Professional Documents
Culture Documents
Calculating-ISO-7886-TOGC
Calculating-ISO-7886-TOGC
Uploaded by
ﺵﻓﻴﺮﺍ ﺭﺷﻲ ﺍﻧﺎCopyright:
Available Formats
You might also like
- Kristle Joy Dimayuga - Activity 1Document5 pagesKristle Joy Dimayuga - Activity 1Kristle Dimayuga100% (3)
- Solution - Fractional Flow Equation Application Class WorkDocument13 pagesSolution - Fractional Flow Equation Application Class WorkgebrilleNo ratings yet
- Tabel Dan Grafik SondirDocument10 pagesTabel Dan Grafik SondirVidio StoryNo ratings yet
- Mallas de Acero Inoxidable C-304 Y C-316: Mesh #Diametro Del Alambre (MM) Luz de La Malla (MM)Document2 pagesMallas de Acero Inoxidable C-304 Y C-316: Mesh #Diametro Del Alambre (MM) Luz de La Malla (MM)Diego Miguel Aliaga GomezNo ratings yet
- Ficha Mallas de Acero InoxidableDocument2 pagesFicha Mallas de Acero InoxidablejuanNo ratings yet
- UntitledDocument6 pagesUntitledRBV DESIGN & BUILD SERVICESNo ratings yet
- Espectro E030 2003 Peru - MiDocument9 pagesEspectro E030 2003 Peru - MiOscarQuirogaNo ratings yet
- Material A Material CDocument9 pagesMaterial A Material Calpin95No ratings yet
- Diametro Nominal (MM) (Pulg)Document2 pagesDiametro Nominal (MM) (Pulg)daniela caryNo ratings yet
- Perhitungan Nakayasu DAM ParitDocument16 pagesPerhitungan Nakayasu DAM ParitArnold SiregarNo ratings yet
- Laporan Kendali Fuzzy Logic Kelompok RianDocument11 pagesLaporan Kendali Fuzzy Logic Kelompok RianNina FloydNo ratings yet
- JL VP Tiempo Total Tiempo de CicloDocument22 pagesJL VP Tiempo Total Tiempo de CicloJohana Andrea Suarez RubianoNo ratings yet
- Lam 1 Data PrimerDocument4 pagesLam 1 Data PrimerUrshalina MasiaraNo ratings yet
- Movimeinto de Tierras Ramon CastillaDocument19 pagesMovimeinto de Tierras Ramon CastillaGiancarlos AguinagaNo ratings yet
- Lab PotenciometriaDocument15 pagesLab PotenciometrialarguedasNo ratings yet
- CRECIMIENTODocument3 pagesCRECIMIENTODeysi Roxana Suclupe GarciaNo ratings yet
- CRECIMIENTODocument3 pagesCRECIMIENTODeysi Roxana Suclupe GarciaNo ratings yet
- Calibracion Materiales InterpolacionDocument8 pagesCalibracion Materiales InterpolacionKatherine BautistaNo ratings yet
- Calculator TablaDocument8 pagesCalculator TablaFlorin MociranNo ratings yet
- Estacion: AYAVIRI Departamento: LIMA Provincia: YAUYOS Latitud:12°22'55.49'' Longitud:76°8'12.2''Document4 pagesEstacion: AYAVIRI Departamento: LIMA Provincia: YAUYOS Latitud:12°22'55.49'' Longitud:76°8'12.2''kennerNo ratings yet
- Volumen de Reporte: Progresiva Area de Corte M2 Area de Relleno M2 Vol. de Corte Vol. de RellenoDocument9 pagesVolumen de Reporte: Progresiva Area de Corte M2 Area de Relleno M2 Vol. de Corte Vol. de RellenoyuniorfredNo ratings yet
- Espectro E030 2003 Peru - MCDocument9 pagesEspectro E030 2003 Peru - MCOscarQuirogaNo ratings yet
- Disignation Des Ouvrages U Dimensions L L H/ep: #De Prix NPS RessultatsDocument3 pagesDisignation Des Ouvrages U Dimensions L L H/ep: #De Prix NPS RessultatsOussama BousalimNo ratings yet
- Levantamento Vigas SienaDocument97 pagesLevantamento Vigas SienaMurilo AssadNo ratings yet
- Reade Particle Size Conversion AnsiDocument1 pageReade Particle Size Conversion AnsijhscribdaccNo ratings yet
