Professional Documents
Culture Documents
First Page
First Page
Uploaded by
shihen0 ratings0% found this document useful (0 votes)
2 views1 pageOriginal Title
first-page
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
2 views1 pageFirst Page
First Page
Uploaded by
shihenCopyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 1
844 SECTION X Liver Affected by Other Conditions or Diseases
56
Drug-Induced Liver Injury
HARSHAD DEVARBHAVI, H ERBERT L. BONKOVSKY,
MARK RUSSO, AND NAGA C HALASANI
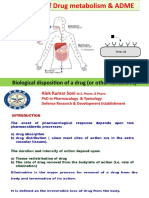
A B B R E V I AT I O N S The multistep process for the metabolism of drugs and chemi-
cals is summarized in Fig. 56-1. Most such chemicals are ingested
ADR adverse drug reaction orally and absorbed, chiefly in the proximal part of the small
ALF acute liver failure intestine. Some of them undergo initial metabolism within the
ALT alanine aminotransferase gastrointestinal tract. Parent compounds and/or metabolites then
AP alkaline phosphatase enter the splanchnic blood, from which they are eventually deliv-
AST aspartate aminotransferase ered by the portal circulation to the liver. Depending on the
ATP adenosine triphosphate xenobiotic in question, cells within the liver absorb a variable
BHDS botanicals, herbal products, and dietary supplements proportion of the compound. This initial removal of compounds
CI confidence interval from the portal blood, which is the chief blood supply to the liver,
CYP cytochrome P450 is called the first-pass effect of the liver.
DILI drug-induced liver injury The uptake into liver cells occurs primarily, but not solely, into
DILIN Drug-Induced Liver Injury Network hepatocytes. During the past several years a number of transport-
FDA Food and Drug Administration ers of cations and anions have been described and have been
GSH glutathione identified as important in the uptake of endogenous chemicals
HAART highly active antiretroviral therapy and xenobiotics (drugs, foreign chemicals) by liver cells. Once
HLA human leukocyte antigen inside hepatocytes, these chemicals undergo further intracellular
MMPT mitochondrial membrane permeability transition binding and transport. The intracellular mechanisms responsible
NAPQI N-acetyl-p-benzoquinoneimine for such transport are less well understood. It is likely that highly
NARTI nucleoside analog reverse transcriptase inhibitor lipophilic compounds dissolve readily into the membranes of cells
NSAID nonsteroidal antiinflammatory drugs and diffuse widely and quickly within and across such mem-
PI protease inhibitor branes. In contrast, more hydrophilic compounds require protein
SOS sinusoidal obstruction syndrome binding and other means for transport.
ULN upper limit of normal As shown in Fig. 56-1, many (but certainly not all) drugs and
chemicals require an initial oxidation reaction, termed phase I
metabolism. The most common example is hydroxylation cata-
lyzed by 1 of the 57 varieties of cytochrome P450 (CYP). Many
Introduction of these CYPs are found in hepatocytes, and they perform hydrox-
ylation reactions in concert with NADPH (as a source of elec-
In humans and other higher organisms the liver is the principal trons) and cytochrome P450 reductase; for some chemicals,
site for the metabolism of foreign substances. It is responsible cooperation with another heme-containing protein, cytochrome
for absorbing, detoxifying, and excreting an astonishing array of b5, is also required. These enzymes and reactions occur principally
chemical substances, encountered both from outside the organism in the smooth endoplasmic reticulum.
