Professional Documents
Culture Documents
C - 8. (ALKYL & ARYL HALLIDE, ALCOHOL ETHERS & PHENOLS) (ADV)
C - 8. (ALKYL & ARYL HALLIDE, ALCOHOL ETHERS & PHENOLS) (ADV)
Uploaded by
revohe2640Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
C - 8. (ALKYL & ARYL HALLIDE, ALCOHOL ETHERS & PHENOLS) (ADV)
C - 8. (ALKYL & ARYL HALLIDE, ALCOHOL ETHERS & PHENOLS) (ADV)
Uploaded by
revohe2640Copyright:
Available Formats
ANDHERI / BORIVALI / DADAR / CHEMBUR / THANE / MULUND/ NERUL / POWAI
IIT – JEE CRASH COURSE (ADV.) MARKS: 124
TIME: 75 MIN. DATE:
TOPIC: ALKYL & ARYL HALLIDE, ALCOHOL ETHERS & PHENOLS
SECTION–I (Multiple Choice Questions)
This section contains 06 multiple choice questions. Each question has 4 choices (A), (B), (C) and
(D) for its answer, out which ONLY ONE is correct. (+3,- 1)
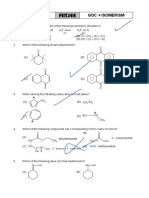
1.
OH
D
CHClBrI
?
t--BuO
NH2
(A) O OH (B) O OH
C C
H H
NC NC
(C) O OH (D) O OH
C C
H D
Cl Cl
NC NC
2.
O O
(i) Mg(Hg) in benzene conc
+
A B
H3C (ii) H3O
CH H2SO4
3
Zn (Hg)
conc HCl
C
The product (C) in the above sequence of reactions will be
(A) (B)
H3 C
H3 C CH3 H3C
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 1
(C) CH3 (D)
H3C CH3
3. During the zeisel estimation of an aromatic ether C 9 H12 O 3 ,1.68g produces 4.7 g of yellow ppt.
what is the structure of the ether?
(A) OCH3 (B) OCH3
OCH3 OCH3
CH2OH OCH3
(C) OCH3 (D) OH
CH2OH
HOH2C CH2OH H3C
OCH3
4.
OH CHO
OCH3
Br2
+
CH3O G Br
G
The rate of reaction is moderately affected by the nature of G. Choose the correct decreasing rate of
reaction with various substituents as G.
(A) NO 2 Br H CH3 OCH3 (B) OCH3 CH3 H Br NO2
(C) OCH 3 Br CH 3 H NO 2 (D) NO 2 OCH 3 Br H CH3
5. Find the product of the following reaction:
R R
CH2
R O
(A) R O (B) R
and CH 3 CH CH 2 R
R R
O R
(C) R (D) R
H
andH2C R
CH2
R R O R
O
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 2
6. The following compounds on treatment with HI at 273 K give an alcohol and alkyl iodide which of
these form an alcohol that gives positive haloform reaction?
CH3 CH3 CH3
O CH CH3 CH3 CH O C CH3
2
CH3
(I) (II)
CH3
CH3 CH3CH2O C CH3
CH3CH2O CH CH3 CH3
(III) (IV)
(A) II, III, IV (B) I, II, III, IV (C) III only (D) I, II, III
SECTION-II (Multiple Choice Questions)
This section contains 06 multiple choice questions. Each question has 4 choices (A), (B), (C) and
(D) for its answer, out which ONE OR MORE is/are correct. (+4,- 2)
