephrotoxicity. unchanged in the feces. The portion that is absorbed is excreted by the
kidney; a total daily intake of 10 g for 3 days yields a blood concentration
Netilmicin below that associated with systemic toxicity if renal function is normal.
Netilmicin (not marketed in the U.S.) is similar to gentamicin and Neomycin and polymyxin B have been used for irrigation of the bladder
tobramycin in its pharmacokinetic properties and dosage (Panwalker to prevent bacteriuria and bacteremia associated with indwelling cathe-
et al., 1978). Its antibacterial activity is broad against aerobic ters. For this purpose, 1 mL of a preparation containing 40 mg neomycin
gram-negative bacilli. Like amikacin, it is not metabolized by most of and 200,000 units polymyxin B per milliliter is diluted in 1 L of 0.9%
the aminoglycoside-inactivating enzymes; thus, it may be active against sodium chloride solution and is used for continuous irrigation of the
certain bacteria that are resistant to gentamicin (with the exception of urinary bladder through appropriate catheter systems. The bladder is
resistant enterococci). Netilmicin is useful for the treatment of serious irrigated at the rate of 1 L every 24 h.
infections owing to susceptible Enterobacteriaceae and other aerobic Hypersensitivity reactions, primarily skin rashes, occur in 6%–8% of
gram-negative bacilli. The recommended dose of netilmicin for com- patients when neomycin is applied topically. The most important toxic
plicated urinary tract infections in adults is 1.5–2 mg/kg every 12 h. effects of neomycin are ototoxicity and nephrotoxicity; as a consequence,
For other serious systemic infections, a total daily dose of 4–7 mg/kg is the drug is no longer available for parenteral administration. Neuromus-
administered as a single dose or two to three divided doses. Children cular blockade with respiratory paralysis also has occurred after irriga-
should receive 3–7 mg/kg/d in two to three divided doses; neonates tion of wounds or serosal cavities. Individuals treated with 4–6 g/d of the
receive 3.5–5 mg/kg/d as a single daily dose. The t1/2 for elimination drug by mouth sometimes develop a sprue-like syndrome with diarrhea,
is usually 2–2.5 h in adults and increases with renal insufficiency. steatorrhea, and azotorrhea. Overgrowth of yeasts in the intestine also
Netilmicin may produce ototoxicity and nephrotoxicity. may occur.
Brunton_Ch58_p1039-p1048.indd 1045 08/09/17 5:58 PM
1046 Paromomycin of intestinal cryptosporidiosis and giardiasis, which can be particularly
challenging to treat in immunocompromised patients. Orally admin-
Paromomycin (also known as aminosidine) is an aminoglycoside that
istered paromomycin is associated with dose-related gastrointestinal
is structurally related to neomycin. It has antibacterial activity simi-
toxicity, including nausea, abdominal pain, and diarrhea. A topical
lar to other aminoglycosides but has particularly notable antiparasitic
formulation is also used internationally for treatment of cutaneous leish-
activity. Parasites that are usually susceptible to paromomycin include
CHAPTER 58 AMINOGLYCOSIDES
maniasis (Ben Salah et al., 2013).
Leishmania spp., Entamoeba histolytica, Giardia lamblia, and Cryptospo-
ridium parvum. Internationally, the parenteral form is used as a treatment Kanamycin
of infections due to Leishmania spp. (visceral leishmaniasis), although
this formulation is not available in the U.S. (Sundar et al., 2007). Like Kanamycin is amongst the most toxic aminoglycosides, and there are few
other aminoglycosides, it has poor systemic absorption when adminis- indications for its use. Its primary remaining indication is for treatment of
tered orally; this characteristic is exploited to achieve high luminal con- extensively drug-resistant tuberculosis; even in this condition, less-toxic
centrations in the treatment of intestinal parasitic diseases. It is available alternatives are generally preferred.
as oral capsules and indicated for treatment of intestinal amebiasis at a Acknowledgment: Henry F. Chambers contributed to this chapter in recent
dose of 25–35 mg/kg/d in three divided doses. It is also used for treatment editions of this book. We have retained some of his text in the current edition.
