Professional Documents
Culture Documents
0 ratings0% found this document useful (0 votes)
2 viewsHW5
HW5
Uploaded by
Bassel Eissadescription of two phase flowl.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You might also like
- Adventurers' Guild - SwordtagDocument12 pagesAdventurers' Guild - Swordtagtruly epicNo ratings yet
- Hilti HSA AnchorsDocument4 pagesHilti HSA AnchorsandreashendiNo ratings yet
- BC-5380 Operation Manual PDFDocument550 pagesBC-5380 Operation Manual PDFAdriana Zuñiga Vega100% (1)
- Natuzza Evolo Mother Natuzza and Her Gifts Given by GodDocument7 pagesNatuzza Evolo Mother Natuzza and Her Gifts Given by GodNurli Girsang100% (1)
- Solution To Exercise 1 - PVT and Material Balance EquationsDocument4 pagesSolution To Exercise 1 - PVT and Material Balance EquationsAlbertlb AB100% (1)
- Monoslope Wind NSCPDocument8 pagesMonoslope Wind NSCPNicole100% (1)
- Formulario Termodinamica-2Document1 pageFormulario Termodinamica-2Anonymous 2x6VE65kNo ratings yet
- HWDocument2 pagesHWkesinlikle çakmaNo ratings yet
- WeeDocument34 pagesWeerozNo ratings yet
- 4 Properties of GasDocument9 pages4 Properties of GasLuffy01No ratings yet
- CH 6 - (New) Chemical Equilibrium-Nguyen Quang LongDocument81 pagesCH 6 - (New) Chemical Equilibrium-Nguyen Quang LongPHƯƠNG ĐẶNG YẾNNo ratings yet
- Determination of The Potential Work and Power Used To Compress Gas in The Gas Transport Through PipelinesDocument1 pageDetermination of The Potential Work and Power Used To Compress Gas in The Gas Transport Through PipelinesmohammadNo ratings yet
- Compressors Efficiency Calculations5Document2 pagesCompressors Efficiency Calculations5Ahmed HassanNo ratings yet
- Homework Week 5: 3 Ext Ext 3 ExtDocument10 pagesHomework Week 5: 3 Ext Ext 3 ExtIsabelle SimãoNo ratings yet
- Single Phase - Part 2Document20 pagesSingle Phase - Part 2MUHAMMAD LUQMAN HAKIMI MOHD ZAMRINo ratings yet
- Chemistry A Chemical Equilibrium KineticsDocument56 pagesChemistry A Chemical Equilibrium Kineticsokumuenock000No ratings yet
- ANs 2022Document4 pagesANs 202222149018No ratings yet
- 17 22 ProcessingDocument30 pages17 22 Processingnkrish21856No ratings yet
- Physical Chemistry Complete Outlines 2017Document20 pagesPhysical Chemistry Complete Outlines 2017Aicha DahmaniNo ratings yet
- Mathcad - Flujo en Gasoducto12sDocument5 pagesMathcad - Flujo en Gasoducto12sffNo ratings yet
- State of Matter Gases and Liquids - Short Notes - Arjuna NEET 2024Document2 pagesState of Matter Gases and Liquids - Short Notes - Arjuna NEET 2024shraddha2572sharmaNo ratings yet
- γ Yi∗Mi: Densidad Del GasDocument11 pagesγ Yi∗Mi: Densidad Del GasAnonymous ab4mVmPNo ratings yet
- Light Emitting Diode (LED) PrinciplesDocument18 pagesLight Emitting Diode (LED) PrinciplesSrinivas MishraNo ratings yet
- HBT 2Document23 pagesHBT 2Sandeep KalpiNo ratings yet
- Facts and Formulae SheetDocument7 pagesFacts and Formulae SheethenryNo ratings yet
- Chemical Reaction Fundimantal For Chemical Engineering - Part337Document1 pageChemical Reaction Fundimantal For Chemical Engineering - Part337tysir sarhanNo ratings yet