(i.e., xenobiotics) and from within the organism, including many Following the initial hydroxylation reaction, one of several
substances synthesized by the liver itself. In the toxicology litera- additional reactions leads to the addition of more water-soluble
ture, there is a distinction between toxins, which are naturally moieties to the initial hydroxylated product. The enzymes
occurring poisons, and toxicants, which can be derived from any responsible for this so-called phase II metabolism are chiefly
source. In general, the liver and kidneys are chiefly responsible for glucuronosyltransferases, sulfotransferases, and enzymes that add
maintenance of the internal milieu of chemicals within narrow glutathione (GSH) or products of the reduced thiol form of
concentration gradients. These organs also function to remove GSH (e.g., glutathione transferases). The key substrates for these
potentially toxic compounds from organisms. In general, toxic conjugation reactions are uridine diphosphoglucuronic acid, 3′-
compounds of lower molecular weight and higher water solubil- phosphoadenosine 5′-phosphosulfate, and reduced GSH (the tri-
ity are excreted chiefly by the kidneys through glomerular filtra- peptide L-γ-glutamyl-L-cysteinylglycine).
tion and/or tubular secretion. In contrast, larger, more lipophilic The third phase of hepatic drug metabolism—the transport of
substances must be absorbed and undergo initial metabolism by the parent drug and/or its metabolites out of hepatocytes—can
the liver before their excretion either in the bile and feces or in occur in one of the following ways: drugs and/or metabolites
the urine. can be transported across the plasma membrane, with eventual
844
You might also like
- McKees Pathology of The SkinDocument1,819 pagesMcKees Pathology of The Skincatalink2191% (11)
- METABOLISM OF XENOBIOTICS-revDocument28 pagesMETABOLISM OF XENOBIOTICS-revEdo Pramana PutraNo ratings yet
- Derry Iodine Regeneration-1 PDFDocument9 pagesDerry Iodine Regeneration-1 PDFrelativelabs2075No ratings yet
- BioenergeticsDocument73 pagesBioenergeticsJiela Mae MamaatNo ratings yet
- Xenobiotic MetabolismDocument64 pagesXenobiotic MetabolismBelajar dan berdoaNo ratings yet
- 1 - XenobioticsDocument24 pages1 - XenobioticsgeenaksamuelNo ratings yet
- Drugs Induced HepatoxicityDocument10 pagesDrugs Induced HepatoxicityAgus HaryantoNo ratings yet
- BCH 220 - DR ErhunseDocument6 pagesBCH 220 - DR ErhunseSuccess OlamideNo ratings yet
- Drug Nmetabolism - BookDocument20 pagesDrug Nmetabolism - Bookcheonsa 1004No ratings yet
- Fiskes&biokim Amenida Halawa-6120055Document15 pagesFiskes&biokim Amenida Halawa-6120055Festinidar GuloNo ratings yet
- 1 - Biochemistry Notes by Dr. M A Bari Siddiqui: DrmentorsDocument3 pages1 - Biochemistry Notes by Dr. M A Bari Siddiqui: DrmentorsShiva KumarNo ratings yet
- Drug BiotransformationDocument17 pagesDrug Biotransformation4mvqwc9h7kNo ratings yet
- BCH421 CA - 2021 Revised-1Document5 pagesBCH421 CA - 2021 Revised-1idriscognitoleadsNo ratings yet
- 4 DerlemeDocument6 pages4 DerlemeŞahin ŞahinNo ratings yet
- Prescribing in Liver Disease Issue 2 Volume 32 Australian PrescriberDocument6 pagesPrescribing in Liver Disease Issue 2 Volume 32 Australian PrescriberAnonymous 9dVZCnTXSNo ratings yet