Drug Facts for Your Personal Formulary: Aminoglycosides
Drug Therapeutic Uses Clinical Pharmacology and Tips
Aminoglycosides—Inhibitors of Bacterial Protein Synthesis
General: Bactericidal, no GI absorption (<1%), oral administration used only for bowel decontamination or intestinal parasites, poor
CSF penetration, renal elimination, nephrotoxicity, ototoxicity (cochlear and vestibular), neuromuscular blockade
Gentamicin (IV) • UTI • Good activity vs. Enterobacteriaceae, Pseudomonas
• Peritonitis • Some activity vs. Neisseria, Haemophilus,
• Endocarditis in combination with a cell-wall Moraxella
active agent • Synergistic activity when combined with a cell-wall
• Plague agent against many organisms
• Tularemia • Vestibular > cochlear toxicity
• Toxicity primarily renal and reversible
Tobramycin (IV, inhalation) • UTI • Similar to gentamicin, with better activity against
• Lung infections, including cystic fibrosis Pseudomonas aeruginosa
exacerbations • Cochlear ≈ vestibular toxicity
• Nosocomial sepsis of unknown origin
Amikacin (IV) • UTI • Similar to tobramycin, with activity against
• Lung infections, including cystic fibrosis some gram-negative bacilli resistant to other
exacerbations aminoglycosides
• Nosocomial sepsis of unknown origin • Activity against a variety of mycobacteria
• Mycobacterial infections • Cochlear > vestibular toxicity
Streptomycin (IV) • Endocarditis in combination with a cell-wall • Similar to gentamicin, with activity against some
active agent gentamicin-resistant enterococci
• Tuberculosis • Activity against Mycobacterium tuberculosis
• Plague • Vestibular > cochlear toxicity
• Tularemia • Vestibular toxicity is irreversible
Neomycin (PO, topical; urologic irrigation) • Minor skin infections • Similar activity to gentamicin but only used topically,
• Bowel preparation prior to intra-abdominal surgery not systemically
• Bladder irrigation • Can cause skin rash
Paromomycin (PO, IM, topical) • Cryptosporidia infection • Diarrhea, nausea, vomiting
• Intestinal amebiasis • IM use for visceral leishmaniasis
• Leishmaniasis • Topical use for cutaneous leishmaniasis
Blair DC, et al. Inactivation of amikacin and gentamicin by carbenicillin
Bibliography in patients with end-stage renal failure. Antimicrob Agents Chemother,
American Thoracic Society. Guidelines for the management of adults with 1982, 22:376–379.
hospital-acquired, ventilator-associated, and healthcare-associated Bliziotis IA, et al. Effect of aminoglycoside and beta-lactam combination
pneumonia. Am J Resp Crit Care Med, 2005, 171:388–416. therapy versus beta-lactam monotherapy on the emergence of
Barclay ML, et al. Once-daily aminoglycoside therapy: is it less toxic antimicrobial resistance: a meta-analysis of randomized, controlled
than multiple daily doses and how should it be monitored? Clin trials. Clin Infect Dis, 2005, 41:149–158.
Pharmacokinet, 1999, 36:89–98. Boulanger LL, et al. Gentamicin and tetracyclines for the treatment of
Bartal C, et al. Pharmacokinetic dosing of aminoglycosides: a controlled human plague: review of 75 cases in New Mexico, 1985–1999. Clin
trial. Am J Med, 2003, 114:194–198. Infect Dis, 2004, 38:663–669.
Ben Salah A, et al. Topical paromomycin with or without gentamicin for Brummett RE, Morrison RB. The incidence of aminoglycoside antibiotic-
cutaneous leishmaniasis. N Engl J Med, 2013, 368:524–532. induced hearing loss. Arch Otolaryngol Head Neck Surg, 1990, 116:406–410.