- Pemeriksaan Kekuatan Tanah Dengna Siondir: Grafik KonsolidasiDocument18 pagesPemeriksaan Kekuatan Tanah Dengna Siondir: Grafik KonsolidasiAdji PriwantaraNo ratings yet
- Graficas ConsolidacionDocument9 pagesGraficas ConsolidacionAngie SamudioNo ratings yet
- Measurement Sheet of Log No. 101Document5 pagesMeasurement Sheet of Log No. 101ejazNo ratings yet
- P 30 30 in R - in R (Rate) In/hrDocument14 pagesP 30 30 in R - in R (Rate) In/hrAbdullah AL QahtaniNo ratings yet
- MQDocument4 pagesMQJOMARY AGALEDNo ratings yet
- Buizen2018 Welded-SeamlessDocument8 pagesBuizen2018 Welded-SeamlesshtcdisirexemailNo ratings yet
- Simula Encargos 1.2Document2 pagesSimula Encargos 1.2Fabio.soares RhNo ratings yet
- Measurement MorayurDocument9 pagesMeasurement MorayurAssistant Engineer PWD Bridges Section, MannarkkadNo ratings yet
- Grade Tonne Report TODASLASCATEGORIASDocument11 pagesGrade Tonne Report TODASLASCATEGORIASAlfaro RoblesNo ratings yet
- Tata Cara Kerja Pembuatan Data Curah Hujan Data Curah Hujan HarianDocument1 pageTata Cara Kerja Pembuatan Data Curah Hujan Data Curah Hujan Harianfaisal rizqiNo ratings yet
- Waterflooding 3 y 4Document8 pagesWaterflooding 3 y 4Jorge Blanco ChoqueNo ratings yet
- Filter Media 15.1.2015Document17 pagesFilter Media 15.1.2015Prashant SinghNo ratings yet
- Kumulativo Kada CBR - Versaun TetunDocument45 pagesKumulativo Kada CBR - Versaun TetunmgiNo ratings yet
- The Engineering Toolbox: Compressed Air - Pressure Drop in PipelinesDocument6 pagesThe Engineering Toolbox: Compressed Air - Pressure Drop in PipelinesWael WaelNo ratings yet
- Laporan Sementara Job 7 - Kel 5Document4 pagesLaporan Sementara Job 7 - Kel 5Rakha MaulanaNo ratings yet
- Plantilla AlcantarilladoDocument2 pagesPlantilla Alcantarilladodaniel 93No ratings yet
- Washability Test Kel 1Document4 pagesWashability Test Kel 1siti maghfiraNo ratings yet
- Pressure DropDocument6 pagesPressure DropamrNo ratings yet
- Pressure DropDocument6 pagesPressure Dropdept.processnamoNo ratings yet
- Planilha para Simulação de Tratamento AcústicoDocument4 pagesPlanilha para Simulação de Tratamento AcústicoRodrigo FilhoNo ratings yet
- Simulación Reacción EDocument67 pagesSimulación Reacción EMilagros Morales VergarayNo ratings yet
- Wit MND NL STD Alg 20200101Document45 pagesWit MND NL STD Alg 20200101sharath_278No ratings yet
- Espectro de RespuestaDocument4 pagesEspectro de RespuestaCarlos RodriguezNo ratings yet
- 6058 Many2Document6 pages6058 Many2api-3699305No ratings yet
- EstadisticaDocument2 pagesEstadisticaJuan BonifazNo ratings yet
- Espectro De Diseño Sismico Gbds: Espectro Zona 3-Suelo S4 (1.5 Kg/cm2>σadm ≥ 0.5Kg/cm2)Document4 pagesEspectro De Diseño Sismico Gbds: Espectro Zona 3-Suelo S4 (1.5 Kg/cm2>σadm ≥ 0.5Kg/cm2)OscarQuirogaNo ratings yet
- Viga L (XX) - Pasto: 4% Vertical (CM)Document6 pagesViga L (XX) - Pasto: 4% Vertical (CM)Sebastian BernalNo ratings yet
- Nro Identificación Parcial 1 Parcial 2 Taller 1 Taller 2 Tarea 1 Tarea 2 Tarea 3 Tarea 4 TAREAS (2.0) E. PARCIALES (1.0) TALLERES (1.5)Document1 pageNro Identificación Parcial 1 Parcial 2 Taller 1 Taller 2 Tarea 1 Tarea 2 Tarea 3 Tarea 4 TAREAS (2.0) E. PARCIALES (1.0) TALLERES (1.5)Diego RoldanNo ratings yet
- PH V PH V: TitranDocument4 pagesPH V PH V: TitranStephen BagunaNo ratings yet