7. Which of the following reactions are correctly interpreted?
(A) CH3
H2O
Hg OAc 2
NaHB4
Product is a mixture of
diastereomers
CH3
(B) CH3 CH3 CH3 O
Ag
CH3 C C C C CH3
OH Cl
CH3
(racemic mixture)
(C) CH3 CH3
Ts Cl CH3 CH 2SH
OH A SCH2CH3
(D)
Pt
O O H 2 HO OH
8. Identify the correct statements:
(A) 1, 2, 1, 3 and 1, 4 dioxanes are used as solvents in reactions involving Grignard reagent
OCH3
CH3 CH 2 CH CH OH and CH3 CH H32C4 CH
O OH OCH3
(B) all give positive tollen’s
reagent test
(C) The alcohol which reacts most readily with Na metal will give fastest turbidity with lucas reagent
(D) Acylation of salicylic acid in basic medium occurs on phenolic oxygen
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 3
9. Compound (X) C 4 H8O decolonizes Baeyers reagent. It undergoes hydrolysis in dil H 2SO4 to give
(Y) and (Z) where both (Y) and (Z) give positive iodoform test while only (Y) responds to Tollen’s
test but (Z) does not. Choose the correct statements of the following.
OH
(A) Y
CH3 CH CH CHO
(B) Z
PCC/CH 2 Cl 2
Y
O3 / Zn /H 2 O
(C) X Y other product
HI aq.KOH
(D) X Y W W Z
10. Which of the following can give E 1 CB reaction
(A) OH (B) H
CH 3 CH CN Cl C CF3
Cl
(C) O OH (D) HCF2 CCl3
H3C CH3
CH3
11. Identify the correct statements
(A) Isobutene reacts faster than propene with HCl
(B) The total number of stereoisomer obtained when 1, 6 – dimethyl cyclohexene reacts with HBr in
presence of peroxide is 8.
(C)
Cl F
undergoes SNAr2 faster than
NO2 NO2
(D) The decreasing leaving group ability follows the order NH 3 PH3 AsH3 SbH 3
12. Which of the following paths is/are feasible for preparation of divinylether
(A) H C ONa +H C
2 X
2
(B) H C OH + HO conc
2 X
H 2SO 4
Low temperature
(C) i Hg OCOCF3 2
CH 2 CH 2
ii CH 2 CH OH
iii NaBH 4
(D) ClCH 2 CH 2 OH i conc H 2SO 4
ii NaNH 2
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 4
SECTION – III (Paragraph Type)
This section contains 2 multiple choice questions relating to 1 paragraph. Each question has four
choices (A), (B), (C) and (D) out of which ONLY ONE is correct. (+3,- 1)
PARAGRAPH FOR QUE. NOS. 13 & 14
13. The compound (C) is
(A) OH (B) Cl
Cl OH
Cl Cl
Cl Cl
(C) OH (D) Both (A) and (B)
Cl
Cl
Cl
14. The compound (E) is
(A) OH OH (B) OH Cl
Cl Cl Cl Cl
Cl Cl Cl Cl
Cl Cl Cl OH
(C) OH OH (D) Both (A) and (C)
Cl Cl
Cl Cl Cl
Cl
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 5
PARAGRAPH FOR QUE. NOS. 15 & 16
15. Identify the correct statements:-
(A) The products (F) and (G) are structural isomers
(B) The products (F) and (G) are geometrical isomers
(C) The compound (D) is
CH3
O
CH3
H3C CH3
(D) The compound (I) is
OH
16. The reaction and mechanism involved in formation of (H) from (A) respectively are:-
(A) Elimination reaction, E1 (B) Elimination reaction, E1CB
(C) Williamsous synthesis SN 2 (D) Williamsous synthesis SN1
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 6
PARAGRAPH FOR QUE. NOS. 15 & 16
HBr
C8 H13 Br B C
Meso R acemate
(A)
t-BuONa
t-BuOH
Br2
Br
(A)
C8 H12 Br
COOEt
COOEt
(F)
O3 / Zn/ H 2 O G
17. Compound (G) is
(A) O (B) O
COOEt
COOEt
COOEt
COOEt O
O
(C) O (D) O O
COOEt COOEt
COOEt COOEt
O
O O
18. Compound (D) is
(A) CH3 (B) CH2
CH2 CH2
(C) CH2 (D) CH3
CH3
CH3
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 7
SECTION – IV (Integer Answer Type)
This section contains 08 questions. The answer to each of the questions is a single digit integer,
ranging from 0 to 9. The correct digit below the question number in the ORS is to be bubbled.
