Professional Documents
Culture Documents
Hydrocarbon _ DPP 04 __ Lakshya JEE 2025
Hydrocarbon _ DPP 04 __ Lakshya JEE 2025
Uploaded by
amaankhansethiCopyright:
Available Formats
You might also like
- Turgeon - Hematology Study QuestionsDocument65 pagesTurgeon - Hematology Study QuestionsGertrudeshane IletoNo ratings yet
- Swiggy Case Study - DE RejectDocument2 pagesSwiggy Case Study - DE RejectNirmal Kumawat0% (1)
- Chapter 13Document10 pagesChapter 13janineNo ratings yet
- Sample Paper - Campus Recruitment Test-Chemistry Medical PDFDocument4 pagesSample Paper - Campus Recruitment Test-Chemistry Medical PDFAbhijeet Parkhi50% (2)
- 01 - 2018 - IEEE - Robotics For Sugarcane Cultivation Analysis of Billet Quality Using Computer VisionDocument8 pages01 - 2018 - IEEE - Robotics For Sugarcane Cultivation Analysis of Billet Quality Using Computer VisionaryoNo ratings yet
- SRG Organic Chemistry AssignmentDocument22 pagesSRG Organic Chemistry Assignmenttrt4s5bmqwNo ratings yet
- 006) Hydrocarbon - DPP 02 Yakeen 20 2024 (Legend)Document2 pages006) Hydrocarbon - DPP 02 Yakeen 20 2024 (Legend)Himanshu PuriNo ratings yet
- Hydrocarbons _ Practice SheetDocument6 pagesHydrocarbons _ Practice SheetthebajranggunNo ratings yet
- Ques&Ans NEET2020 (Code E4) Answer KeyDocument21 pagesQues&Ans NEET2020 (Code E4) Answer Keyseetharaman8341No ratings yet
- Haloalkane & Haloarene _ DPP 05Document3 pagesHaloalkane & Haloarene _ DPP 05shaanmukherjee2020No ratings yet
- MCQ Based Paper (Isc Pattern) : Mark: 70 Time 90 MinDocument4 pagesMCQ Based Paper (Isc Pattern) : Mark: 70 Time 90 MinDisha MukherjeeNo ratings yet
- Class 12th NEET Chemistry Sample Paper 4 Alka Meena Mem Vijay Kr. Kashyap Questions Final 1Document5 pagesClass 12th NEET Chemistry Sample Paper 4 Alka Meena Mem Vijay Kr. Kashyap Questions Final 1revathiprasady1987No ratings yet
- Chemical Equilibrium - DPP 01 (Of Lec 02) - Arjuna JEE 2024Document2 pagesChemical Equilibrium - DPP 01 (Of Lec 02) - Arjuna JEE 2024nrashmi743No ratings yet
- ISC 2023 Chemistry Question PaperDocument8 pagesISC 2023 Chemistry Question PaperT rud0No ratings yet
- Electrolysis QPDocument14 pagesElectrolysis QPSartaj KabirNo ratings yet
- GOC - DPP 12 (Extra DPP) - Prayas JEE 2024Document3 pagesGOC - DPP 12 (Extra DPP) - Prayas JEE 2024smrutichandanmohantaNo ratings yet
- KVS LR M.SDocument5 pagesKVS LR M.Syogendra.pandey03No ratings yet
- NEET 2020 Question Paper With Answer Key E4 F4 G4 H4Document101 pagesNEET 2020 Question Paper With Answer Key E4 F4 G4 H4Sanket SawantNo ratings yet
- NEET 2020 Question Paper Set E3 PDFDocument21 pagesNEET 2020 Question Paper Set E3 PDFZalaslad HackerNo ratings yet
- Born-Haber Cycles QuestionsDocument5 pagesBorn-Haber Cycles QuestionsotoczylowskaNo ratings yet
- NEET Full Test - 4Document7 pagesNEET Full Test - 4Yash KapoorNo ratings yet
- ISC 2015 Chemistry Paper 1 Theory Solved PaperDocument27 pagesISC 2015 Chemistry Paper 1 Theory Solved PaperSatish Chandra BhartiNo ratings yet
- Hydrocarbons EX 1 - Tatva ModuleDocument19 pagesHydrocarbons EX 1 - Tatva ModuleS. S. Naren Karthik ShunmugamNo ratings yet
- Aldehydes and Ketones - Practice Sheet & Solution 02 (Of Lecture 07) - Drona - 12th - NEET 2023 2Document5 pagesAldehydes and Ketones - Practice Sheet & Solution 02 (Of Lecture 07) - Drona - 12th - NEET 2023 2Sara NimareNo ratings yet
- Neet Model - 6Document20 pagesNeet Model - 6krishna kamleshNo ratings yet