Brunton_Ch58_p1039-p1048.indd 1046 08/09/17 5:58 PM
You might also like
- The AminoglycosidesDocument8 pagesThe AminoglycosidesVangenique Nieves AgrudaNo ratings yet
- Risk Management PlanDocument4 pagesRisk Management PlanHannah BalucaNo ratings yet
- DP On AglDocument12 pagesDP On AglDeepikaNo ratings yet
- Protein Synthesis Inhibitors - Second SessionalDocument10 pagesProtein Synthesis Inhibitors - Second Sessionaljhanavi rajeshNo ratings yet
- Protein Systhesis InhibitorsDocument31 pagesProtein Systhesis InhibitorsCabaddu AngieNo ratings yet
- Aminoglycoside Antibiotics: Mechanism of ActionDocument9 pagesAminoglycoside Antibiotics: Mechanism of Actionprabhakaran payamNo ratings yet
- Chapter 45 - Aminoglycosides SpectinomycinDocument6 pagesChapter 45 - Aminoglycosides SpectinomycinrheaasarenNo ratings yet
- AminoglycosidesDocument20 pagesAminoglycosidesPrasad SangishettyNo ratings yet
- AminoglycosideDocument22 pagesAminoglycosideRabi ShahNo ratings yet
- (BACTE-LEC) 8. Antimicrobial Susceptibility TestingDocument6 pages(BACTE-LEC) 8. Antimicrobial Susceptibility TestingCarlo CataagNo ratings yet
- Aminoglycoside: Systemic AminoglycosidesDocument47 pagesAminoglycoside: Systemic AminoglycosidesPawan PatelNo ratings yet
- Protein Synthesis InhibitorsDocument35 pagesProtein Synthesis InhibitorsHarsha MaheshwariNo ratings yet
- Aminoglycosides (17.07.2017)Document44 pagesAminoglycosides (17.07.2017)Habibul Kowser (Rishat)No ratings yet
- Streptomycin PDFDocument7 pagesStreptomycin PDFHdjdNo ratings yet
- Antibiotics GuideDocument33 pagesAntibiotics Guidej7qs46h8wrNo ratings yet
- Antibiotics Chart For MicrobiologyDocument5 pagesAntibiotics Chart For MicrobiologyTrevorNo ratings yet
- Pathophysiology Assignment 5Document5 pagesPathophysiology Assignment 5venkatasaiyuvarajNo ratings yet
- Inhibitors of Protein SynthesisDocument2 pagesInhibitors of Protein Synthesiselsayed barhomeNo ratings yet
- Protein Synthesis Inhibitors: Tasneem SmeratDocument78 pagesProtein Synthesis Inhibitors: Tasneem Smeratansam hirbaweNo ratings yet
- AMINOGLYCOSIDESDocument45 pagesAMINOGLYCOSIDESAbdullah EmadNo ratings yet
- Amino Glyc o SidesDocument27 pagesAmino Glyc o SidesAyesha AkbarNo ratings yet
- Table: Selected Antibacterial Antibiotics Antibiotics THAT . Mechanism of Action Names of Drugs Notes and ProblemsDocument4 pagesTable: Selected Antibacterial Antibiotics Antibiotics THAT . Mechanism of Action Names of Drugs Notes and ProblemsTJNo ratings yet
- 46 Aminoglycosides-2023Document16 pages46 Aminoglycosides-2023deo okiaNo ratings yet
- HandoutDocument17 pagesHandoutAlmalyn TimbayanNo ratings yet
- QuinolonDocument10 pagesQuinolonkimanh phạmNo ratings yet
- Aminoglikosida Farkol 2 2023Document20 pagesAminoglikosida Farkol 2 2023ghazialghifariNo ratings yet
- Aminoglycosides: Amlan GangulyDocument14 pagesAminoglycosides: Amlan GangulyAli Akand AsifNo ratings yet
- Aminoglycosides, Macrolides, Chloramphenicol, LincomycinsDocument7 pagesAminoglycosides, Macrolides, Chloramphenicol, LincomycinsMello DiaxNo ratings yet
- ANTIBIOTIKA REVIEW-2021 enDocument45 pagesANTIBIOTIKA REVIEW-2021 enBeby CantikqNo ratings yet
- P'cology of Amin & Spect (2022-P)Document42 pagesP'cology of Amin & Spect (2022-P)Tewodros TeshomeNo ratings yet