- Compressors Efficiency Calculations5Document11 pagesCompressors Efficiency Calculations5Ahmed HassanNo ratings yet
- Formula ListDocument3 pagesFormula ListRaihanNo ratings yet
- VPS CarbeneDocument6 pagesVPS CarbeneChem StudentNo ratings yet
- Formula Sheet End-TermDocument1 pageFormula Sheet End-TermAbdul Fatir KhanNo ratings yet
- Air Receivers Volume CalculationDocument86 pagesAir Receivers Volume CalculationHgagselim SelimNo ratings yet
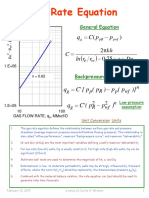
- Gas Rate EquationDocument2 pagesGas Rate EquationTripoli ManoNo ratings yet
- Dal Tons LawDocument5 pagesDal Tons LawajaiforceNo ratings yet
- Module1 Basic Equations ToolsDocument89 pagesModule1 Basic Equations ToolsEridaniNo ratings yet
- Single Phase SystemDocument61 pagesSingle Phase SystemNoorhalieza AliNo ratings yet
- ME 4061 Compressible FlowDocument18 pagesME 4061 Compressible FlowFatih İnalNo ratings yet
- AmplifierDocument20 pagesAmplifierHua-Chien ChangNo ratings yet
- KW RPM MM FT Min T N N: Internacional InglesDocument2 pagesKW RPM MM FT Min T N N: Internacional InglesAdriana Elorza RamosNo ratings yet
- 3 FTP - 2-WT PDFDocument34 pages3 FTP - 2-WT PDFMohama KarimiNo ratings yet
- Gas PropertiesDocument9 pagesGas PropertiesReza Gustarani DaneswariNo ratings yet
- PVT Behavior of Fluida & EOSDocument53 pagesPVT Behavior of Fluida & EOSEka WahyuNo ratings yet
- 3 HomogeneouseDocument26 pages3 HomogeneouseSayd KamalNo ratings yet
- HFIC Chapter 10 HF VCO DesignDocument67 pagesHFIC Chapter 10 HF VCO DesignEnricoLiaNo ratings yet
- Solution To Statistical Physics Exam: 29th June 2015Document13 pagesSolution To Statistical Physics Exam: 29th June 2015*83*22*No ratings yet
- Review Physics 2: Date: Xx/Xx/20XxDocument21 pagesReview Physics 2: Date: Xx/Xx/20XxDương Hà Trúc TâmNo ratings yet
- Capacidad Portante de Suelos Cohesivos (TERZAGHI) Brinch-Hansen Corto Plazo (Sin Drenar)Document2 pagesCapacidad Portante de Suelos Cohesivos (TERZAGHI) Brinch-Hansen Corto Plazo (Sin Drenar)RUBEN DARIO VILLCA QUIÑONESNo ratings yet
- Gas Rate Equation: P P C QDocument2 pagesGas Rate Equation: P P C QSagar DadhichNo ratings yet
- Gas RateDocument2 pagesGas RateArif KhanNo ratings yet
- Phys210 Formula Sheet - May2019Document8 pagesPhys210 Formula Sheet - May2019Franco FrecheroNo ratings yet
- Thermodynamics, Formulas and Constants: W Q U PDV WDocument1 pageThermodynamics, Formulas and Constants: W Q U PDV WMaarten ElingNo ratings yet
- Formulario Termodinamica 1Document2 pagesFormulario Termodinamica 1Jesus LyroyNo ratings yet
- Air Receivers Volume CalculationDocument83 pagesAir Receivers Volume CalculationsudarwantoNo ratings yet
- Lecture 7. Vacuum Technology: 7-1. Kinetic Theory of GasesDocument12 pagesLecture 7. Vacuum Technology: 7-1. Kinetic Theory of Gases최종윤No ratings yet
- 2 FTP - 1-WTDocument35 pages2 FTP - 1-WTMohama KarimiNo ratings yet
- Chapter10 Vcos 2Document70 pagesChapter10 Vcos 2toto byNo ratings yet
- P V V P: 1 ConstanteDocument3 pagesP V V P: 1 ConstanteAlex Mollo VillcaNo ratings yet
- CHAPTER 3-Gas-Liquid Density-Kays Rules-BBDocument24 pagesCHAPTER 3-Gas-Liquid Density-Kays Rules-BBMustafa Al-GhoulNo ratings yet