- Chemistry 101 SPRING 2005: Final Form B SECTIONS 501-511 Dr. Keeney-KennicuttDocument15 pagesChemistry 101 SPRING 2005: Final Form B SECTIONS 501-511 Dr. Keeney-KennicuttKalaiarasu SelvarajanNo ratings yet
- 4-Poll ChemistryDocument2 pages4-Poll Chemistryprathikmstar55No ratings yet
- Nakha: Important InstructionsDocument24 pagesNakha: Important InstructionsImran ChoudhuryNo ratings yet
- EM - Sci - G10 - T3 - I, II PP Ans - 2018Document21 pagesEM - Sci - G10 - T3 - I, II PP Ans - 2018KaveenNo ratings yet
- JEE Main Full Mock Test 8Document10 pagesJEE Main Full Mock Test 8Aditya SinghNo ratings yet
- ChemistryDocument12 pagesChemistryAditya SinghNo ratings yet
- Sample Paper - Campus Recruitment Test-Chemistry MedicalDocument4 pagesSample Paper - Campus Recruitment Test-Chemistry MedicalRani PandeyNo ratings yet
- D and F Block Elements - DPP 03 (Of Lec-04) - Yakeen 2.0 2024 (Legend)Document2 pagesD and F Block Elements - DPP 03 (Of Lec-04) - Yakeen 2.0 2024 (Legend)Aayushi gargNo ratings yet
- Unit Unit Unit Unit Unit: I. Multiple Choice Questions (Type-I)Document13 pagesUnit Unit Unit Unit Unit: I. Multiple Choice Questions (Type-I)Om AgrawalNo ratings yet
- Chemistry Class Test: AIIMS - 10.05.2019Document8 pagesChemistry Class Test: AIIMS - 10.05.2019DrNaresh SahuNo ratings yet
- Gujarat Technological University: InstructionsDocument2 pagesGujarat Technological University: Instructionsadi411No ratings yet
- Che 4 Sure Shot BothDocument24 pagesChe 4 Sure Shot BothPAVEENA KNo ratings yet
- BSC 4 Sem Chemistry Ay 2201 S 2022Document4 pagesBSC 4 Sem Chemistry Ay 2201 S 2022patelwajid772No ratings yet
- XI-Chemistry Chapter Test-9-HydrogenDocument3 pagesXI-Chemistry Chapter Test-9-Hydrogencakof67215No ratings yet
- Jee Main 2024 Shift1 Chemistry Jan27Document4 pagesJee Main 2024 Shift1 Chemistry Jan27Krishna - the GeniusNo ratings yet
- 615d6ef770dc4 Alkane ExerciseDocument6 pages615d6ef770dc4 Alkane ExerciseNguyên Phạm ThuNo ratings yet
- Organic Chemistry MergeDocument397 pagesOrganic Chemistry Mergekd79329No ratings yet
- National Eligibility Cum Entrance Test Neet (Ug) ,: Phase-2 (CODE:BB-QQ-XX)Document19 pagesNational Eligibility Cum Entrance Test Neet (Ug) ,: Phase-2 (CODE:BB-QQ-XX)Vivek SinghNo ratings yet
- Annual Examination (2021-22) : CHEMISTRY - (043) Class - XiDocument4 pagesAnnual Examination (2021-22) : CHEMISTRY - (043) Class - XiNitin HansNo ratings yet
- (Total 1 Mark) : IB Questionbank Chemistry 1Document4 pages(Total 1 Mark) : IB Questionbank Chemistry 1Talha HasanNo ratings yet
- Chemical Kinetics - DPP 09 - Lakshya JEE 2024Document2 pagesChemical Kinetics - DPP 09 - Lakshya JEE 2024Hrishith SavirNo ratings yet
- First Year Higher Secondary Examination June 2022 CHEMISTRY - ANSWER KEY (Unofficial)Document7 pagesFirst Year Higher Secondary Examination June 2022 CHEMISTRY - ANSWER KEY (Unofficial)Ashkar AshrafNo ratings yet
- Mathongo Jee Main 2015Document28 pagesMathongo Jee Main 2015rishithhr rajeevNo ratings yet
- Organic Chemistry - Top 500 Question Bank For JEE Main by MathonGo PDFDocument65 pagesOrganic Chemistry - Top 500 Question Bank For JEE Main by MathonGo PDFDeepak BinwalNo ratings yet
- Chemistry Complete PaperDocument5 pagesChemistry Complete PaperNitin HansNo ratings yet
- Organic Chemistry - Some Basic Principles and Techniques _ Practice Sheet (1)Document6 pagesOrganic Chemistry - Some Basic Principles and Techniques _ Practice Sheet (1)thebajranggunNo ratings yet
