Professional Documents
Culture Documents
How to Prisma
How to Prisma
Uploaded by
Christine YauCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
How to Prisma
How to Prisma
Uploaded by
Christine YauCopyright:
Available Formats
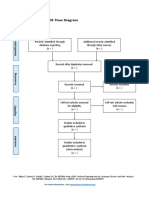
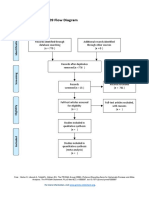
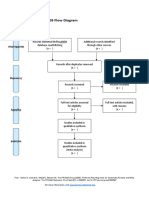
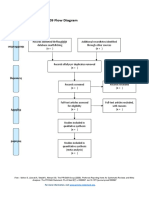
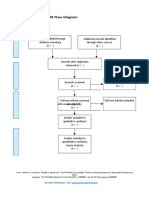
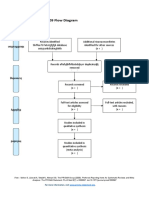
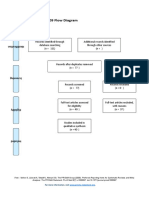
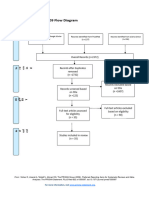
PRISMA 2009 Flow Diagram – HOW TO
tifi
tio
en
ca
Id
Records identified through Additional records identified
database searching through other sources
(n = ) (n = )
Records after duplicates removed
(n = )
en
Sc
re
in
g
Records screened Records excluded
(n = ) (n = )
gib
ilit
Eli
Full-text articles assessed Full-text articles excluded,
for eligibility with reasons
(n = ) (n = )
Studies included in
qualitative synthesis
(n = )
ud
ed
In
cl
From: Moher D, Liberati A, Tetzlaff J, Altman DG, The PRISMA Group (2009). Preferred Reporting Items for Systematic Reviews and Meta-
Analyses: The PRISMA Statement. PLoS Med 6(7): e1000097. doi:10.1371/journal.pmed1000097
For more information, visit www.prisma-statement.org.
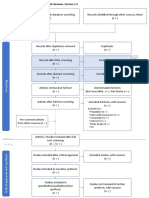
This looks great Julie, you are definitely ready to fill in your PRISMA
So to fill this in….
Records identified through database searching - this is your total number of hits (relevant or
otherwise)
Additional records identified through other sources – this is the number of total hits from
reference list searching
Records after duplicates removed - total number after you have gone through and removed all
duplicates (whether they are relevant or not)
Records screened- this number will be the same as the above (Records after duplicates removed).
After this step, this is where you go through the ones you identified as relevant in your literature
table (double check all duplicates have been removed), apply Cohen’s preview, read etc, and
screen for relevant hits.
Whilst it seems like you are backtracking here this is a rechecking phase that is important for
consistency.
You then record your excluded number (so the number you removed that was irrelevant) in the
Records excluded section of the diagram.
This will give you the number for Full-text articles assessed for eligibility
You then, read the full text of the articles you have left, the ‘relevant’ ones.
and screen out any that are deemed irrelevant or unsuitable (they do not meet your criteria
fully). Record your exclusions and the reasons in the Full-text articles excluded, with reasons
section.
This will give you the final set of papers that you will use in your review.
Once you have completed these steps, you are ready to complete the quality assessment. Begin
by dividing the remaining articles into theoretical, qualitative (and mixed methods), and
quantitative.
Theoretical articles will use the Authority, Accuracy, Coverage, Objectivity, Date, Significance
(AACODS) checklist (Tyndall, 2008).
Qualitative will use CASP
Quantitative will use STROBE.
From: Moher D, Liberati A, Tetzlaff J, Altman DG, The PRISMA Group (2009). Preferred Reporting Items for Systematic Reviews and Meta-
Analyses: The PRISMA Statement. PLoS Med 6(7): e1000097. doi:10.1371/journal.pmed1000097
For more information, visit www.prisma-statement.org.