- Chap 06Document25 pagesChap 06echelon120% (1)
- Air Receivers Volume CalculationDocument83 pagesAir Receivers Volume CalculationAnonymous yt4t6XjgScNo ratings yet
- Formulae Sheet Fundamental Constants: R 0.08314 DM Bar K Mol R 0.08206 DM Atm K Mol R 8.314 J K MolDocument6 pagesFormulae Sheet Fundamental Constants: R 0.08314 DM Bar K Mol R 0.08206 DM Atm K Mol R 8.314 J K MolPauline NgNo ratings yet
- هندسة مكامن II المستوى الثالثDocument2 pagesهندسة مكامن II المستوى الثالثهشام القادريNo ratings yet
- Chemical Engineering Thermodynamics (CHE 303) : Kfupm - Edu.saDocument95 pagesChemical Engineering Thermodynamics (CHE 303) : Kfupm - Edu.sai 3l3jNo ratings yet
- assignment 3Document3 pagesassignment 3Bassel EissaNo ratings yet
- Artificial Lift Methods ComparisonsDocument12 pagesArtificial Lift Methods ComparisonsBassel EissaNo ratings yet
- Final MCQDocument31 pagesFinal MCQBassel EissaNo ratings yet
- Gym Refressing - Stress Relief - More Productive Building Muscules Refused and Have No Time ConvincedDocument1 pageGym Refressing - Stress Relief - More Productive Building Muscules Refused and Have No Time ConvincedBassel EissaNo ratings yet
- Candito 6 Week Program 1Document22 pagesCandito 6 Week Program 1Melysunn ReevsNo ratings yet
- Engine Training Manual - D114 SeriesDocument86 pagesEngine Training Manual - D114 SeriesMuhammad Imran Aftab83% (6)
- 81161i Piattaforme 04-2018 EngDocument20 pages81161i Piattaforme 04-2018 EngEvandro Amaral RodriguesNo ratings yet
- GR WU - 25th - 29 TH April, 2022Document10 pagesGR WU - 25th - 29 TH April, 2022Nishtha JainNo ratings yet
- Illusory SuperiorityDocument16 pagesIllusory Superiorityyzydrbabh5No ratings yet
- Parad ShivlingDocument2 pagesParad ShivlingsharathVEMNo ratings yet
- 74LS245Document2 pages74LS245api-3711187No ratings yet
- DR - B - R - Kerur, Dept - of Physics, GUGDocument8 pagesDR - B - R - Kerur, Dept - of Physics, GUGkerurbrk2009No ratings yet
- Consolidation (2) - 4th SEMDocument3 pagesConsolidation (2) - 4th SEMDipankar NathNo ratings yet
- Arduino Earthquake CodeDocument3 pagesArduino Earthquake Codekimchen edenelleNo ratings yet
- Non Verbal CommunicationDocument26 pagesNon Verbal Communicationrooroli74No ratings yet
- Chouvy 2014Document8 pagesChouvy 2014Fadwa BADRANANo ratings yet
- Conduction Power Loss in MOSFET Synchronous Rectifier With Parallel-Connected Schottky Barrier DiodeDocument7 pagesConduction Power Loss in MOSFET Synchronous Rectifier With Parallel-Connected Schottky Barrier DiodeMuhammad Saqib AliNo ratings yet
- Low-Level Equilibrium Trap - Wikipedia PDFDocument12 pagesLow-Level Equilibrium Trap - Wikipedia PDFMiss MishraNo ratings yet
- 3504 S 63 Rev 0 ENDocument2 pages3504 S 63 Rev 0 ENIbrahim AsifNo ratings yet
- TM 9 2320 304 14 and PDocument1,321 pagesTM 9 2320 304 14 and PjordanloNo ratings yet
- Human Anatomy NotesDocument2 pagesHuman Anatomy NotesSamantha LiberatoNo ratings yet
- TLE9 - Q2 - Mod4 Week 8Document24 pagesTLE9 - Q2 - Mod4 Week 8Ceasar Ryan AsuncionNo ratings yet
- RP-091353 Report RAN 45 SevilleDocument155 pagesRP-091353 Report RAN 45 SevilleDellNo ratings yet