- Alkenes 2 QP AnsDocument11 pagesAlkenes 2 QP AnshhheeeNo ratings yet
- Microsoft Word - Redox Review - IB-Short QuestionsDocument8 pagesMicrosoft Word - Redox Review - IB-Short Questionsmccano99No ratings yet
- S6 Test 5Document7 pagesS6 Test 5XD XDNo ratings yet
- Chem - 24.03.2020 - Full Test - Naresh Sir: ChemistryDocument7 pagesChem - 24.03.2020 - Full Test - Naresh Sir: ChemistryDrNaresh SahuNo ratings yet
- Re - Aipmt 2015 Test Paper With Answer & Solution (Held On Saturday 25 JULY, 2015Document19 pagesRe - Aipmt 2015 Test Paper With Answer & Solution (Held On Saturday 25 JULY, 2015Jessica ShamoonNo ratings yet
- 穩底班Quiz topic 8 part 1-3 10份Document18 pages穩底班Quiz topic 8 part 1-3 10份pinkychan0126No ratings yet
- Che CP CASS NEET-UG (Ques) ENG 2PDocument3 pagesChe CP CASS NEET-UG (Ques) ENG 2PRaktim FactoryNo ratings yet
- Xii Ak PB1Document9 pagesXii Ak PB1Keshav PandeyNo ratings yet
- Kcet Model QP - Chemistry - 15-04-2024Document8 pagesKcet Model QP - Chemistry - 15-04-2024mahadevipatil.patil04No ratings yet
- Complete Organic Chemistry - Topper Assignment 01Document6 pagesComplete Organic Chemistry - Topper Assignment 01gssharmaa2017No ratings yet
- Redox Reactions _ Practice SheetDocument5 pagesRedox Reactions _ Practice SheetthebajranggunNo ratings yet
- Graphene Oxide: Fundamentals and ApplicationsFrom EverandGraphene Oxide: Fundamentals and ApplicationsAyrat M. DimievNo ratings yet
- Palladium Reagents and Catalysts: New Perspectives for the 21st CenturyFrom EverandPalladium Reagents and Catalysts: New Perspectives for the 21st CenturyNo ratings yet
- Eng Test AllDocument2 pagesEng Test AllWaseem AbbasNo ratings yet
- Fs 276 LFV 02Document12 pagesFs 276 LFV 02Pablo MgNo ratings yet
- DS Lab4 UnlockedDocument2 pagesDS Lab4 UnlockedSri JulapallyNo ratings yet
- Boron For Liquid Fuel Engines-A Review On Synthesis, Dispersion Stability in Liquid Fuel and Combustion AspectsDocument29 pagesBoron For Liquid Fuel Engines-A Review On Synthesis, Dispersion Stability in Liquid Fuel and Combustion Aspectsmaa bloNo ratings yet
- APM2613 - 202 - 0 - 2022 (A02 Feedback)Document21 pagesAPM2613 - 202 - 0 - 2022 (A02 Feedback)Darrel van OnselenNo ratings yet
- Lums Sse Test Sample Paper.Document16 pagesLums Sse Test Sample Paper.999 X100% (1)
- Deep FoundationsDocument23 pagesDeep FoundationsMushaid Ali SyedNo ratings yet
- Theory and Its Implication by Eyuel KokebDocument6 pagesTheory and Its Implication by Eyuel KokebEyuel KokebNo ratings yet
- Peel Synchro GuidelinesDocument20 pagesPeel Synchro GuidelinesSyed Imam100% (1)
- Nadia Karima Izzaty 1306369466 Dept. Teknik Sipil: Time (H) Rate (In/h) Direct Runoff (CFS) RM (In.)Document7 pagesNadia Karima Izzaty 1306369466 Dept. Teknik Sipil: Time (H) Rate (In/h) Direct Runoff (CFS) RM (In.)Nadia KarimaNo ratings yet
- Modules Music 6Document23 pagesModules Music 6Diether Mercado PaduaNo ratings yet
- Circle CPPDocument7 pagesCircle CPPAnugrah AgrawalNo ratings yet
- Domain Specific Languages in Practice: International Journal of Computer Applications April 2015Document5 pagesDomain Specific Languages in Practice: International Journal of Computer Applications April 2015Radian Nata KristiantoNo ratings yet