- Aminoglycosides.: Chapter No. 02 Week # 07Document2 pagesAminoglycosides.: Chapter No. 02 Week # 07Alee Iz HarNo ratings yet
- AntifungalagentsDocument52 pagesAntifungalagentsspriyansh202No ratings yet
- Inhibitor of Bacterial Protein SynthesisDocument83 pagesInhibitor of Bacterial Protein SynthesisNdayisaba CorneilleNo ratings yet
- Aminoglycoside: From Wikipedia, The Free EncyclopediaDocument11 pagesAminoglycoside: From Wikipedia, The Free Encyclopediaawajahat100% (3)
- ANTITUBERCULOUSDocument11 pagesANTITUBERCULOUSScribdTranslationsNo ratings yet
- MicrobialsDocument5 pagesMicrobialsMARIEMIL FOLLOSONo ratings yet
- CH 10 Microbiology Lecture PresentationDocument56 pagesCH 10 Microbiology Lecture PresentationRiriNo ratings yet
- Bacteriostatic Agents: Drugs Which Bind To The 50s Ribosomal UnitDocument3 pagesBacteriostatic Agents: Drugs Which Bind To The 50s Ribosomal UnitJoshua Trinidad100% (1)
- Amr SeminarDocument48 pagesAmr SeminarSwijalNo ratings yet
- Antibióticos en NefrotoxicidadDocument15 pagesAntibióticos en NefrotoxicidadHilma BermudezNo ratings yet
- Ch12 Part1Document15 pagesCh12 Part1lasanders601No ratings yet
- Antibacterial Class 03Document80 pagesAntibacterial Class 03tsegaab yosephNo ratings yet
- Department of Pharmacology: Prof. Dr. Asya RehmanDocument15 pagesDepartment of Pharmacology: Prof. Dr. Asya RehmanGareth BaleNo ratings yet
- Antibiotic Susceptibility TestingDocument46 pagesAntibiotic Susceptibility Testingditta antNo ratings yet
- AntimikrobaDocument50 pagesAntimikrobaarisita firmanNo ratings yet
- H01033 Hoá Dư C 1 Lecture 4 AminoglycosidesDocument27 pagesH01033 Hoá Dư C 1 Lecture 4 AminoglycosidesTor NgânNo ratings yet
- 6 - Lectins and PolypeptidesDocument6 pages6 - Lectins and PolypeptidesJonafe JuntillaNo ratings yet
- Notes MacrolidesDocument6 pagesNotes MacrolidesAlessa Marie BadonNo ratings yet
- TuberculosisDocument2 pagesTuberculosisElle ReyesNo ratings yet
- Pharmacology of AntimicrobialsDocument31 pagesPharmacology of AntimicrobialsJosette LeyvaNo ratings yet
- Antimicrobal Drugs #Dental 1Document30 pagesAntimicrobal Drugs #Dental 1ggNo ratings yet
- Antimicrobial Agents and Vaccinations - UpdatedDocument62 pagesAntimicrobial Agents and Vaccinations - Updatedjohn tsuiNo ratings yet
- Aminoglycosides: Official Reprint From Uptodate ©2021 Uptodate, Inc. And/Or Its Affiliates. All Rights ReservedDocument17 pagesAminoglycosides: Official Reprint From Uptodate ©2021 Uptodate, Inc. And/Or Its Affiliates. All Rights ReservedVanessa GomesNo ratings yet
- Tonghop ProDocument16 pagesTonghop Prokimanh phạmNo ratings yet
- General Characteristics of Antimicrobial Drug1Document5 pagesGeneral Characteristics of Antimicrobial Drug1vishuop061No ratings yet
- Antimicrobial AgentsDocument2 pagesAntimicrobial AgentsFahima RahmanNo ratings yet
- Haematology-Oncology McqsDocument80 pagesHaematology-Oncology McqsKarim MohamedNo ratings yet
- AminoglycosidesDocument1 pageAminoglycosidesmariam ibrahimNo ratings yet
- Antibiotic 1Document13 pagesAntibiotic 1ريدان عياشNo ratings yet
- 8 - AntibioticsDocument8 pages8 - AntibioticsArshad AbbasNo ratings yet
- 7 Natural Remedies For Epstein Barr VirusDocument40 pages7 Natural Remedies For Epstein Barr VirusalbinutaNo ratings yet