- Consum Cherestea Decembrie 2014 Tabel de Calcul Paleti Din Lemn PTR Tabla Palet Din LemnDocument8 pagesConsum Cherestea Decembrie 2014 Tabel de Calcul Paleti Din Lemn PTR Tabla Palet Din LemnGheorghita FanelNo ratings yet
- Sample Size EstimatorDocument8 pagesSample Size EstimatorRiyan MardiyantaNo ratings yet
- Copia de Flujo - JC LucasDocument37 pagesCopia de Flujo - JC LucasALEJANDRA GUTIERREZ SAAVEDRANo ratings yet
- Concentración de Equilibrio de Adsorbato Cantidad Específica de Equilibrio de AdsorbatoDocument5 pagesConcentración de Equilibrio de Adsorbato Cantidad Específica de Equilibrio de AdsorbatoDayNo ratings yet
- Intensidades Maximas IDFDocument12 pagesIntensidades Maximas IDFEricEspinozaAcostaNo ratings yet
- Llogaritja e KG Te LamarinesDocument17 pagesLlogaritja e KG Te LamarinesArmond KurtiNo ratings yet
- Math Practice Simplified: Decimals & Percents (Book H): Practicing the Concepts of Decimals and PercentagesFrom EverandMath Practice Simplified: Decimals & Percents (Book H): Practicing the Concepts of Decimals and PercentagesRating: 5 out of 5 stars5/5 (3)
- Vehicle ChecklistDocument1 pageVehicle ChecklistAbdus SamadNo ratings yet
- Optimization of Methanol ProductionDocument14 pagesOptimization of Methanol ProductionMuhammad AliNo ratings yet
- Portfolio in My Immersion Work 2.0Document41 pagesPortfolio in My Immersion Work 2.0floreson 16No ratings yet
- RESEARCHDocument70 pagesRESEARCHRizza Manabat PacheoNo ratings yet
- Komatsu Views 2014 No.31 (Ing)Document3 pagesKomatsu Views 2014 No.31 (Ing)Carlos Alfredo LauraNo ratings yet
- Mandate Hub User GuideDocument53 pagesMandate Hub User GuideMuruganand RamalingamNo ratings yet
- A Study On Problem and Prospects of Women Entrepreneur in Coimbatore and Tirupur QuestionnaireDocument6 pagesA Study On Problem and Prospects of Women Entrepreneur in Coimbatore and Tirupur QuestionnaireAsheek ShahNo ratings yet
- Thorn Outdoor CatalogueDocument46 pagesThorn Outdoor CatalogueEmeka Patrick OgbuNo ratings yet
- FCE Exam 3 ListeningDocument6 pagesFCE Exam 3 ListeningSaul MendozaNo ratings yet
- Ch. 9 Sampling Distributions and Confidence Intervals For ProportionsDocument22 pagesCh. 9 Sampling Distributions and Confidence Intervals For ProportionsThanh PhamNo ratings yet
- Einstein - Remarks On Bertrand RussellDocument15 pagesEinstein - Remarks On Bertrand RussellGabriel Rogé Such100% (1)
- Jawapan Topikal Kertas 1 Tingkatan 4Document16 pagesJawapan Topikal Kertas 1 Tingkatan 4qistinaunie07No ratings yet
- (Appendix 4A) Teacher Reflection Form For T I-III For RPMS SY 2021-2022Document10 pages(Appendix 4A) Teacher Reflection Form For T I-III For RPMS SY 2021-2022Quia Ferraren UmayaoNo ratings yet
- The Changing Politics of Foreign PolicyDocument3 pagesThe Changing Politics of Foreign PolicyLuís PatriotaNo ratings yet
- RESMETH Instructors ManualDocument32 pagesRESMETH Instructors ManualportucasbasNo ratings yet
- Valkokari TIMReview August2015Document8 pagesValkokari TIMReview August2015Héctor BallesterosNo ratings yet
- Thesis Statement About Business AdministrationDocument4 pagesThesis Statement About Business Administrationokxyghxff100% (2)
- Specification For Marker PostsDocument5 pagesSpecification For Marker PostsTai ChoonNo ratings yet