- Module 5n6 Answer Sheet Is Anatomy and Physiology (Abegail C. Relunia - Bsed 3c-Science)Document6 pagesModule 5n6 Answer Sheet Is Anatomy and Physiology (Abegail C. Relunia - Bsed 3c-Science)Abegail ReluniaNo ratings yet
- Nav2013 Enus Upgrade 03Document56 pagesNav2013 Enus Upgrade 03Wagz KagiriNo ratings yet
- Polycythaemia Vera Presenting As Massive Splenic Infarction and Liquefaction PDFDocument2 pagesPolycythaemia Vera Presenting As Massive Splenic Infarction and Liquefaction PDFtedmozbiNo ratings yet
- Semi-Active Fuzzy Control of A Wind-Excited Tall Building Using Multi-Objectivegenetic AlgorithmDocument16 pagesSemi-Active Fuzzy Control of A Wind-Excited Tall Building Using Multi-Objectivegenetic Algorithmalexkoch2No ratings yet
- A Creep Life Assessment Method For Boiler Pipes Using Small Punch Creep TestDocument6 pagesA Creep Life Assessment Method For Boiler Pipes Using Small Punch Creep TestAndrea CalderaNo ratings yet
- Transportation Engineering 1 Question Bank - Mumbai UniversityDocument10 pagesTransportation Engineering 1 Question Bank - Mumbai UniversityMegha Thomas0% (1)
- The Bass Player's Companion: A G Uide To S Ound, C Oncepts, and T Echnique For The E Lectric BassDocument55 pagesThe Bass Player's Companion: A G Uide To S Ound, C Oncepts, and T Echnique For The E Lectric BassBassman BronNo ratings yet
- AI - AI417DE01 Lab - Week 09-22102363Document8 pagesAI - AI417DE01 Lab - Week 09-22102363thinhbui1907No ratings yet
- Data-Driven Unit Tests: (Fact) and (Theory)Document6 pagesData-Driven Unit Tests: (Fact) and (Theory)Ronesh KumarNo ratings yet
- A Critical Review of Heat Transfer Through Helical Coils of Circular Cross Section PDFDocument9 pagesA Critical Review of Heat Transfer Through Helical Coils of Circular Cross Section PDFManuel CortesNo ratings yet
- Vinay Sharma - inDocument3 pagesVinay Sharma - inVinay SharmaNo ratings yet
- 2009 06 23 CAD CMX Technical Report 164Document99 pages2009 06 23 CAD CMX Technical Report 164César GuerreroNo ratings yet
- Datasheet EE800Document3 pagesDatasheet EE800AdyMichaelNo ratings yet
Hydrocarbon _ DPP 04 __ Lakshya JEE 2025
Hydrocarbon _ DPP 04 __ Lakshya JEE 2025
Uploaded by
amaankhansethiCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Hydrocarbon _ DPP 04 __ Lakshya JEE 2025
Hydrocarbon _ DPP 04 __ Lakshya JEE 2025
Uploaded by
amaankhansethiCopyright:
Available Formats
1
LAKSHYA JEE 2025
Hydrocarbon DPP-4
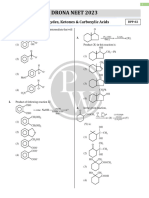
1. Which product is obtained at the anode by the 6. Assertion : Iodination of alkanes is reversible.
electrolysis of sodium butanoate : Reason : The byproduct HI is a strong reducing
(1) Butane + CO2 (2) Pentane + H2 agent, and reduces alkyl iodide.
(3) Hexane + CO2 (4) Hexane + H2 (1) Assertion is correct, reason is correct; reason is
a correct explanation for assertion.
2. In the reaction (2) Assertion is correct, reason is correct; reason is
CH3COONa + H 2O ⎯⎯⎯⎯
electrolysis
→ not a correct explanation for assertion
(3) Assertion is correct, reason is incorrect
Read the statement given below and choose the
(4) Assertion is incorrect, reason is correct.
option containing correct statement -
(a) Gas produced at anode gives lime water test
7. How many monochloro derivatives are possible by
(b) One of the gas produced at cathode gives pop
neopentane?
sound test
(1) 1 (2) 3
(c) The pH of the reaction mixture increases as
(3) 4 (4) 6
reaction progresses.
(d) Alkane produced in this reaction can show
conformational isomerism.