- Notes From Drug MetabolismDocument3 pagesNotes From Drug MetabolismDaniil LyalkoNo ratings yet
- Pha 201 Lecture NoteDocument10 pagesPha 201 Lecture NoteDebby14No ratings yet
- Elimination of DrugsDocument39 pagesElimination of Drugsdeepshah6068No ratings yet
- Shahed Unit I Principles of Drug Therapy Part IIDocument2 pagesShahed Unit I Principles of Drug Therapy Part IIEsdras DountioNo ratings yet
- Chapter III Pharmacokinetics: Durge Raj GhalanDocument64 pagesChapter III Pharmacokinetics: Durge Raj GhalanDurge Raj Ghalan100% (3)
- Drug Metabolism ADME PrincipalDocument38 pagesDrug Metabolism ADME PrincipalA. Kumar Soni100% (1)
- Metabolism of Xenobiotics by HarperDocument6 pagesMetabolism of Xenobiotics by HarperDr Sanjeeb Kumar Dey BaidyaNo ratings yet
- Xeno BioticsDocument27 pagesXeno BioticsMoses OrisaNo ratings yet
- Drug EliminationDocument18 pagesDrug EliminationBriana NdayisabaNo ratings yet
- Pharmacology: Modern Pharmacology With Clinical Application (6th Ed., P. 34) - IllinoisDocument7 pagesPharmacology: Modern Pharmacology With Clinical Application (6th Ed., P. 34) - IllinoisTazkiyatan IsriaNo ratings yet
- Anaesthesia Science 17Document1 pageAnaesthesia Science 17KelompokC IPDNo ratings yet
- Metabolism in Liver Kidney and PlacentaDocument22 pagesMetabolism in Liver Kidney and PlacentaBikash SahNo ratings yet
- Topic 2Document43 pagesTopic 22022451102No ratings yet
- Elimination: G. SrikarDocument23 pagesElimination: G. SrikarSowjanya NekuriNo ratings yet
- PK-PD 2022Document38 pagesPK-PD 2022احمد علىNo ratings yet
- Chapter 4 Drug MetabolismDocument14 pagesChapter 4 Drug MetabolismNicole AguilarNo ratings yet
- Lecture 2 - Basic Concepts of PharmacokineticsDocument63 pagesLecture 2 - Basic Concepts of Pharmacokineticsahmadslayman1No ratings yet
- Lecture 2 Drug Metabolism Phase IIDocument27 pagesLecture 2 Drug Metabolism Phase IIfeegame7No ratings yet
- Absorption and Distribution - 2023Document34 pagesAbsorption and Distribution - 2023Rhythm BhardwajNo ratings yet
- A Dictionary of Terms Used in Drug MetabolismDocument7 pagesA Dictionary of Terms Used in Drug MetabolismOksana KamenetskaNo ratings yet
- Metabolism of XenobioticsDocument30 pagesMetabolism of XenobioticsCzarina RiveraNo ratings yet
- A. Principles of Drug MetabolismDocument4 pagesA. Principles of Drug MetabolismLynette EvangelistaNo ratings yet
- Bioavailability (Autosaved)Document7 pagesBioavailability (Autosaved)Sunu RodriguezNo ratings yet
- Pharma 4Document11 pagesPharma 4Ibrahim dhainyNo ratings yet
- Drug Transport Across Cell Membrane: Dr. Salman H. RizviDocument67 pagesDrug Transport Across Cell Membrane: Dr. Salman H. RizviIrum RafeeqNo ratings yet
- Metabolic Changes of DrugsDocument103 pagesMetabolic Changes of DrugsDaniel Wang100% (2)
- 2 Pharmacokinetics MetabolismDocument2 pages2 Pharmacokinetics MetabolismLloyd Selorio ArboledaNo ratings yet
- 1 s2.0 S1472029923001285 MainDocument7 pages1 s2.0 S1472029923001285 MainPoramate PITAK-ARNNOPNo ratings yet
- Metabolism of XenobioticsDocument84 pagesMetabolism of XenobioticsJNo ratings yet