- MKT Marunda Center ProfileDocument47 pagesMKT Marunda Center ProfileMuhammad AbidinNo ratings yet
- Corrigendum 18.10.2011Document4 pagesCorrigendum 18.10.2011Ravi KalesNo ratings yet
- Lab Assignment 11313Document2 pagesLab Assignment 11313abhaybaranwalNo ratings yet
- CJR FILSAFAT Sem 1Document17 pagesCJR FILSAFAT Sem 1Jesica RaivitaNo ratings yet
- Self-Efficacy Mediated Spiritual Leadership On Citizenship Behavior Towards The Environment of Employees at Harapan Keluarga HospitalDocument7 pagesSelf-Efficacy Mediated Spiritual Leadership On Citizenship Behavior Towards The Environment of Employees at Harapan Keluarga HospitalInternational Journal of Innovative Science and Research TechnologyNo ratings yet
- A. B. 0.99 M /KG 0.79 M /KG 0.89 M /KG 0.69m /KGDocument143 pagesA. B. 0.99 M /KG 0.79 M /KG 0.89 M /KG 0.69m /KGRyan Togonon100% (1)
- Internal Combustion Engines - R. K. RajputDocument352 pagesInternal Combustion Engines - R. K. RajputmeetbalakumarNo ratings yet
- Java Atm PtojectDocument10 pagesJava Atm PtojectKrinal kathiriyaNo ratings yet
- 5S Implementation Manual Part 2Document63 pages5S Implementation Manual Part 2jgprasadNo ratings yet
- General Chemistry 1 Module 1 Answer Sheet (Page 1 of 3) : Examples of Physical Properties IncludeDocument6 pagesGeneral Chemistry 1 Module 1 Answer Sheet (Page 1 of 3) : Examples of Physical Properties IncludeJeremiah Se-engNo ratings yet
- Literature Review of The Job SatisfactionDocument3 pagesLiterature Review of The Job SatisfactionsuryaNo ratings yet
Calculating-ISO-7886-TOGC
Calculating-ISO-7886-TOGC
Uploaded by
ﺵﻓﻴﺮﺍ ﺭﺷﻲ ﺍﻧﺎCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Calculating-ISO-7886-TOGC
Calculating-ISO-7886-TOGC
Uploaded by
ﺵﻓﻴﺮﺍ ﺭﺷﻲ ﺍﻧﺎCopyright:
Available Formats
Syringe accuracy (TOGC) estimates for ISO 7886-1
P Phillips, Director, Surgical Materials Testing Laboratory
August 10, 2016 - Revision: 1.4
Introduction
This paper shows the method and for using table 1 in ISO 7886-1 to calculate the Tolerance on graduated
capacity (TOGC) for syringes, and displays calculated values for different combinations of syringe size and
expelled volume. It is available online as a pdf.
For example, if using a 1ml syringe to deliver a particular volume of expelled liquid, ISO 7886-1 states that
the tolerance on the graduated capacity is
• ± (1.5% syringe vol + 2% of the expelled volume) for volumes < 0.5ml, and
• ± 5% of the expelled volume for volumes ≥ 0.5ml.
Similar but different tolerances are used depending on the syringe size for syringes up to 50ml of nominal
capacity.
Worked examples
As a worked example, lets look at delivering 4ml from a 10ml syringe.
• 4ml is less than half the nominal volume of a 10ml syringe so we use the first column (‘less than half
nominal capacity’)
• (1.5% × 10) + (1% × 4) = 0.15 + 0.04 = 0.19ml
• As a percentage of the expelled volume (4ml), 4.75%
If instead we look at delivering an 8ml dose from a 10ml syringe:
• 8ml is greater than half the nominal volume of a 10ml syringe so we use the first column (‘equal to or
greater than half nominal capacity’)
• 4% × 8 = 0.32ml
• As a percentage of the expelled volume, 4%
Tolerance Calculation Tables
The following tables show how the error varies as the delivery volume changes for different size syringes.