(1) (a), (b), (c) and (d) 8. major product is :
(2) (a), (d)
(3) (b), (c), (d)
(1) (2)
(4) (a), (b), (c)
3. Which of the following statements are correct ? (3) (4)
(i) The rate of reactivity of alkanes with halogens is
F2 > Cl2 > Br2 > I2.
(ii) Rate of replacement of hydrogens of alkanes is
3° > 2º > 1°
9. ⎯⎯⎯
Cl2 /hv
→ Monochloro derivative (X); X
(iii) Fluorination of alkanes is a very slow process. is :
(iv) Iodination of alkanes is too violent to be (1) 6 (2) 7
controlled. (3) 8 (3) 5
(1) (i), (ii) and (iii) (2) (i) and (ii)
(3) (ii) and (iii) (4) (i) and (iv)
10. Decline can be prepared by
Cl
4. No. of required O2 mole for complete combustion of
one mole of propane - (1) ⎯⎯⎯
Na
Ether
→
(1) 7 (2) 5 Cl
(3) 16 (4) 10 COONa
(2) ⎯⎯⎯⎯→
Electrolysis
5. Assertion : CH4 does not react with Cl2 in dark.
Reason : Chlorination of CH4 takes place in sunlight. COONa
(1) Assertion is correct, reason is correct; reason is (3) Cl
a correct explanation for assertion. ⎯⎯⎯⎯
Mg(1eq.)
→
(2) Assertion is correct, reason is correct; reason is
Cl
not a correct explanation for assertion
(3) Assertion is correct, reason is incorrect (4) Cl Cl ⎯⎯⎯⎯
Na/Ether
→
(4) Assertion is incorrect, reason is correct.
2
Answer Key
1. (3)
2. (1)
3. (2)
4. (2)
5. (2)
6. (1)
7. (1)
8. (2)
9. (3)
10. (2)
PW Web/App - https://smart.link/7wwosivoicgd4
Library- https://smart.link/sdfez8ejd80if
You might also like
- Turgeon - Hematology Study QuestionsDocument65 pagesTurgeon - Hematology Study QuestionsGertrudeshane IletoNo ratings yet
- Swiggy Case Study - DE RejectDocument2 pagesSwiggy Case Study - DE RejectNirmal Kumawat0% (1)
- Chapter 13Document10 pagesChapter 13janineNo ratings yet
- Sample Paper - Campus Recruitment Test-Chemistry Medical PDFDocument4 pagesSample Paper - Campus Recruitment Test-Chemistry Medical PDFAbhijeet Parkhi50% (2)
- 01 - 2018 - IEEE - Robotics For Sugarcane Cultivation Analysis of Billet Quality Using Computer VisionDocument8 pages01 - 2018 - IEEE - Robotics For Sugarcane Cultivation Analysis of Billet Quality Using Computer VisionaryoNo ratings yet
- SRG Organic Chemistry AssignmentDocument22 pagesSRG Organic Chemistry Assignmenttrt4s5bmqwNo ratings yet
- 006) Hydrocarbon - DPP 02 Yakeen 20 2024 (Legend)Document2 pages006) Hydrocarbon - DPP 02 Yakeen 20 2024 (Legend)Himanshu PuriNo ratings yet
- Hydrocarbons _ Practice SheetDocument6 pagesHydrocarbons _ Practice SheetthebajranggunNo ratings yet
- Ques&Ans NEET2020 (Code E4) Answer KeyDocument21 pagesQues&Ans NEET2020 (Code E4) Answer Keyseetharaman8341No ratings yet
- Haloalkane & Haloarene _ DPP 05Document3 pagesHaloalkane & Haloarene _ DPP 05shaanmukherjee2020No ratings yet
- MCQ Based Paper (Isc Pattern) : Mark: 70 Time 90 MinDocument4 pagesMCQ Based Paper (Isc Pattern) : Mark: 70 Time 90 MinDisha MukherjeeNo ratings yet
- Class 12th NEET Chemistry Sample Paper 4 Alka Meena Mem Vijay Kr. Kashyap Questions Final 1Document5 pagesClass 12th NEET Chemistry Sample Paper 4 Alka Meena Mem Vijay Kr. Kashyap Questions Final 1revathiprasady1987No ratings yet
- Chemical Equilibrium - DPP 01 (Of Lec 02) - Arjuna JEE 2024Document2 pagesChemical Equilibrium - DPP 01 (Of Lec 02) - Arjuna JEE 2024nrashmi743No ratings yet
- ISC 2023 Chemistry Question PaperDocument8 pagesISC 2023 Chemistry Question PaperT rud0No ratings yet