(+4, 0)
19.
O O
i AlCl
2 + O O
3
ii HCl A B
Zn Hg / conc HCl
O O
C D
H3 PO4
E F
Zn Hg / conc HCl
G
The number of benzene rings in (G) is:
20. The total number of bonds present in the intermediate meisenheimer complex of the following
reaction is
Cl NR2
R2 N H HCl
H3C CH3 H3C CH3
CN CN
21.
O O CH3
O
H3C O
O O
H3C O
O CH3
Cl
O O O OHO
H3C
The ratio of the number of moles of Grignard reagent consumed by the above compound to the
number of moles of ethanol formed in the reaction will be
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 8
22. The total number of moles of HIO 4 needed for the oxidation of
OH
O OH
OH
is :
OH
OH
OH CH3
23. The number of molecules among the following which give haloform reaction:
O O O O O
CH3 C O C CH3 , CH3 C C OH , CH3 C Cl
O O Cl O
CH3 C OCH3 , CH3 C NH2 , CH3 CH C CH3 ,
O O OH O
CH3 , ,
C C CH3 CH C CH3 CH3 CH C CH3
Cl OH
24.
H3 C CH3
NBS
h
Total number of products obtained (excluding stereoisomers)
25. The total number of product including minor product and stereoisomers possible in the following
reaction are:
CH3
H
CH3
OH
26. The number of ethers among the following which can be prepared using Williamsons synthesis are:
O O
H3C CH3
CH 2 CH O CH CH 2
CH3
CH3 CH3 O CH3
O CH3
H3 C O CH3 O CH3
H3 C
O CH3
CH3
CH3
O 2N
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 9
SECTION - V (Matrix Match Type)
This section 1 Question. Each question has four statements Given in Column - I and four statements
in Column – II. Any given statement in Column – I can have correct matching with one or more
statement (s) given in column II. (+8, 0)
27. Match the column:-
Column – I Column – II
(A) NO2 (P) SArE 2
3
EtO
2
Br 1
Cl
(B) OH (Q) Reaction at position – 1
3
2 Br2
Fe
1
NO2
(C) OCH3 (R) Reaction at position – 2
3 HNO
3
H 2 SO4
2
1
Br
(D) 1 (S) Reaction at position – 3
2
Br2
Fe
3
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 10
28.
Column – I Column – II
(A) CH3 (P) SN 2 reaction on treatment with alcoholic
CH3 C Cl NaSH
CH2CH3
(B) Br (Q) E 2 reaction on treatment with alcoholic KOH
(C) Cl (R) SN Ar reaction on treatment with fused NaOH
H3 C CH2-CH2Cl
(D) CH3 (S) SN1 reaction with aq- KOH
Br
H3C CH3
29.
Column – I Column – II
(A) COOH (P) CO 2 gas will be evolved on addition of
HO3S NaHCO3
OH
CH2-OH
(B) OH (Q) H 2 gas will be evolved on addition of Na
metal
(C) C CH (R) H 2 O will be formed on addition of NaOH
CH2OH
(D) OH (S) NH3 gas will be evolved on addition of
NaNH 2
OH
OH
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 11
30.