The columns are as follows:
• Syringe Size - Nominal capacity of syringe in ml
• Expelled Vol - the volume intended to be delivered from the syringe in ml (the volume called ‘expelled
volume’ in ISO 7886, although this may more accurately be called the intended volume of delivery)
• Tolerance ± (ml) - Tolerance on graduated capacity (TOGC) calculated in ml as per Table 1, EN
ISO 7886-1:1997
1
Syringe accuracy estimates for ISO 7886-1
• Tolerance ± (%) - Tolerance on graduated capacity calculated as a % of the Expelled Vol as per
Table 1, EN ISO 7886-1:1997
The tables have been generated with the ‘R’1 language using ‘R Markdown’2 . This document and the original
Rmarkdown code are available from the SMTL website3 .
1 https://en.wikipedia.org/wiki/R_(programming_language)
2 http://rmarkdown.rstudio.com/
3 http://smtl.co.uk/pete-phillips/224-togc-from-iso-7886-1.html
August 10, 2016 2 Revision: 1.4
Syringe accuracy estimates for ISO 7886-1
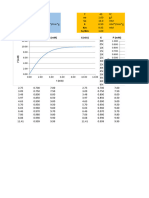
Syringe Size Expelled Vol Tolerance ± (ml) Tolerance ± (%)
1 0.10 0.017 17.00
1 0.20 0.019 9.50
1 0.30 0.021 7.00
1 0.40 0.023 5.75
1 0.50 0.025 5.00
1 0.60 0.030 5.00
1 0.70 0.035 5.00
1 0.80 0.040 5.00
1 0.90 0.045 5.00
1 1.00 0.050 5.00
Table 1: Syringe Accuracy Vol - Syringe Size = 1 ml
Syringe Size Expelled Vol Tolerance ± (ml) Tolerance ± (%)
2 0.10 0.032 32.00
2 0.20 0.034 17.00
2 0.30 0.036 12.00
2 0.40 0.038 9.50
2 0.50 0.040 8.00
2 0.60 0.042 7.00
2 0.70 0.044 6.29
2 0.80 0.046 5.75
2 0.90 0.048 5.33
2 1.00 0.050 5.00
2 1.10 0.055 5.00
2 1.20 0.060 5.00
2 1.30 0.065 5.00
2 1.40 0.070 5.00
2 1.50 0.075 5.00
2 1.60 0.080 5.00
2 1.70 0.085 5.00
2 1.80 0.090 5.00
2 1.90 0.095 5.00
2 2.00 0.100 5.00
Table 2: Syringe Accuracy Vol - Syringe Size = 2 ml
August 10, 2016 3 Revision: 1.4
Syringe accuracy estimates for ISO 7886-1
Syringe Size Expelled Vol Tolerance ± (ml) Tolerance ± (%)
3 0.10 0.047 47.00
3 0.20 0.049 24.50
3 0.30 0.051 17.00
3 0.40 0.053 13.25
3 0.50 0.055 11.00
3 0.60 0.057 9.50
3 0.70 0.059 8.43
3 0.80 0.061 7.62
3 0.90 0.063 7.00
3 1.00 0.065 6.50
3 1.10 0.067 6.09
3 1.20 0.069 5.75
3 1.30 0.071 5.46
3 1.40 0.073 5.21
3 1.50 0.075 5.00
3 1.60 0.080 5.00
3 1.70 0.085 5.00
3 1.80 0.090 5.00
3 1.90 0.095 5.00
3 2.00 0.100 5.00
3 2.10 0.105 5.00
3 2.20 0.110 5.00
3 2.30 0.115 5.00
3 2.40 0.120 5.00
3 2.50 0.125 5.00
3 2.60 0.130 5.00
3 2.70 0.135 5.00
3 2.80 0.140 5.00
3 2.90 0.145 5.00
3 3.00 0.150 5.00
Table 3: Syringe Accuracy Vol - Syringe Size = 3 ml
August 10, 2016 4 Revision: 1.4
Syringe accuracy estimates for ISO 7886-1
Syringe Size Expelled Vol Tolerance ± (ml) Tolerance ± (%)
5 0.10 0.076 76.00
5 0.20 0.077 38.50
5 0.30 0.078 26.00
5 0.40 0.079 19.75
5 0.50 0.080 16.00
5 0.60 0.081 13.50
5 0.70 0.082 11.71
5 0.80 0.083 10.37
5 0.90 0.084 9.33
5 1.00 0.085 8.50
5 1.10 0.086 7.82
5 1.20 0.087 7.25
5 1.30 0.088 6.77