- Electrolysis QPDocument14 pagesElectrolysis QPSartaj KabirNo ratings yet
- GOC - DPP 12 (Extra DPP) - Prayas JEE 2024Document3 pagesGOC - DPP 12 (Extra DPP) - Prayas JEE 2024smrutichandanmohantaNo ratings yet
- KVS LR M.SDocument5 pagesKVS LR M.Syogendra.pandey03No ratings yet
- NEET 2020 Question Paper With Answer Key E4 F4 G4 H4Document101 pagesNEET 2020 Question Paper With Answer Key E4 F4 G4 H4Sanket SawantNo ratings yet
- NEET 2020 Question Paper Set E3 PDFDocument21 pagesNEET 2020 Question Paper Set E3 PDFZalaslad HackerNo ratings yet
- Born-Haber Cycles QuestionsDocument5 pagesBorn-Haber Cycles QuestionsotoczylowskaNo ratings yet
- NEET Full Test - 4Document7 pagesNEET Full Test - 4Yash KapoorNo ratings yet
- ISC 2015 Chemistry Paper 1 Theory Solved PaperDocument27 pagesISC 2015 Chemistry Paper 1 Theory Solved PaperSatish Chandra BhartiNo ratings yet
- Hydrocarbons EX 1 - Tatva ModuleDocument19 pagesHydrocarbons EX 1 - Tatva ModuleS. S. Naren Karthik ShunmugamNo ratings yet
- Aldehydes and Ketones - Practice Sheet & Solution 02 (Of Lecture 07) - Drona - 12th - NEET 2023 2Document5 pagesAldehydes and Ketones - Practice Sheet & Solution 02 (Of Lecture 07) - Drona - 12th - NEET 2023 2Sara NimareNo ratings yet
- Neet Model - 6Document20 pagesNeet Model - 6krishna kamleshNo ratings yet
- Chemistry 101 SPRING 2005: Final Form B SECTIONS 501-511 Dr. Keeney-KennicuttDocument15 pagesChemistry 101 SPRING 2005: Final Form B SECTIONS 501-511 Dr. Keeney-KennicuttKalaiarasu SelvarajanNo ratings yet
- 4-Poll ChemistryDocument2 pages4-Poll Chemistryprathikmstar55No ratings yet
- Nakha: Important InstructionsDocument24 pagesNakha: Important InstructionsImran ChoudhuryNo ratings yet
- EM - Sci - G10 - T3 - I, II PP Ans - 2018Document21 pagesEM - Sci - G10 - T3 - I, II PP Ans - 2018KaveenNo ratings yet
- JEE Main Full Mock Test 8Document10 pagesJEE Main Full Mock Test 8Aditya SinghNo ratings yet
- ChemistryDocument12 pagesChemistryAditya SinghNo ratings yet
- Sample Paper - Campus Recruitment Test-Chemistry MedicalDocument4 pagesSample Paper - Campus Recruitment Test-Chemistry MedicalRani PandeyNo ratings yet
- D and F Block Elements - DPP 03 (Of Lec-04) - Yakeen 2.0 2024 (Legend)Document2 pagesD and F Block Elements - DPP 03 (Of Lec-04) - Yakeen 2.0 2024 (Legend)Aayushi gargNo ratings yet
- Unit Unit Unit Unit Unit: I. Multiple Choice Questions (Type-I)Document13 pagesUnit Unit Unit Unit Unit: I. Multiple Choice Questions (Type-I)Om AgrawalNo ratings yet
- Chemistry Class Test: AIIMS - 10.05.2019Document8 pagesChemistry Class Test: AIIMS - 10.05.2019DrNaresh SahuNo ratings yet
- Gujarat Technological University: InstructionsDocument2 pagesGujarat Technological University: Instructionsadi411No ratings yet
- Che 4 Sure Shot BothDocument24 pagesChe 4 Sure Shot BothPAVEENA KNo ratings yet
- BSC 4 Sem Chemistry Ay 2201 S 2022Document4 pagesBSC 4 Sem Chemistry Ay 2201 S 2022patelwajid772No ratings yet
- XI-Chemistry Chapter Test-9-HydrogenDocument3 pagesXI-Chemistry Chapter Test-9-Hydrogencakof67215No ratings yet
- Jee Main 2024 Shift1 Chemistry Jan27Document4 pagesJee Main 2024 Shift1 Chemistry Jan27Krishna - the GeniusNo ratings yet
- 615d6ef770dc4 Alkane ExerciseDocument6 pages615d6ef770dc4 Alkane ExerciseNguyên Phạm ThuNo ratings yet
- Organic Chemistry MergeDocument397 pagesOrganic Chemistry Mergekd79329No ratings yet