You might also like
- Productize Yourself - How Jack Butcher Built His 1M Year Online BusinessDocument33 pagesProductize Yourself - How Jack Butcher Built His 1M Year Online BusinessJim100% (4)
- PRISMA 2009 Flow Diagram MS WordDocument1 pagePRISMA 2009 Flow Diagram MS WordYuliza ArianiNo ratings yet
- ROSES Flow Diagram For Systematic ReviewsDocument1 pageROSES Flow Diagram For Systematic Reviewswhy daNo ratings yet
- PRISMA Flow DiagramDocument1 pagePRISMA Flow DiagramavfNo ratings yet
- Mythic RacesDocument173 pagesMythic RacesRobert Howell100% (9)
- Iso 18295 1 2017Document11 pagesIso 18295 1 2017Duilio HomarNo ratings yet
- PRISMA 2009 Flow Diagram PDFDocument1 pagePRISMA 2009 Flow Diagram PDFসোমনাথ মহাপাত্রNo ratings yet
- PRISMA 2020 Flow Diagram For New Systematic Reviews Which Included Searches of Databases, Registers and Other SourcesDocument1 pagePRISMA 2020 Flow Diagram For New Systematic Reviews Which Included Searches of Databases, Registers and Other SourcesSurya WijayaNo ratings yet
- PRISMA Flow Diagram: For More Information, VisitDocument1 pagePRISMA Flow Diagram: For More Information, VisitimranNo ratings yet
- PRISMA-2009-Flow-Diagram-MS-WordDocument1 pagePRISMA-2009-Flow-Diagram-MS-WordLarissa KischenahNo ratings yet
- PRISMA 2009 Flow Diagram PDFDocument1 pagePRISMA 2009 Flow Diagram PDFKalis WarenNo ratings yet
- PRISMA 2009 Flow Diagram: For More Information, VisitDocument1 pagePRISMA 2009 Flow Diagram: For More Information, VisitpedriyepseNo ratings yet
- PRISMA 2009 Flow DiagramDocument1 pagePRISMA 2009 Flow DiagramKou YashiroNo ratings yet
- PRISMA 2009 Flow DiagramDocument1 pagePRISMA 2009 Flow DiagramAndrés Felipe Obregón HernándezNo ratings yet
- PRISMA 2009 Flow Diagram: For More Information, VisitDocument1 pagePRISMA 2009 Flow Diagram: For More Information, VisitpedriyepseNo ratings yet
- PRISMA 2009 Flow Diagram: For More Information, VisitDocument1 pagePRISMA 2009 Flow Diagram: For More Information, VisitpedriyepsegsgsgsNo ratings yet
- PRISMA 2009 Flow Diagram: For More Information, VisitDocument1 pagePRISMA 2009 Flow Diagram: For More Information, VisitpedriyepsegsgsgsNo ratings yet
- PRISMA 2009 Flow DiagramDocument1 pagePRISMA 2009 Flow DiagramnurulNo ratings yet
- PRISMA 2009 Flow DiagramDocument1 pagePRISMA 2009 Flow DiagrampedriyepsegsgsgsNo ratings yet
- PRISMA 2009 Flow Diagram: For More Information, VisitDocument1 pagePRISMA 2009 Flow Diagram: For More Information, VisitpedriyepsegututsgsgsNo ratings yet
- PRISMA 2009 Flow Diagram: For More Information, VisitDocument1 pagePRISMA 2009 Flow Diagram: For More Information, VisitpedriyepsegututsgsgskhkhNo ratings yet
- PRISMA 2009 Flow Diagram: For More Information, VisitDocument1 pagePRISMA 2009 Flow Diagram: For More Information, VisitRahma Aulia KhairunnisaNo ratings yet
- PRISMA 2009 Flow Diagram: For More Information, VisitDocument1 pagePRISMA 2009 Flow Diagram: For More Information, VisitpedriyepsegututsgsgskhkhNo ratings yet