- Rai Work Experience SheetDocument2 pagesRai Work Experience Sheetmark langcayNo ratings yet
- Concepts of ProsthoDocument22 pagesConcepts of ProsthoKirti SharmaNo ratings yet
- 50 Items HADocument7 pages50 Items HAToni Marie Buenconsejo PunzalanNo ratings yet
- Tristill - Brochure - March 2017 New A4 Three FoldDocument2 pagesTristill - Brochure - March 2017 New A4 Three FoldLesley HollardNo ratings yet
- Case Study On PneumoniaDocument12 pagesCase Study On PneumoniaRintu JenaNo ratings yet
- The Structure and Function of The Cardiovascular SystemDocument28 pagesThe Structure and Function of The Cardiovascular SystemShamaeNogaNo ratings yet
- The Dorsal Skinfold Chamber: A Versatile Tool For Preclinical Research in Tissue Engineering and Regenerative MedicineDocument14 pagesThe Dorsal Skinfold Chamber: A Versatile Tool For Preclinical Research in Tissue Engineering and Regenerative MedicineTanvir DaphedarNo ratings yet
- Extracts of Justicia Wynaadensis LeavesDocument9 pagesExtracts of Justicia Wynaadensis LeavesPawan KumarNo ratings yet
- SBL 4 - Xerostomia - FINALDocument16 pagesSBL 4 - Xerostomia - FINALCara ChanNo ratings yet
- HCD2Document2 pagesHCD2fwijaya888No ratings yet
- Nursing Practice II EdgeworthDocument9 pagesNursing Practice II EdgeworthRichard Ines Valino100% (1)
- Communicable DiseaseDocument38 pagesCommunicable DiseaseDrShrikant JahagirdarNo ratings yet
- AF SeedingDocument6 pagesAF SeedingdonkeyendutNo ratings yet
- Chemical Product and Company Information: Material Safety Data SheetDocument5 pagesChemical Product and Company Information: Material Safety Data Sheetkirandas_mullasseryNo ratings yet
- Narrative Report - NSTP 2nd Sem Chapter 3Document1 pageNarrative Report - NSTP 2nd Sem Chapter 3Clint Kove YtemNo ratings yet
- Who Killed Christopher Goodman? by Allan Wolf Chapter SamplerDocument21 pagesWho Killed Christopher Goodman? by Allan Wolf Chapter SamplerCandlewick Press50% (2)
- Daftar PustakaDocument3 pagesDaftar PustakamemeeeyyyNo ratings yet
- Sample Academic Essay in APA Style (6 Edition)Document3 pagesSample Academic Essay in APA Style (6 Edition)Papa YawNo ratings yet
- N410 SyllabusDocument20 pagesN410 SyllabusTwobucktinNo ratings yet
- When To See A DoctorDocument2 pagesWhen To See A DoctorPooja ChapdiNo ratings yet
- نموذج انقاذ حياة الجديدDocument2 pagesنموذج انقاذ حياة الجديدraad_alghamdi_10% (1)
- HIV Transmission Through Breastfeeding: A Review of Available EvidenceDocument33 pagesHIV Transmission Through Breastfeeding: A Review of Available EvidenceJessica RamosNo ratings yet
- Psychophysiological Disorders - Kanna KDocument16 pagesPsychophysiological Disorders - Kanna KKannan KrishnamurthyNo ratings yet
- Formatif NBSSDocument17 pagesFormatif NBSSCYNTHIA ARISTANo ratings yet
- Escape From The Jade Colossus: by Bruce R. CordellDocument30 pagesEscape From The Jade Colossus: by Bruce R. CordellJuh Freitas38% (8)
- CGHS Enclosure 1Document77 pagesCGHS Enclosure 1bhupendrapawar279No ratings yet
- Paediatric Proton Therapy (Thomas 2019)Document9 pagesPaediatric Proton Therapy (Thomas 2019)Milda InayahNo ratings yet
- Surgical Hand Hygiene PosterDocument2 pagesSurgical Hand Hygiene PosterIrenne WibowoNo ratings yet