Column – I Column – II
(A) CH3 (P) Saytzeff alkene is the major product
Ph H
CH3O K
Ph Br
CH3
(B) CH3 (Q) Hofmann’s alkene is the major product
Br
C2 H 5 O K
(C) (R) Stereo specific reaction
OH
+
N
Me
Me
(D) CH3 (S) A single stereoisomer will be obtained
C2H 5O K
F
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 12
ANDHERI / BORIVALI / DADAR / CHEMBUR / THANE / MULUND/ NERUL / POWAI
TOPIC: (ANSWER KEY)
1. (D)
t BuO
CHClBrI C
H Cl
carbene
Carbenes with phenols show Reimer – Tiemann reaction, with 1o amines carbylamine reaction and
with alkenes a cycloaddition reaction
2. (B)
i Mg Hg in benzene
ii H O
3
H3C OO CH3
H3C OH OHCH3
Pinacol
Pinacol-pinacolone conc H2SO4
rearrangement
Zn Hg
H3C HC
conc.HCl 3
H3C H3C O
3. (A)
In zeisels method of estimation of ethers. The ether is treated with HI and then the iodide with
AgNO3 to give a yellow ppt of AgI
R OR HI ROH RI
RI AgNO3 AgI
Yellow ppt
Mol weight of C9 H12O3 168
1.68
No. of moles taken 0.01mole .
168
4.7
No. of moles of AgI produced 0.02moles
235
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 13
4. (B)
OH
Br
Br
G
H3C
CH3O G + OH
O
HC OH
Br
+
CH3O
G
H
CHO
G
5. (D)
The reaction involved is claisen Rearrangement
R O R O
R R H2C R R H3C
R O
R CH2
R
6. (B)
CH 3 CH 3 CH 3
HI
CH3 CH O CH CH 3
273K
CH 3 CH CH 3 CH 3 CH I
OH
CH 3 CH3
CH3 CH O C CH 3 3
HI
273K
CH 3CH CH3 CH3 C I
OH CH3
CH3 CH 3
HI
CH3 CH 2 O CH CH 3
273K
CH 3 CH 2 I CH3 CH OH
CH 3 CH 2 O C CH 3 3
HI
273K
CH 3CH 2 OH CH 3 3 CI
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 14
7. (BD)
CH3 CH3 OH
OMDM
(A) OH + CH3
CH3 H CH3
CH3 H
CH3 CH3 CH3 CH3
Ag
(B) CH3 C C H3C
AgCl C C
OH Cl OH
O CH3
CH3 CH3
CH3 C C
H
+ C
O
H
CH3
CH3
(both the enantiomers are
obtained)
CH3 CH3 CH3
OH OTs SCH2CH3
TsCl CH3 CH 2SH
(C)
8. (D)
(A) 1, 2 – dioxane being a peroxide is not a very stable compound
Tautomerium
(B) CH 3 CH 2 CH CH OH CH 3CH 2CH 2CHO gives tollen’s reagent test.
O OH
Hemiacetals responds to Tollen’s reagent test
OCH3
acetals do not respond to
CH3 CH 2 4 CH Tollens test
OCH3
(C) Acidic strength in alcohols is 1o 2o 3o
O
OH ONa O C R
COOH basic
COONa COONa
Medium
(D) NaOH O
C
R Cl
NaCl
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 15
9. (ABD)
CH 2 CH O CH 2 CH 3
H O/H
CH 2 CH OH CH3CH 2 OH
2
X Z
Y
CH3CHO
10. (BD)
E1CB occurs on alkyl halides with electron withdrawing groups on carbon
11. (AB)
H3C H3C
H o
(A) CH2 CH
3 3
H3C H3C
H
H3C CH H3C CH
H3C2 2o 3
H3C Me
(B) HBr Me Br
peroxide
12. (D)
conc H 2SO 4
2Cl CH 2 CH 2 OH 413K
ClCH 2CH 2 OCH 2CH 2Cl
NaNH 2
CH 2 CH O CH CH 2
13. (C)
14. (A)
Sol. 13& 14
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 16
15. (B)
16. (C)
Sol. 15&16.