- Chemistry ReportDocument29 pagesChemistry ReportJenneth Cabinto DalisanNo ratings yet
- Countrys ClimateDocument20 pagesCountrys ClimateErika Jayne100% (1)
- Gcse Geography Coursework EvaluationDocument8 pagesGcse Geography Coursework Evaluationiyldyzadf100% (2)
- Piping Vent & DrainsDocument8 pagesPiping Vent & Drainsswati2584100% (2)
- Spohn Performance, Inc.: Part# D94-02-TB-DS - Adjustable Front Track BarDocument5 pagesSpohn Performance, Inc.: Part# D94-02-TB-DS - Adjustable Front Track BarJameson PowersNo ratings yet
- THESIS Evaluation of CO2 Supermarket Refrigeration Systems. Field Measurements in Three SupermarketsDocument83 pagesTHESIS Evaluation of CO2 Supermarket Refrigeration Systems. Field Measurements in Three SupermarketsP. ReobNo ratings yet
- J.saintifika Uji Bioavailabilitas Dan BioekivalensiDocument8 pagesJ.saintifika Uji Bioavailabilitas Dan BioekivalensiAhmad MujahidinNo ratings yet
- NTN TR73 en P014Document6 pagesNTN TR73 en P014harshal161987No ratings yet
HW5
HW5
Uploaded by
Bassel Eissa0 ratings0% found this document useful (0 votes)
2 views1 pagedescription of two phase flowl.
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this Documentdescription of two phase flowl.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
0 ratings0% found this document useful (0 votes)
2 views1 pageHW5
HW5
Uploaded by
Bassel Eissadescription of two phase flowl.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
You are on page 1of 1
Question 2:
Derive the formation volume factor equation using equation of state.
1. From the Ideal Gas Law:
znRT
V=
p
At standard conditions, temperature Tsc is 60°F, which is 520°R (Rankine), and standard pressure Psc is 14.7 psi. The
volume at standard conditions Vsc is typically 1 scf. Thus, using the Ideal Gas Law at standard conditions:
nRTsc
Vsc=
Psc
For 1 scf, we simplify:
( nRTsc )
Vsc= =1
Ps c
The gas formation volume factor Bg is the volume of gas at reservoir conditions per volume at standard conditions:
znRT
V p
Bg= =
Vsc Vsc
nRTsc
Since: Vsc=
Psc
znRT
P znRT
∗Psc
nRTsc P
Bg= =
Psc nRTsc
Cancel out the nR terms:
zT
∗Psc
P
Bg=
Tsc
The universal gas constant R in ft³-psi/°R-lb-mol is approximately 10.732. We introduce the conversion factor 0.02827
to convert from the standard cubic feet at standard conditions to cubic feet per scf:
1 520
0.02827 ft 3= ft 3 psi /° R−lbmol ×
T sc 14.7
Therefore:
0.02827 zT
Bg=
P
You might also like
- Adventurers' Guild - SwordtagDocument12 pagesAdventurers' Guild - Swordtagtruly epicNo ratings yet
- Hilti HSA AnchorsDocument4 pagesHilti HSA AnchorsandreashendiNo ratings yet
- BC-5380 Operation Manual PDFDocument550 pagesBC-5380 Operation Manual PDFAdriana Zuñiga Vega100% (1)
- Natuzza Evolo Mother Natuzza and Her Gifts Given by GodDocument7 pagesNatuzza Evolo Mother Natuzza and Her Gifts Given by GodNurli Girsang100% (1)
- Solution To Exercise 1 - PVT and Material Balance EquationsDocument4 pagesSolution To Exercise 1 - PVT and Material Balance EquationsAlbertlb AB100% (1)
- Monoslope Wind NSCPDocument8 pagesMonoslope Wind NSCPNicole100% (1)
- Formulario Termodinamica-2Document1 pageFormulario Termodinamica-2Anonymous 2x6VE65kNo ratings yet