- PRISMA 2009 Flow Diagram: For More Information, VisitDocument1 pagePRISMA 2009 Flow Diagram: For More Information, VisitpedriyepsegututsgsgskhkhNo ratings yet
- PRISMA 2009 Flow Diagram: For More Information, VisitDocument1 pagePRISMA 2009 Flow Diagram: For More Information, VisitpedriyepsegututsgsgskhkhNo ratings yet
- Bmjopen 2020 December 10 12 Inline Supplementary Material 3Document1 pageBmjopen 2020 December 10 12 Inline Supplementary Material 3Alessandra Miranda PadillaNo ratings yet
- PRISMA 2020 Flow Diagram For Updated Systematic Reviews Which Included Searches of Databases, Registers and Other SourcesDocument1 pagePRISMA 2020 Flow Diagram For Updated Systematic Reviews Which Included Searches of Databases, Registers and Other SourcesSurya WijayaNo ratings yet
- PRISMA 2009 Flow DiagramDocument1 pagePRISMA 2009 Flow DiagramsamiratumananNo ratings yet
- PRISMA 2009 Flow Diagram PreenchidoDocument1 pagePRISMA 2009 Flow Diagram PreenchidoSDNo ratings yet
- PRISMA - 2020 - Flow - Diagram - Updated. JK Menambah Yg SDH Ada. TP Register OnlyDocument1 pagePRISMA - 2020 - Flow - Diagram - Updated. JK Menambah Yg SDH Ada. TP Register Onlyfachry albabNo ratings yet
- ROSES Flow Diagram For Systematic MapsDocument1 pageROSES Flow Diagram For Systematic MapsLina DiazNo ratings yet
- ROSES Flow Diagram For Systematic MapsDocument1 pageROSES Flow Diagram For Systematic MapsJoanne WongNo ratings yet
- PRISMA 2009 Flow Diagram PreenchidoDocument1 pagePRISMA 2009 Flow Diagram PreenchidoSDNo ratings yet
- MMC 1Document1 pageMMC 1hopenmazeNo ratings yet
- PRISMA 2009 Flow Diagram: For More Information, VisitDocument1 pagePRISMA 2009 Flow Diagram: For More Information, VisitAndrés Felipe Obregón HernándezNo ratings yet
- PRISMA - Urban e KhouryDocument1 pagePRISMA - Urban e KhourypasqualiniericaNo ratings yet
- Flow DiagramDocument1 pageFlow DiagramdiribaNo ratings yet
- PRISMA 2020 Flow Diagram New SRs v1Document1 pagePRISMA 2020 Flow Diagram New SRs v1Krisna ayuNo ratings yet
- PRISMA 2020 Flow Diagram For New Systematic Reviews Which Included Searches of Databases and Registers OnlyDocument2 pagesPRISMA 2020 Flow Diagram For New Systematic Reviews Which Included Searches of Databases and Registers OnlyJavier Gio AlvarezNo ratings yet
- Draft Diagram PRISMA Paper ESGDocument1 pageDraft Diagram PRISMA Paper ESGniar196No ratings yet
- AbstrakDocument2 pagesAbstrakIndraNo ratings yet
- PRISMA 2020 Flow Diagram New SRs v2Document1 pagePRISMA 2020 Flow Diagram New SRs v2Ivan PradhanaNo ratings yet
- PRISMA 2020 Flow Diagram New SRs v1Document1 pagePRISMA 2020 Flow Diagram New SRs v1eclipsesolidNo ratings yet
- PRISMA 2009 Flow Diagram: For More Information, VisitDocument1 pagePRISMA 2009 Flow Diagram: For More Information, VisitabdulNo ratings yet
- Dissertation StructureDocument4 pagesDissertation StructureLonny AtienoNo ratings yet
- NEW - Konvalesen Plasma - PRISMA 2009 Flow DiagramDocument1 pageNEW - Konvalesen Plasma - PRISMA 2009 Flow DiagramReynardi SutantoNo ratings yet