17. (B)
18. (B)
Sol. 17 & 18
Br Br
CH2 Me Me
HBr
+
Me Me Me
Br Br Br
(meso) (C)
(B) optically active
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 17
t-BuONa
t-BuOH
COOEt
CH2 COOEt
EtOOC
CH2 COOEt
(D) (F)
O3 | Zn | H 2O
O
COOEt
COOEt
O
19. (3)
O O
A=
HOOC COOH
O
COOH
B=
HOOC
O
C=
HOOC COOH
COOH
D=
HOOC
E=
O O
O
F=
G=
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 18
20. (4)
Cl Cl NHR2
NHR2
H3C CH3 H3C CH3
C C
N N
intermediate
Cl
NR2 NHR2
H
H3C CH3H3C CH3
CN CN
21. (7)
No. of moles of Grignard reagent used will be 14 and number of moles of ethylalcohol formed are 2.
22. (6)
OH
O OH
OH
OH
OH
OH CH3
23. (5)
Cl O O O OH
CH3COCOOH , CH3 CH C CH3 , CH3 C C CH3 , CH C CH3
O Cl
and CH3 CH C CH3 respond to haloform reaction
OH
24. (4)
Br
Me Me Me Me Me Me
Br
Br
Br
Me
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 19
25. (7)
CH3
CH3 CH3 CH3 CH3
CH3 CH3 CH3
CH3 CH2
CH3 CH3
26. (4)
27. ( A Q; B P, R;C P,S; D P, Q )
28. ( A Q,S; B R; C P, Q; D Q,S )
29. ( A P, Q, R,S; B Q, R,S; C Q,S; D Q,S )
30. ( A P, R,S; B Q, R,S;C Q; D Q, R,S )
CENTERS: MUMBAI / DELHI / AKOLA / KOLKATA / LUCKNOW / NASHIK / GOA / PUNE # 20
You might also like
- Topic 3.1 - Thermal Concepts Formative Assessment NAMEDocument2 pagesTopic 3.1 - Thermal Concepts Formative Assessment NAMEvekid47899No ratings yet
- Sample Acs Final ExamDocument27 pagesSample Acs Final Examjilo100% (2)
- Ape Assignment 3Document7 pagesApe Assignment 3Atharva KulkarniNo ratings yet
- Org 1 PDFDocument4 pagesOrg 1 PDFTanmay KumarNo ratings yet
- JEE Main Organic Compound Containing Halogens Important QuestionsDocument15 pagesJEE Main Organic Compound Containing Halogens Important QuestionsRuchitha VNo ratings yet
- A - 1 (Isomerism, Reaction Mechanism) - Question PaperDocument11 pagesA - 1 (Isomerism, Reaction Mechanism) - Question PaperSachin DedhiaNo ratings yet
- JEE Advanced Alcohols, Phenols and Ethers Important QuestionsDocument17 pagesJEE Advanced Alcohols, Phenols and Ethers Important QuestionsPiyush kumarNo ratings yet
- Haloalkanes SC (Adv)Document10 pagesHaloalkanes SC (Adv)dhikiviyu666No ratings yet
- JEE (Advanced) - 2018 TEST PAPER With Solution: (Exam Date: 20-05-2018) Part-1: ChemistryDocument13 pagesJEE (Advanced) - 2018 TEST PAPER With Solution: (Exam Date: 20-05-2018) Part-1: Chemistrysaravanaajani2012No ratings yet
- Alkyl Halides and Aryl HalidesDocument16 pagesAlkyl Halides and Aryl Halidesvardesh100% (1)
- Chemistry Test PaperDocument12 pagesChemistry Test Papersougata_rintu9598No ratings yet
- Alcohol EtherDocument27 pagesAlcohol EtherslkvalarNo ratings yet
- Assessment - 90 (Chemistry) QDocument6 pagesAssessment - 90 (Chemistry) QParam shahNo ratings yet
- Quiz-Aldehyde & Ketone-VdDocument5 pagesQuiz-Aldehyde & Ketone-VdSatyam AgrawalNo ratings yet
- Alcohols Phenols EtherDocument55 pagesAlcohols Phenols EtherAnanya AgrawalNo ratings yet