- HWDocument2 pagesHWkesinlikle çakmaNo ratings yet
- WeeDocument34 pagesWeerozNo ratings yet
- 4 Properties of GasDocument9 pages4 Properties of GasLuffy01No ratings yet
- CH 6 - (New) Chemical Equilibrium-Nguyen Quang LongDocument81 pagesCH 6 - (New) Chemical Equilibrium-Nguyen Quang LongPHƯƠNG ĐẶNG YẾNNo ratings yet
- Determination of The Potential Work and Power Used To Compress Gas in The Gas Transport Through PipelinesDocument1 pageDetermination of The Potential Work and Power Used To Compress Gas in The Gas Transport Through PipelinesmohammadNo ratings yet
- Compressors Efficiency Calculations5Document2 pagesCompressors Efficiency Calculations5Ahmed HassanNo ratings yet
- Homework Week 5: 3 Ext Ext 3 ExtDocument10 pagesHomework Week 5: 3 Ext Ext 3 ExtIsabelle SimãoNo ratings yet
- Single Phase - Part 2Document20 pagesSingle Phase - Part 2MUHAMMAD LUQMAN HAKIMI MOHD ZAMRINo ratings yet
- Chemistry A Chemical Equilibrium KineticsDocument56 pagesChemistry A Chemical Equilibrium Kineticsokumuenock000No ratings yet
- ANs 2022Document4 pagesANs 202222149018No ratings yet
- 17 22 ProcessingDocument30 pages17 22 Processingnkrish21856No ratings yet
- Physical Chemistry Complete Outlines 2017Document20 pagesPhysical Chemistry Complete Outlines 2017Aicha DahmaniNo ratings yet
- Mathcad - Flujo en Gasoducto12sDocument5 pagesMathcad - Flujo en Gasoducto12sffNo ratings yet
- State of Matter Gases and Liquids - Short Notes - Arjuna NEET 2024Document2 pagesState of Matter Gases and Liquids - Short Notes - Arjuna NEET 2024shraddha2572sharmaNo ratings yet
- γ Yi∗Mi: Densidad Del GasDocument11 pagesγ Yi∗Mi: Densidad Del GasAnonymous ab4mVmPNo ratings yet
- Light Emitting Diode (LED) PrinciplesDocument18 pagesLight Emitting Diode (LED) PrinciplesSrinivas MishraNo ratings yet
- HBT 2Document23 pagesHBT 2Sandeep KalpiNo ratings yet
- Facts and Formulae SheetDocument7 pagesFacts and Formulae SheethenryNo ratings yet
- Chemical Reaction Fundimantal For Chemical Engineering - Part337Document1 pageChemical Reaction Fundimantal For Chemical Engineering - Part337tysir sarhanNo ratings yet
- Compressors Efficiency Calculations5Document11 pagesCompressors Efficiency Calculations5Ahmed HassanNo ratings yet
- Formula ListDocument3 pagesFormula ListRaihanNo ratings yet
- VPS CarbeneDocument6 pagesVPS CarbeneChem StudentNo ratings yet
- Formula Sheet End-TermDocument1 pageFormula Sheet End-TermAbdul Fatir KhanNo ratings yet
- Air Receivers Volume CalculationDocument86 pagesAir Receivers Volume CalculationHgagselim SelimNo ratings yet
- Gas Rate EquationDocument2 pagesGas Rate EquationTripoli ManoNo ratings yet
- Dal Tons LawDocument5 pagesDal Tons LawajaiforceNo ratings yet
- Module1 Basic Equations ToolsDocument89 pagesModule1 Basic Equations ToolsEridaniNo ratings yet
- Single Phase SystemDocument61 pagesSingle Phase SystemNoorhalieza AliNo ratings yet
- ME 4061 Compressible FlowDocument18 pagesME 4061 Compressible FlowFatih İnalNo ratings yet
- AmplifierDocument20 pagesAmplifierHua-Chien ChangNo ratings yet