- National Eligibility Cum Entrance Test Neet (Ug) ,: Phase-2 (CODE:BB-QQ-XX)Document19 pagesNational Eligibility Cum Entrance Test Neet (Ug) ,: Phase-2 (CODE:BB-QQ-XX)Vivek SinghNo ratings yet
- Annual Examination (2021-22) : CHEMISTRY - (043) Class - XiDocument4 pagesAnnual Examination (2021-22) : CHEMISTRY - (043) Class - XiNitin HansNo ratings yet
- (Total 1 Mark) : IB Questionbank Chemistry 1Document4 pages(Total 1 Mark) : IB Questionbank Chemistry 1Talha HasanNo ratings yet
- Chemical Kinetics - DPP 09 - Lakshya JEE 2024Document2 pagesChemical Kinetics - DPP 09 - Lakshya JEE 2024Hrishith SavirNo ratings yet
- First Year Higher Secondary Examination June 2022 CHEMISTRY - ANSWER KEY (Unofficial)Document7 pagesFirst Year Higher Secondary Examination June 2022 CHEMISTRY - ANSWER KEY (Unofficial)Ashkar AshrafNo ratings yet
- Mathongo Jee Main 2015Document28 pagesMathongo Jee Main 2015rishithhr rajeevNo ratings yet
- Organic Chemistry - Top 500 Question Bank For JEE Main by MathonGo PDFDocument65 pagesOrganic Chemistry - Top 500 Question Bank For JEE Main by MathonGo PDFDeepak BinwalNo ratings yet
- Chemistry Complete PaperDocument5 pagesChemistry Complete PaperNitin HansNo ratings yet
- Organic Chemistry - Some Basic Principles and Techniques _ Practice Sheet (1)Document6 pagesOrganic Chemistry - Some Basic Principles and Techniques _ Practice Sheet (1)thebajranggunNo ratings yet
- Alkenes 2 QP AnsDocument11 pagesAlkenes 2 QP AnshhheeeNo ratings yet
- Microsoft Word - Redox Review - IB-Short QuestionsDocument8 pagesMicrosoft Word - Redox Review - IB-Short Questionsmccano99No ratings yet
- S6 Test 5Document7 pagesS6 Test 5XD XDNo ratings yet
- Chem - 24.03.2020 - Full Test - Naresh Sir: ChemistryDocument7 pagesChem - 24.03.2020 - Full Test - Naresh Sir: ChemistryDrNaresh SahuNo ratings yet
- Re - Aipmt 2015 Test Paper With Answer & Solution (Held On Saturday 25 JULY, 2015Document19 pagesRe - Aipmt 2015 Test Paper With Answer & Solution (Held On Saturday 25 JULY, 2015Jessica ShamoonNo ratings yet
- 穩底班Quiz topic 8 part 1-3 10份Document18 pages穩底班Quiz topic 8 part 1-3 10份pinkychan0126No ratings yet
- Che CP CASS NEET-UG (Ques) ENG 2PDocument3 pagesChe CP CASS NEET-UG (Ques) ENG 2PRaktim FactoryNo ratings yet
- Xii Ak PB1Document9 pagesXii Ak PB1Keshav PandeyNo ratings yet
- Kcet Model QP - Chemistry - 15-04-2024Document8 pagesKcet Model QP - Chemistry - 15-04-2024mahadevipatil.patil04No ratings yet
- Complete Organic Chemistry - Topper Assignment 01Document6 pagesComplete Organic Chemistry - Topper Assignment 01gssharmaa2017No ratings yet
- Redox Reactions _ Practice SheetDocument5 pagesRedox Reactions _ Practice SheetthebajranggunNo ratings yet
- Graphene Oxide: Fundamentals and ApplicationsFrom EverandGraphene Oxide: Fundamentals and ApplicationsAyrat M. DimievNo ratings yet
- Palladium Reagents and Catalysts: New Perspectives for the 21st CenturyFrom EverandPalladium Reagents and Catalysts: New Perspectives for the 21st CenturyNo ratings yet
- Eng Test AllDocument2 pagesEng Test AllWaseem AbbasNo ratings yet
- Fs 276 LFV 02Document12 pagesFs 276 LFV 02Pablo MgNo ratings yet
- DS Lab4 UnlockedDocument2 pagesDS Lab4 UnlockedSri JulapallyNo ratings yet
- Boron For Liquid Fuel Engines-A Review On Synthesis, Dispersion Stability in Liquid Fuel and Combustion AspectsDocument29 pagesBoron For Liquid Fuel Engines-A Review On Synthesis, Dispersion Stability in Liquid Fuel and Combustion Aspectsmaa bloNo ratings yet