- PRISMA 2020 Flow Diagram SimpleDocument1 pagePRISMA 2020 Flow Diagram Simplecallytedford5No ratings yet
- PRISMA 2009 Flow Diagram: For More Information, VisitDocument1 pagePRISMA 2009 Flow Diagram: For More Information, VisitManJ_mchNo ratings yet
- PRISMA 2009 Flow Diagram: 1. Youth, or Adolescents (N 100) 2. Irrelevant With Tubercolusi (N 35) 3Document1 pagePRISMA 2009 Flow Diagram: 1. Youth, or Adolescents (N 100) 2. Irrelevant With Tubercolusi (N 35) 3aisah andiniNo ratings yet
- PRISMA Diagram TemplateDocument1 pagePRISMA Diagram TemplateRitashree DasguptaNo ratings yet
- Prisma PediculosisDocument1 pagePrisma PediculosisAni TikaNo ratings yet
- Diagram PrismaDocument1 pageDiagram PrismaevamotretNo ratings yet
- PRISMA 2020 Flow Diagram New SRs v1Document1 pagePRISMA 2020 Flow Diagram New SRs v1Research and Academic AMSA-BrawijayaNo ratings yet
- PRISMA 2009 Flow DiagramDocument1 pagePRISMA 2009 Flow Diagrammoutasim mohammadNo ratings yet
- Prisma PediculosisDocument1 pagePrisma PediculosisAni TikaNo ratings yet
- Kalman Filter Tuning For Digital Protection Applications: Jose A. Rosendo MaciasDocument5 pagesKalman Filter Tuning For Digital Protection Applications: Jose A. Rosendo MaciasFabien CallodNo ratings yet
- PRISMA - 2020 - Flow - Diagram - New - SRs - v1 2Document1 pagePRISMA - 2020 - Flow - Diagram - New - SRs - v1 2Francisco Castillo VázquezNo ratings yet
- Figure 1 - ExampleDocument1 pageFigure 1 - ExamplediegofilipebNo ratings yet
- fpsyg-11-562506Document29 pagesfpsyg-11-562506Christine YauNo ratings yet
- 01-The-Effectiveness-of-EMDR-for-Medically-Unexplained-Symptoms-A-Systematic-Literature-ReviewDocument32 pages01-The-Effectiveness-of-EMDR-for-Medically-Unexplained-Symptoms-A-Systematic-Literature-ReviewChristine YauNo ratings yet
- SUMARI access via the WSU LibraryDocument2 pagesSUMARI access via the WSU LibraryChristine YauNo ratings yet
- Bringing Up Kids in The Early Years - Chapter 1aDocument5 pagesBringing Up Kids in The Early Years - Chapter 1aChristine YauNo ratings yet
- Small Group Experience VS One To One CounsellingDocument2 pagesSmall Group Experience VS One To One CounsellingChristine YauNo ratings yet
- Safe and Together ModelDocument1 pageSafe and Together ModelChristine YauNo ratings yet
- Cozolino & Davis (2017) Ch2 How People ChangeDocument10 pagesCozolino & Davis (2017) Ch2 How People ChangeChristine YauNo ratings yet
- From The Australian Childhood Foundation (ACF) Safe-Secure Children & Yound People Affected by Family ViolenceDocument52 pagesFrom The Australian Childhood Foundation (ACF) Safe-Secure Children & Yound People Affected by Family ViolenceChristine YauNo ratings yet
- Treatments For Anger in Specific Populations Theor... - (3. Emotion-Focused Therapy For Anger in Complex Trauma)Document41 pagesTreatments For Anger in Specific Populations Theor... - (3. Emotion-Focused Therapy For Anger in Complex Trauma)Christine YauNo ratings yet