5 1.40 0.089 6.36
5 1.50 0.090 6.00
5 1.60 0.091 5.69
5 1.70 0.092 5.41
5 1.80 0.093 5.17
5 1.90 0.094 4.95
5 2.00 0.095 4.75
5 2.10 0.096 4.57
5 2.20 0.097 4.41
5 2.30 0.098 4.26
5 2.40 0.099 4.12
5 2.50 0.100 4.00
5 2.60 0.104 4.00
5 2.70 0.108 4.00
5 2.80 0.112 4.00
5 2.90 0.116 4.00
5 3.00 0.120 4.00
5 3.50 0.140 4.00
5 4.00 0.160 4.00
5 4.50 0.180 4.00
5 5.00 0.200 4.00
Table 4: Syringe Accuracy Vol - Syringe Size = 5 ml
August 10, 2016 5 Revision: 1.4
Syringe accuracy estimates for ISO 7886-1
Syringe Size Expelled Vol Tolerance ± (ml) Tolerance ± (%)
6 0.10 0.091 91.00
6 0.20 0.092 46.00
6 0.30 0.093 31.00
6 0.40 0.094 23.50
6 0.50 0.095 19.00
6 0.60 0.096 16.00
6 0.70 0.097 13.86
6 0.80 0.098 12.25
6 0.90 0.099 11.00
6 1.00 0.100 10.00
6 1.10 0.101 9.18
6 1.20 0.102 8.50
6 1.30 0.103 7.92
6 1.40 0.104 7.43
6 1.50 0.105 7.00
6 1.60 0.106 6.62
6 1.70 0.107 6.29
6 1.80 0.108 6.00
6 1.90 0.109 5.74
6 2.00 0.110 5.50
6 2.10 0.111 5.29
6 2.20 0.112 5.09
6 2.30 0.113 4.91
6 2.40 0.114 4.75
6 2.50 0.115 4.60
6 2.60 0.116 4.46
6 2.70 0.117 4.33
6 2.80 0.118 4.21
6 2.90 0.119 4.10
6 3.00 0.120 4.00
6 2.50 0.115 4.60
6 3.00 0.120 4.00
6 3.50 0.140 4.00
6 4.00 0.160 4.00
6 4.50 0.180 4.00
6 5.00 0.200 4.00
6 5.50 0.220 4.00
6 6.00 0.240 4.00
Table 5: Syringe Accuracy Vol - Syringe Size = 6 ml
August 10, 2016 6 Revision: 1.4
Syringe accuracy estimates for ISO 7886-1
Syringe Size Expelled Vol Tolerance ± (ml) Tolerance ± (%)
10 0.10 0.151 151.00
10 0.20 0.152 76.00
10 0.30 0.153 51.00
10 0.40 0.154 38.50
10 0.50 0.155 31.00
10 0.60 0.156 26.00
10 0.70 0.157 22.43
10 0.80 0.158 19.75
10 0.90 0.159 17.67
10 1.00 0.160 16.00
10 1.50 0.165 11.00
10 2.00 0.170 8.50
10 2.50 0.175 7.00
10 3.00 0.180 6.00
10 3.50 0.185 5.29
10 4.00 0.190 4.75
10 4.50 0.195 4.33
10 5.00 0.200 4.00
10 5.50 0.220 4.00
10 6.00 0.240 4.00
10 6.50 0.260 4.00
10 7.00 0.280 4.00
10 7.50 0.300 4.00
10 8.00 0.320 4.00
10 8.50 0.340 4.00
10 9.00 0.360 4.00
10 9.50 0.380 4.00
10 10.00 0.400 4.00
Table 6: Syringe Accuracy Vol - Syringe Size = 10 ml
August 10, 2016 7 Revision: 1.4
Syringe accuracy estimates for ISO 7886-1
Syringe Size Expelled Vol Tolerance ± (ml) Tolerance ± (%)
20 0.10 0.301 301.00
20 0.20 0.302 151.00
20 0.30 0.303 101.00
20 0.40 0.304 76.00
20 0.50 0.305 61.00
20 0.60 0.306 51.00
20 0.70 0.307 43.86
20 0.80 0.308 38.50
20 0.90 0.309 34.33
20 1.00 0.310 31.00
20 1.50 0.315 21.00
20 2.00 0.320 16.00
20 2.50 0.325 13.00
20 3.00 0.330 11.00
20 3.50 0.335 9.57
20 4.00 0.340 8.50
20 4.50 0.345 7.67
20 5.00 0.350 7.00
20 5.50 0.355 6.45
20 6.00 0.360 6.00
20 6.50 0.365 5.62
20 7.00 0.370 5.29
20 7.50 0.375 5.00
20 8.00 0.380 4.75
20 8.50 0.385 4.53
20 9.00 0.390 4.33
20 9.50 0.395 4.16
20 10.00 0.400 4.00
20 10.50 0.420 4.00
20 11.00 0.440 4.00
20 11.50 0.460 4.00
20 12.00 0.480 4.00
20 12.50 0.500 4.00
20 13.00 0.520 4.00
20 13.50 0.540 4.00