- Kuliah FarmakologiDocument74 pagesKuliah FarmakologiHanifNo ratings yet
- Renal Metabolism and Hypertension Review ArticleDocument12 pagesRenal Metabolism and Hypertension Review ArticlePATRICIA JACQUELINE SARMIENTO GOMEZNo ratings yet
- Adarsh Yadav 005Document14 pagesAdarsh Yadav 005Abdul WahidNo ratings yet
- Drug Metabolism: A Fascinating Link Between Chemistry and BiologyDocument24 pagesDrug Metabolism: A Fascinating Link Between Chemistry and BiologyAdnanNo ratings yet
- Drug Metabolism and Excretion by AsadDocument19 pagesDrug Metabolism and Excretion by AsadAsadullah ghalibNo ratings yet
- Biopharm M5-M8Document15 pagesBiopharm M5-M8Peniel ResurreccionNo ratings yet
- Biotransfermation of XenobioticsDocument34 pagesBiotransfermation of XenobioticsМохіт Кумар ЯмпатіNo ratings yet
- Liver and Xenobiotic MetabolismDocument13 pagesLiver and Xenobiotic MetabolismAmitNo ratings yet
- Bibi 2008Document10 pagesBibi 2008Karunagaran KNo ratings yet
- Pharmacokinetics Metabolism 2020Document70 pagesPharmacokinetics Metabolism 2020Levani KartvelishviliNo ratings yet
- Xenobiotic FKBiotransformationDocument100 pagesXenobiotic FKBiotransformationMuhammad SyaifullahNo ratings yet
- Running Head: (GAGTAN, ANDREI RAPHAEL, L.)Document9 pagesRunning Head: (GAGTAN, ANDREI RAPHAEL, L.)Edmarkmoises ValdezNo ratings yet
- 03 - Principles of PharmacokineticsDocument25 pages03 - Principles of PharmacokineticsSimonNo ratings yet
- Pharmacological Research: ReviewDocument8 pagesPharmacological Research: ReviewValiza nasya faatihahNo ratings yet
- Activity 11 MetabolismDocument6 pagesActivity 11 MetabolismdavenNo ratings yet
- The Journal of Clinical Pharma - 2013 - Urquhart - Nuclear Receptors and The Regulation of Drug Metabolizing Enzymes andDocument13 pagesThe Journal of Clinical Pharma - 2013 - Urquhart - Nuclear Receptors and The Regulation of Drug Metabolizing Enzymes andCutest guddyNo ratings yet
- Pharmaco KineticsDocument10 pagesPharmaco KineticsVithyaah ParameswaranNo ratings yet
- PPAR Agonists in Primary Biliary CholangitisDocument4 pagesPPAR Agonists in Primary Biliary CholangitiscehborrotoNo ratings yet
- Rubin 2003Document11 pagesRubin 2003shihenNo ratings yet
- Activated Monocytes Tissue Factor Expression - Sci Rep 2017Document9 pagesActivated Monocytes Tissue Factor Expression - Sci Rep 2017shihenNo ratings yet
- Rapid Testing Methods of Drugs of Abuse EDocument118 pagesRapid Testing Methods of Drugs of Abuse EshihenNo ratings yet
- Segment 001 of XN-3000 - IFU - AP - EN (1709)Document156 pagesSegment 001 of XN-3000 - IFU - AP - EN (1709)shihenNo ratings yet
- XN-Series Analytical PrincipalDocument8 pagesXN-Series Analytical PrincipalshihenNo ratings yet
- Segment 002 of XN-3000 - IFU - AP - EN (1709)Document156 pagesSegment 002 of XN-3000 - IFU - AP - EN (1709)shihenNo ratings yet
- Diestro Electrolyte Analyzer 103ap-V4r-V1.0-March-2022-EngDocument133 pagesDiestro Electrolyte Analyzer 103ap-V4r-V1.0-March-2022-EngshihenNo ratings yet
- XN-2000 Ifu Eu 1105Document412 pagesXN-2000 Ifu Eu 1105shihenNo ratings yet
- Dirui BCC-3600 User Manual 1160105408 2014-07Document110 pagesDirui BCC-3600 User Manual 1160105408 2014-07shihenNo ratings yet