- KW RPM MM FT Min T N N: Internacional InglesDocument2 pagesKW RPM MM FT Min T N N: Internacional InglesAdriana Elorza RamosNo ratings yet
- 3 FTP - 2-WT PDFDocument34 pages3 FTP - 2-WT PDFMohama KarimiNo ratings yet
- Gas PropertiesDocument9 pagesGas PropertiesReza Gustarani DaneswariNo ratings yet
- PVT Behavior of Fluida & EOSDocument53 pagesPVT Behavior of Fluida & EOSEka WahyuNo ratings yet
- 3 HomogeneouseDocument26 pages3 HomogeneouseSayd KamalNo ratings yet
- HFIC Chapter 10 HF VCO DesignDocument67 pagesHFIC Chapter 10 HF VCO DesignEnricoLiaNo ratings yet
- Solution To Statistical Physics Exam: 29th June 2015Document13 pagesSolution To Statistical Physics Exam: 29th June 2015*83*22*No ratings yet
- Review Physics 2: Date: Xx/Xx/20XxDocument21 pagesReview Physics 2: Date: Xx/Xx/20XxDương Hà Trúc TâmNo ratings yet
- Capacidad Portante de Suelos Cohesivos (TERZAGHI) Brinch-Hansen Corto Plazo (Sin Drenar)Document2 pagesCapacidad Portante de Suelos Cohesivos (TERZAGHI) Brinch-Hansen Corto Plazo (Sin Drenar)RUBEN DARIO VILLCA QUIÑONESNo ratings yet
- Gas Rate Equation: P P C QDocument2 pagesGas Rate Equation: P P C QSagar DadhichNo ratings yet
- Gas RateDocument2 pagesGas RateArif KhanNo ratings yet
- Phys210 Formula Sheet - May2019Document8 pagesPhys210 Formula Sheet - May2019Franco FrecheroNo ratings yet
- Thermodynamics, Formulas and Constants: W Q U PDV WDocument1 pageThermodynamics, Formulas and Constants: W Q U PDV WMaarten ElingNo ratings yet
- Formulario Termodinamica 1Document2 pagesFormulario Termodinamica 1Jesus LyroyNo ratings yet
- Air Receivers Volume CalculationDocument83 pagesAir Receivers Volume CalculationsudarwantoNo ratings yet
- Lecture 7. Vacuum Technology: 7-1. Kinetic Theory of GasesDocument12 pagesLecture 7. Vacuum Technology: 7-1. Kinetic Theory of Gases최종윤No ratings yet
- 2 FTP - 1-WTDocument35 pages2 FTP - 1-WTMohama KarimiNo ratings yet
- Chapter10 Vcos 2Document70 pagesChapter10 Vcos 2toto byNo ratings yet
- P V V P: 1 ConstanteDocument3 pagesP V V P: 1 ConstanteAlex Mollo VillcaNo ratings yet
- CHAPTER 3-Gas-Liquid Density-Kays Rules-BBDocument24 pagesCHAPTER 3-Gas-Liquid Density-Kays Rules-BBMustafa Al-GhoulNo ratings yet
- Chap 06Document25 pagesChap 06echelon120% (1)
- Air Receivers Volume CalculationDocument83 pagesAir Receivers Volume CalculationAnonymous yt4t6XjgScNo ratings yet
- Formulae Sheet Fundamental Constants: R 0.08314 DM Bar K Mol R 0.08206 DM Atm K Mol R 8.314 J K MolDocument6 pagesFormulae Sheet Fundamental Constants: R 0.08314 DM Bar K Mol R 0.08206 DM Atm K Mol R 8.314 J K MolPauline NgNo ratings yet
- هندسة مكامن II المستوى الثالثDocument2 pagesهندسة مكامن II المستوى الثالثهشام القادريNo ratings yet
- Chemical Engineering Thermodynamics (CHE 303) : Kfupm - Edu.saDocument95 pagesChemical Engineering Thermodynamics (CHE 303) : Kfupm - Edu.sai 3l3jNo ratings yet
- assignment 3Document3 pagesassignment 3Bassel EissaNo ratings yet
- Artificial Lift Methods ComparisonsDocument12 pagesArtificial Lift Methods ComparisonsBassel EissaNo ratings yet
- Final MCQDocument31 pagesFinal MCQBassel EissaNo ratings yet