- Assessment - 91 (Chemistry) QDocument6 pagesAssessment - 91 (Chemistry) QParam shahNo ratings yet
- Alcohol, Ether and PhenolDocument4 pagesAlcohol, Ether and PhenolKushagra SrivastavaNo ratings yet
- Sheet-5-Hydro CarbonDocument9 pagesSheet-5-Hydro CarbonZooper lNo ratings yet
- Alcohols IIT PACEDocument19 pagesAlcohols IIT PACEDushyant GuptaNo ratings yet
- 1-50 QuestionsDocument48 pages1-50 Questionsbolla reddyNo ratings yet
- CSIR Test Paper - 16Document32 pagesCSIR Test Paper - 16Vineeth V TNo ratings yet
- A - 2 (Isomerism, Reaction Mechantism) - Question PaperDocument14 pagesA - 2 (Isomerism, Reaction Mechantism) - Question PaperSachin DedhiaNo ratings yet
- C - 9. ALDEHYDE KETONES (RCH SIR)ADVDocument10 pagesC - 9. ALDEHYDE KETONES (RCH SIR)ADVrevohe2640No ratings yet
- Neet Sample Paper: Max. Marks: 180 Duration: 3 HrsDocument38 pagesNeet Sample Paper: Max. Marks: 180 Duration: 3 HrsShiv soniNo ratings yet
- Hydrocarbon Practice Sheet Lakshya JEE AIR 20256630e141a403b10018655772Document24 pagesHydrocarbon Practice Sheet Lakshya JEE AIR 20256630e141a403b10018655772peter88105No ratings yet
- COOHDocument4 pagesCOOHtanvip16No ratings yet
- Quiz - Alkyl & Aryl HalidesDocument9 pagesQuiz - Alkyl & Aryl HalidesAdipta GainNo ratings yet
- CHM B44Y Test 3Document15 pagesCHM B44Y Test 3Quốc NguyễnNo ratings yet
- Black Board Problems For JEE Advanced Set-7Document8 pagesBlack Board Problems For JEE Advanced Set-7DikshantNo ratings yet
- Isomerism DPPDocument4 pagesIsomerism DPPRAGHUL MNo ratings yet
- Module AG Sir - Organic Advanced - HydrocarbonsDocument16 pagesModule AG Sir - Organic Advanced - Hydrocarbonskaransharma690No ratings yet
- Goc + IsomerismDocument5 pagesGoc + IsomerismRohail HussainNo ratings yet
- ALCOHOL SHORT TEST-FINALDocument2 pagesALCOHOL SHORT TEST-FINALSparsh SinghNo ratings yet
- Jumbo Home Test-1 - FinalDocument59 pagesJumbo Home Test-1 - FinalSayali SachinNo ratings yet
- Quiz-Alcohol Ether & Phenols-Rsk - RGVDocument6 pagesQuiz-Alcohol Ether & Phenols-Rsk - RGVAtharva GanjuNo ratings yet
- Class Test-6 - Carboxylic Acid - Amines JEE Adv - CC - AnsDocument6 pagesClass Test-6 - Carboxylic Acid - Amines JEE Adv - CC - Ansbruh pogNo ratings yet
- CHM 2201 - Tutorial # 7-2017Document2 pagesCHM 2201 - Tutorial # 7-2017antonio latenNo ratings yet
- Assessment - 97 (Chemistry) QDocument6 pagesAssessment - 97 (Chemistry) QParam shahNo ratings yet
- Alkyl Halides and Aryl Halides - QBDocument23 pagesAlkyl Halides and Aryl Halides - QBNETHAKANI SUJATHA100% (1)
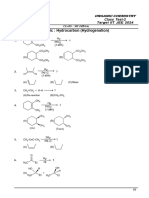
- MT - 6 Paper - II (Question Paper) NewDocument7 pagesMT - 6 Paper - II (Question Paper) Newmaster aexpeckNo ratings yet
- Class Test-2 - Hydrocarbon (Hydrogenation) - Without AnswerDocument8 pagesClass Test-2 - Hydrocarbon (Hydrogenation) - Without AnswerYuvarajNo ratings yet