20 14.00 0.560 4.00
20 14.50 0.580 4.00
20 15.00 0.600 4.00
20 15.50 0.620 4.00
20 16.00 0.640 4.00
20 16.50 0.660 4.00
20 17.00 0.680 4.00
20 17.50 0.700 4.00
20 18.00 0.720 4.00
20 18.50 0.740 4.00
20 19.00 0.760 4.00
20 19.50 0.780 4.00
20 20.00 0.800 4.00
Table 7: Syringe Accuracy Vol - Syringe Size = 20 ml
August 10, 2016 8 Revision: 1.4
Syringe accuracy estimates for ISO 7886-1
APPENDIX A
SMTL is a UK NHS Medical Device Testing Laboratory, funded centrally to provide testing and medical
device technical services to the Welsh NHS. SMTL also provides medical device testing and technical services
on a commercial basis to the global medical device industry. SMTL are accredited to ISO 17025 for testing
medical devices.
Surgical Materials Testing Laboratory,
Princess of Wales Hospital,
Coity Road,
Bridgend,
CF31 1RQ
Tel: 01656-752820
Web: http://www.smtl.co.uk/ & http://www.medidex.com/
August 10, 2016 9 Revision: 1.4
You might also like
- Kristle Joy Dimayuga - Activity 1Document5 pagesKristle Joy Dimayuga - Activity 1Kristle Dimayuga100% (3)
- Solution - Fractional Flow Equation Application Class WorkDocument13 pagesSolution - Fractional Flow Equation Application Class WorkgebrilleNo ratings yet
- Tabel Dan Grafik SondirDocument10 pagesTabel Dan Grafik SondirVidio StoryNo ratings yet
- Mallas de Acero Inoxidable C-304 Y C-316: Mesh #Diametro Del Alambre (MM) Luz de La Malla (MM)Document2 pagesMallas de Acero Inoxidable C-304 Y C-316: Mesh #Diametro Del Alambre (MM) Luz de La Malla (MM)Diego Miguel Aliaga GomezNo ratings yet
- Ficha Mallas de Acero InoxidableDocument2 pagesFicha Mallas de Acero InoxidablejuanNo ratings yet
- UntitledDocument6 pagesUntitledRBV DESIGN & BUILD SERVICESNo ratings yet
- Espectro E030 2003 Peru - MiDocument9 pagesEspectro E030 2003 Peru - MiOscarQuirogaNo ratings yet
- Material A Material CDocument9 pagesMaterial A Material Calpin95No ratings yet
- Diametro Nominal (MM) (Pulg)Document2 pagesDiametro Nominal (MM) (Pulg)daniela caryNo ratings yet
- Perhitungan Nakayasu DAM ParitDocument16 pagesPerhitungan Nakayasu DAM ParitArnold SiregarNo ratings yet
- Laporan Kendali Fuzzy Logic Kelompok RianDocument11 pagesLaporan Kendali Fuzzy Logic Kelompok RianNina FloydNo ratings yet
- JL VP Tiempo Total Tiempo de CicloDocument22 pagesJL VP Tiempo Total Tiempo de CicloJohana Andrea Suarez RubianoNo ratings yet
- Lam 1 Data PrimerDocument4 pagesLam 1 Data PrimerUrshalina MasiaraNo ratings yet
- Movimeinto de Tierras Ramon CastillaDocument19 pagesMovimeinto de Tierras Ramon CastillaGiancarlos AguinagaNo ratings yet
- Lab PotenciometriaDocument15 pagesLab PotenciometrialarguedasNo ratings yet
- CRECIMIENTODocument3 pagesCRECIMIENTODeysi Roxana Suclupe GarciaNo ratings yet
- CRECIMIENTODocument3 pagesCRECIMIENTODeysi Roxana Suclupe GarciaNo ratings yet