- APM2613 - 202 - 0 - 2022 (A02 Feedback)Document21 pagesAPM2613 - 202 - 0 - 2022 (A02 Feedback)Darrel van OnselenNo ratings yet
- Lums Sse Test Sample Paper.Document16 pagesLums Sse Test Sample Paper.999 X100% (1)
- Deep FoundationsDocument23 pagesDeep FoundationsMushaid Ali SyedNo ratings yet
- Theory and Its Implication by Eyuel KokebDocument6 pagesTheory and Its Implication by Eyuel KokebEyuel KokebNo ratings yet
- Peel Synchro GuidelinesDocument20 pagesPeel Synchro GuidelinesSyed Imam100% (1)
- Nadia Karima Izzaty 1306369466 Dept. Teknik Sipil: Time (H) Rate (In/h) Direct Runoff (CFS) RM (In.)Document7 pagesNadia Karima Izzaty 1306369466 Dept. Teknik Sipil: Time (H) Rate (In/h) Direct Runoff (CFS) RM (In.)Nadia KarimaNo ratings yet
- Modules Music 6Document23 pagesModules Music 6Diether Mercado PaduaNo ratings yet
- Circle CPPDocument7 pagesCircle CPPAnugrah AgrawalNo ratings yet
- Domain Specific Languages in Practice: International Journal of Computer Applications April 2015Document5 pagesDomain Specific Languages in Practice: International Journal of Computer Applications April 2015Radian Nata KristiantoNo ratings yet
- Module 5n6 Answer Sheet Is Anatomy and Physiology (Abegail C. Relunia - Bsed 3c-Science)Document6 pagesModule 5n6 Answer Sheet Is Anatomy and Physiology (Abegail C. Relunia - Bsed 3c-Science)Abegail ReluniaNo ratings yet
- Nav2013 Enus Upgrade 03Document56 pagesNav2013 Enus Upgrade 03Wagz KagiriNo ratings yet
- Polycythaemia Vera Presenting As Massive Splenic Infarction and Liquefaction PDFDocument2 pagesPolycythaemia Vera Presenting As Massive Splenic Infarction and Liquefaction PDFtedmozbiNo ratings yet
- Semi-Active Fuzzy Control of A Wind-Excited Tall Building Using Multi-Objectivegenetic AlgorithmDocument16 pagesSemi-Active Fuzzy Control of A Wind-Excited Tall Building Using Multi-Objectivegenetic Algorithmalexkoch2No ratings yet
- A Creep Life Assessment Method For Boiler Pipes Using Small Punch Creep TestDocument6 pagesA Creep Life Assessment Method For Boiler Pipes Using Small Punch Creep TestAndrea CalderaNo ratings yet
- Transportation Engineering 1 Question Bank - Mumbai UniversityDocument10 pagesTransportation Engineering 1 Question Bank - Mumbai UniversityMegha Thomas0% (1)
- The Bass Player's Companion: A G Uide To S Ound, C Oncepts, and T Echnique For The E Lectric BassDocument55 pagesThe Bass Player's Companion: A G Uide To S Ound, C Oncepts, and T Echnique For The E Lectric BassBassman BronNo ratings yet
- AI - AI417DE01 Lab - Week 09-22102363Document8 pagesAI - AI417DE01 Lab - Week 09-22102363thinhbui1907No ratings yet
- Data-Driven Unit Tests: (Fact) and (Theory)Document6 pagesData-Driven Unit Tests: (Fact) and (Theory)Ronesh KumarNo ratings yet
- A Critical Review of Heat Transfer Through Helical Coils of Circular Cross Section PDFDocument9 pagesA Critical Review of Heat Transfer Through Helical Coils of Circular Cross Section PDFManuel CortesNo ratings yet
- Vinay Sharma - inDocument3 pagesVinay Sharma - inVinay SharmaNo ratings yet
- 2009 06 23 CAD CMX Technical Report 164Document99 pages2009 06 23 CAD CMX Technical Report 164César GuerreroNo ratings yet
- Datasheet EE800Document3 pagesDatasheet EE800AdyMichaelNo ratings yet