- Exercise StereochemistryDocument4 pagesExercise StereochemistryPuvaneswary LoganathanNo ratings yet
- JEE Advanced Aldehyde and Ketones Important QuestionsDocument23 pagesJEE Advanced Aldehyde and Ketones Important QuestionsthisissubhaNo ratings yet
- Carboxylic Acid CPPDocument24 pagesCarboxylic Acid CPPGulshan kumarNo ratings yet
- CMB - Mains - 16-07-2022 - After Corrections at 15.58 On 12-07-22Document5 pagesCMB - Mains - 16-07-2022 - After Corrections at 15.58 On 12-07-22Tanush AgarwalNo ratings yet
- Iit Jam Chemistry Core2014Document8 pagesIit Jam Chemistry Core2014Mahendra GanuboyinaNo ratings yet
- Chemistry Paper - I - Question PaperDocument6 pagesChemistry Paper - I - Question PaperKUNALNo ratings yet
- VITEEE Chemistry 2013: - Download FromDocument11 pagesVITEEE Chemistry 2013: - Download FromAnweshMishraNo ratings yet
- JEE Main Hydrocarbons Important Questions (2025)Document15 pagesJEE Main Hydrocarbons Important Questions (2025)amandiwan336No ratings yet
- Quiz-Alkyl Halide-MksDocument6 pagesQuiz-Alkyl Halide-MksSatyam AgrawalNo ratings yet
- 2B-Alcohols, Ethers & Phenols - FINAL - 06!03!14 (86-112)Document27 pages2B-Alcohols, Ethers & Phenols - FINAL - 06!03!14 (86-112)udaysrinivasNo ratings yet
- Aliphatic HydrocarbonsDocument11 pagesAliphatic HydrocarbonsRishabhNo ratings yet
- Manzil Alchol Phenol and Ether Practice Sheet-1: H C H C H C OHDocument6 pagesManzil Alchol Phenol and Ether Practice Sheet-1: H C H C H C OHRenu KalvanshNo ratings yet
- 2008Document7 pages2008prakhar vishwakarmaNo ratings yet
- C - 8. (MAIN) (ALKYL & ARYL HALLIDE, ALCOHOL ETHERS & PHENOLS)Document10 pagesC - 8. (MAIN) (ALKYL & ARYL HALLIDE, ALCOHOL ETHERS & PHENOLS)revohe2640No ratings yet
- QUIZ-ALKYL_HALIDE-MKSDocument6 pagesQUIZ-ALKYL_HALIDE-MKSSreyan MajumderNo ratings yet
- Sheet-3-Hydro CarbonDocument8 pagesSheet-3-Hydro CarbonZooper lNo ratings yet
- CH 44 Organic Reactions - Supp Ex 1 (Updated)Document4 pagesCH 44 Organic Reactions - Supp Ex 1 (Updated)伊貝P-No ratings yet
- The Carbonyl Compound-3Document3 pagesThe Carbonyl Compound-3devender singhNo ratings yet
- 2007Document9 pages2007Anil KumarNo ratings yet
- Stainless Steel Razni StandardiDocument6 pagesStainless Steel Razni StandardiAnonymous HcfA1RNo ratings yet
- Calendering ProcessDocument14 pagesCalendering ProcessRony ShielaNo ratings yet
- Pharmaceutical Formulations-Suspensions and Solutions PDFDocument4 pagesPharmaceutical Formulations-Suspensions and Solutions PDFERICA LILIAN ESGUERRA GILNo ratings yet
- Lutensit a-ES MSAS enDocument6 pagesLutensit a-ES MSAS ensamotnik81No ratings yet
- ANSYS-Based Detailed Thermo-Mechanical Modeling of Complex Thermoelectric Power DesignsDocument3 pagesANSYS-Based Detailed Thermo-Mechanical Modeling of Complex Thermoelectric Power DesignsablueleafNo ratings yet