- Jurassic Park - 1Document3 pagesJurassic Park - 1Christine YauNo ratings yet
- BlueKnot Practice Guidelines 2019Document270 pagesBlueKnot Practice Guidelines 2019Christine YauNo ratings yet
- Properties of Bio-Based PolyurethaneDocument200 pagesProperties of Bio-Based PolyurethaneMust BoujaataNo ratings yet
- Ca Q&aDocument129 pagesCa Q&aRavi TejaNo ratings yet
- Using WebLogic Server Multitenant PDFDocument256 pagesUsing WebLogic Server Multitenant PDFFabian Cabrera0% (1)
- Per 3 Science7Document2 pagesPer 3 Science7Maria Cristina PolNo ratings yet
- Seal Design For High Pressure Offshore-Morton & EvansDocument10 pagesSeal Design For High Pressure Offshore-Morton & EvansMachineryengNo ratings yet
- 10 1 1 590 9326 PDFDocument13 pages10 1 1 590 9326 PDFsbarwal9No ratings yet
- GSCFF APF TechniquesDocument18 pagesGSCFF APF TechniquesMaharaniNo ratings yet
- Reporting VerbsDocument4 pagesReporting VerbsMaksat AbetayevNo ratings yet
- KPMG China Pharmaceutical 201106Document62 pagesKPMG China Pharmaceutical 201106merc2No ratings yet
- B23 PDFDocument3 pagesB23 PDFHector SilvaNo ratings yet
- Social Dimensions in Education Part 1Document4 pagesSocial Dimensions in Education Part 1Laurice Marie GabianaNo ratings yet
- MSG456 Mathematical - Programming (May 2010)Document7 pagesMSG456 Mathematical - Programming (May 2010)dikkanNo ratings yet
- Propylene (Asia-Pacific) 31 Jan 2020Document5 pagesPropylene (Asia-Pacific) 31 Jan 2020Wira Nata S SinuhajiNo ratings yet
- Patterns of Labour ExploitationDocument15 pagesPatterns of Labour ExploitationNeeraj KaundalNo ratings yet
- Build Tight RangeDocument8 pagesBuild Tight RangeMihai MunteanuNo ratings yet
- Jataka Tales 3 PDFDocument3 pagesJataka Tales 3 PDFGhosalNo ratings yet
- Minebea Stepper Motor Part Number Decoding TableDocument5 pagesMinebea Stepper Motor Part Number Decoding TableWijebNo ratings yet
- Semi-Detailed Lesson Plan For Grade-7 Mathematics Prepared By: Jennelyn G. Malayno ObjectivesDocument2 pagesSemi-Detailed Lesson Plan For Grade-7 Mathematics Prepared By: Jennelyn G. Malayno Objectivesjennelyn malaynoNo ratings yet
- MCQ ObgDocument3 pagesMCQ ObgSahil sharma100% (1)
- 1) AISC Standards and PublicationsDocument6 pages1) AISC Standards and PublicationsJonatan MamaniNo ratings yet
- SMART Objective RubricDocument1 pageSMART Objective RubricKeren HapkhNo ratings yet
- ROMI Forecasting ToolDocument2 pagesROMI Forecasting Toolkurtkomeng182No ratings yet
- GCC Lab Manual Mahesh Kumar PDFDocument130 pagesGCC Lab Manual Mahesh Kumar PDFMohan ReddyNo ratings yet
- Urban Coherence: A Morphological DefinitionDocument19 pagesUrban Coherence: A Morphological DefinitionOlgu CaliskanNo ratings yet
- SQL ExceriseDocument10 pagesSQL ExceriseSri VatsaNo ratings yet
- (Updated Constantly) : CCNA 1 (v5.1 + v6.0) Chapter 6 Exam Answers FullDocument16 pages(Updated Constantly) : CCNA 1 (v5.1 + v6.0) Chapter 6 Exam Answers Fullfitroh fitrohNo ratings yet
- Giesekus ModelDocument18 pagesGiesekus Modelvasopoli1No ratings yet