- Impact of Dietary Selenium On Methylmercury Toxicity in Juvenile Atlantic Cod - A Transcriptional SurveyDocument7 pagesImpact of Dietary Selenium On Methylmercury Toxicity in Juvenile Atlantic Cod - A Transcriptional SurveyTiago TorresNo ratings yet
- BMSN2202 - Life Science II (Physiology & Biochemistry) : ReviewDocument11 pagesBMSN2202 - Life Science II (Physiology & Biochemistry) : ReviewMiles HuiNo ratings yet
- Hiv and Aids and Substance Use and AbuseDocument33 pagesHiv and Aids and Substance Use and Abusejohn frits gerard mombayNo ratings yet
- Reflection 1Document4 pagesReflection 1api-272827947No ratings yet
- PCR and Types of PCRDocument23 pagesPCR and Types of PCRadnanNo ratings yet
- NEET UG Biology Evolution PDFDocument22 pagesNEET UG Biology Evolution PDFamsaeangovanNo ratings yet
- Barron Model Test 2 GlossaryDocument2 pagesBarron Model Test 2 GlossaryRenee WuNo ratings yet
- Post-Harvest Physiology and Crop PreservationDocument574 pagesPost-Harvest Physiology and Crop PreservationRendy PramudyaNo ratings yet
- ANTIBODIESDocument8 pagesANTIBODIESlaiba Binta TahirNo ratings yet
- Cell TypesDocument16 pagesCell TypesyvetteNo ratings yet
- Plant and Animal Cell WorksheetDocument3 pagesPlant and Animal Cell WorksheetCorey ColeNo ratings yet
- Effect of Uv On BacteriaDocument11 pagesEffect of Uv On BacteriaDr.Neelam JainNo ratings yet
- S-236546 CompletoDocument361 pagesS-236546 CompletoGustavo MathiasNo ratings yet
- George K. Aghajanian and Elaine Sanders-Bush - SerotoninDocument20 pagesGeorge K. Aghajanian and Elaine Sanders-Bush - SerotoninSour60No ratings yet
- Module 1 Introduction To Animal BreedingDocument13 pagesModule 1 Introduction To Animal BreedingRaiza AwatNo ratings yet
- GamefowlDocument21 pagesGamefowlLeo HiddenValleyNo ratings yet
- Plant and Mammalian Cell Technology (BSB 3163)Document25 pagesPlant and Mammalian Cell Technology (BSB 3163)Anis FatehahNo ratings yet
- Song Zhang Diagnostic Imaging of Mediastinal Dis 230202 214458Document276 pagesSong Zhang Diagnostic Imaging of Mediastinal Dis 230202 214458Marisol DíazNo ratings yet
- Fundamentals of Bio Informatics Multiple Choice Question (GuruKpo)Document7 pagesFundamentals of Bio Informatics Multiple Choice Question (GuruKpo)GuruKPO100% (1)
- Wong Chun SamDocument7 pagesWong Chun SamYuslina YusoffNo ratings yet
- Jagendorf Andre PPDocument15 pagesJagendorf Andre PPNurlaila KhairunnisaNo ratings yet
- Population Genetics: Hardy Weinberg EquilibriumDocument114 pagesPopulation Genetics: Hardy Weinberg EquilibriumribonikNo ratings yet
- HIS-Select Technology GuideDocument6 pagesHIS-Select Technology GuideSigma-AldrichNo ratings yet
- Isolasi Dan Identifikasi Morfologi Jamur Selulolitik Dari Limbah Rumah Tangga Di Desa Sanur Kauh, BaliDocument6 pagesIsolasi Dan Identifikasi Morfologi Jamur Selulolitik Dari Limbah Rumah Tangga Di Desa Sanur Kauh, BaliNur ZuheliNo ratings yet
- CBSE Worksheet-06 Class - VI Science (Components of Food)Document4 pagesCBSE Worksheet-06 Class - VI Science (Components of Food)Ratul RanjanNo ratings yet
- Bioactive PeptidesDocument19 pagesBioactive PeptidesANNU ANN JosephNo ratings yet
- Botany HorsetailDocument3 pagesBotany HorsetailJohn Kevin NocheNo ratings yet