- Ultrasonic Machining Process, Working Principles & AdvantagesDocument3 pagesUltrasonic Machining Process, Working Principles & AdvantagesCiprian-Dumitru CiofuNo ratings yet
- 2.2 Carbohydrate Metabolism Part 2Document13 pages2.2 Carbohydrate Metabolism Part 2John Louis PulidoNo ratings yet
- Lecture 1Document7 pagesLecture 1Chaudhry FahadNo ratings yet
- Section 9 - Welding ControlDocument6 pagesSection 9 - Welding ControlYasser Hammad MohamedNo ratings yet
- Rajah 8.2 Menunjukkan Satu Cawan Bertutup. Cawan Ini Tidak Sesuai Untuk Mengekalkan Suhu Bagi Minuman Panas Dalam Masa Yang LamaDocument10 pagesRajah 8.2 Menunjukkan Satu Cawan Bertutup. Cawan Ini Tidak Sesuai Untuk Mengekalkan Suhu Bagi Minuman Panas Dalam Masa Yang Lamajgd2080No ratings yet
- Industrial Chemistry PDFDocument213 pagesIndustrial Chemistry PDFLucio Peña Zarate100% (1)
- Turbo Machines Module 01 Q No 2a & 2bDocument26 pagesTurbo Machines Module 01 Q No 2a & 2bShadow KnightNo ratings yet
- Advances in Chemical Reactivity Evaluation at Dow: Marabeth Holsinger, PH.DDocument40 pagesAdvances in Chemical Reactivity Evaluation at Dow: Marabeth Holsinger, PH.DsushantNo ratings yet
- Physics A Level DefinitionsDocument12 pagesPhysics A Level DefinitionsNaillah Saba100% (1)
- Melpool 70GDocument21 pagesMelpool 70GAnjosNo ratings yet
- Vol 10 No 2 2017 9 185-194Document11 pagesVol 10 No 2 2017 9 185-194Đào Đình NamNo ratings yet
- ElectroplatingDocument23 pagesElectroplatingJayne Kazandra P. Ortega67% (3)
- Fabrication of Components at Dillinger Huette Heavy FabricationDocument82 pagesFabrication of Components at Dillinger Huette Heavy Fabrication66apenlullenNo ratings yet
- CAPE 1 BIOLOGY - WaterDocument72 pagesCAPE 1 BIOLOGY - WaterTamicka BonnickNo ratings yet
- Week 4 Applying Lime To Correct Soil AcidityDocument19 pagesWeek 4 Applying Lime To Correct Soil AcidityPrincess Genotiva GerancoNo ratings yet
- PHD A Harris Fuels 05-02-12Document279 pagesPHD A Harris Fuels 05-02-12David Jiménez MenaNo ratings yet
- Heat Exchanger ManualDocument14 pagesHeat Exchanger ManualITSNo ratings yet
- Composite Materials ReportDocument47 pagesComposite Materials ReportPatrickNo ratings yet
- ARTICLEJMSrev Macromolchemphys HEMA1992Document35 pagesARTICLEJMSrev Macromolchemphys HEMA1992Milda Syakilla ZaharaNo ratings yet
- Machining MechAnalysis CHPT 3Document38 pagesMachining MechAnalysis CHPT 3Brahim MouchaneNo ratings yet
- Electrolysis of Copper (II) SulphateDocument4 pagesElectrolysis of Copper (II) Sulphateamber_strauss100% (2)
- Modeling of Ambient Air Pollutants ThrouDocument4 pagesModeling of Ambient Air Pollutants ThrouRahul KumarNo ratings yet
- Plasma Orbital Expansion Electrons WaterDocument158 pagesPlasma Orbital Expansion Electrons WaterVincent J. CataldiNo ratings yet