- Gym Refressing - Stress Relief - More Productive Building Muscules Refused and Have No Time ConvincedDocument1 pageGym Refressing - Stress Relief - More Productive Building Muscules Refused and Have No Time ConvincedBassel EissaNo ratings yet
- Candito 6 Week Program 1Document22 pagesCandito 6 Week Program 1Melysunn ReevsNo ratings yet
- Engine Training Manual - D114 SeriesDocument86 pagesEngine Training Manual - D114 SeriesMuhammad Imran Aftab83% (6)
- 81161i Piattaforme 04-2018 EngDocument20 pages81161i Piattaforme 04-2018 EngEvandro Amaral RodriguesNo ratings yet
- GR WU - 25th - 29 TH April, 2022Document10 pagesGR WU - 25th - 29 TH April, 2022Nishtha JainNo ratings yet
- Illusory SuperiorityDocument16 pagesIllusory Superiorityyzydrbabh5No ratings yet
- Parad ShivlingDocument2 pagesParad ShivlingsharathVEMNo ratings yet
- 74LS245Document2 pages74LS245api-3711187No ratings yet
- DR - B - R - Kerur, Dept - of Physics, GUGDocument8 pagesDR - B - R - Kerur, Dept - of Physics, GUGkerurbrk2009No ratings yet
- Consolidation (2) - 4th SEMDocument3 pagesConsolidation (2) - 4th SEMDipankar NathNo ratings yet
- Arduino Earthquake CodeDocument3 pagesArduino Earthquake Codekimchen edenelleNo ratings yet
- Non Verbal CommunicationDocument26 pagesNon Verbal Communicationrooroli74No ratings yet
- Chouvy 2014Document8 pagesChouvy 2014Fadwa BADRANANo ratings yet
- Conduction Power Loss in MOSFET Synchronous Rectifier With Parallel-Connected Schottky Barrier DiodeDocument7 pagesConduction Power Loss in MOSFET Synchronous Rectifier With Parallel-Connected Schottky Barrier DiodeMuhammad Saqib AliNo ratings yet
- Low-Level Equilibrium Trap - Wikipedia PDFDocument12 pagesLow-Level Equilibrium Trap - Wikipedia PDFMiss MishraNo ratings yet
- 3504 S 63 Rev 0 ENDocument2 pages3504 S 63 Rev 0 ENIbrahim AsifNo ratings yet
- TM 9 2320 304 14 and PDocument1,321 pagesTM 9 2320 304 14 and PjordanloNo ratings yet
- Human Anatomy NotesDocument2 pagesHuman Anatomy NotesSamantha LiberatoNo ratings yet
- TLE9 - Q2 - Mod4 Week 8Document24 pagesTLE9 - Q2 - Mod4 Week 8Ceasar Ryan AsuncionNo ratings yet
- RP-091353 Report RAN 45 SevilleDocument155 pagesRP-091353 Report RAN 45 SevilleDellNo ratings yet
- Chemistry ReportDocument29 pagesChemistry ReportJenneth Cabinto DalisanNo ratings yet
- Countrys ClimateDocument20 pagesCountrys ClimateErika Jayne100% (1)
- Gcse Geography Coursework EvaluationDocument8 pagesGcse Geography Coursework Evaluationiyldyzadf100% (2)
- Piping Vent & DrainsDocument8 pagesPiping Vent & Drainsswati2584100% (2)
- Spohn Performance, Inc.: Part# D94-02-TB-DS - Adjustable Front Track BarDocument5 pagesSpohn Performance, Inc.: Part# D94-02-TB-DS - Adjustable Front Track BarJameson PowersNo ratings yet
- THESIS Evaluation of CO2 Supermarket Refrigeration Systems. Field Measurements in Three SupermarketsDocument83 pagesTHESIS Evaluation of CO2 Supermarket Refrigeration Systems. Field Measurements in Three SupermarketsP. ReobNo ratings yet
- J.saintifika Uji Bioavailabilitas Dan BioekivalensiDocument8 pagesJ.saintifika Uji Bioavailabilitas Dan BioekivalensiAhmad MujahidinNo ratings yet
- NTN TR73 en P014Document6 pagesNTN TR73 en P014harshal161987No ratings yet