- Calibracion Materiales InterpolacionDocument8 pagesCalibracion Materiales InterpolacionKatherine BautistaNo ratings yet
- Calculator TablaDocument8 pagesCalculator TablaFlorin MociranNo ratings yet
- Estacion: AYAVIRI Departamento: LIMA Provincia: YAUYOS Latitud:12°22'55.49'' Longitud:76°8'12.2''Document4 pagesEstacion: AYAVIRI Departamento: LIMA Provincia: YAUYOS Latitud:12°22'55.49'' Longitud:76°8'12.2''kennerNo ratings yet
- Volumen de Reporte: Progresiva Area de Corte M2 Area de Relleno M2 Vol. de Corte Vol. de RellenoDocument9 pagesVolumen de Reporte: Progresiva Area de Corte M2 Area de Relleno M2 Vol. de Corte Vol. de RellenoyuniorfredNo ratings yet
- Espectro E030 2003 Peru - MCDocument9 pagesEspectro E030 2003 Peru - MCOscarQuirogaNo ratings yet
- Disignation Des Ouvrages U Dimensions L L H/ep: #De Prix NPS RessultatsDocument3 pagesDisignation Des Ouvrages U Dimensions L L H/ep: #De Prix NPS RessultatsOussama BousalimNo ratings yet
- Levantamento Vigas SienaDocument97 pagesLevantamento Vigas SienaMurilo AssadNo ratings yet
- Reade Particle Size Conversion AnsiDocument1 pageReade Particle Size Conversion AnsijhscribdaccNo ratings yet
- Pemeriksaan Kekuatan Tanah Dengna Siondir: Grafik KonsolidasiDocument18 pagesPemeriksaan Kekuatan Tanah Dengna Siondir: Grafik KonsolidasiAdji PriwantaraNo ratings yet
- Graficas ConsolidacionDocument9 pagesGraficas ConsolidacionAngie SamudioNo ratings yet
- Measurement Sheet of Log No. 101Document5 pagesMeasurement Sheet of Log No. 101ejazNo ratings yet
- P 30 30 in R - in R (Rate) In/hrDocument14 pagesP 30 30 in R - in R (Rate) In/hrAbdullah AL QahtaniNo ratings yet
- MQDocument4 pagesMQJOMARY AGALEDNo ratings yet
- Buizen2018 Welded-SeamlessDocument8 pagesBuizen2018 Welded-SeamlesshtcdisirexemailNo ratings yet
- Simula Encargos 1.2Document2 pagesSimula Encargos 1.2Fabio.soares RhNo ratings yet
- Measurement MorayurDocument9 pagesMeasurement MorayurAssistant Engineer PWD Bridges Section, MannarkkadNo ratings yet
- Grade Tonne Report TODASLASCATEGORIASDocument11 pagesGrade Tonne Report TODASLASCATEGORIASAlfaro RoblesNo ratings yet
- Tata Cara Kerja Pembuatan Data Curah Hujan Data Curah Hujan HarianDocument1 pageTata Cara Kerja Pembuatan Data Curah Hujan Data Curah Hujan Harianfaisal rizqiNo ratings yet
- Waterflooding 3 y 4Document8 pagesWaterflooding 3 y 4Jorge Blanco ChoqueNo ratings yet
- Filter Media 15.1.2015Document17 pagesFilter Media 15.1.2015Prashant SinghNo ratings yet
- Kumulativo Kada CBR - Versaun TetunDocument45 pagesKumulativo Kada CBR - Versaun TetunmgiNo ratings yet
- The Engineering Toolbox: Compressed Air - Pressure Drop in PipelinesDocument6 pagesThe Engineering Toolbox: Compressed Air - Pressure Drop in PipelinesWael WaelNo ratings yet
- Laporan Sementara Job 7 - Kel 5Document4 pagesLaporan Sementara Job 7 - Kel 5Rakha MaulanaNo ratings yet
- Plantilla AlcantarilladoDocument2 pagesPlantilla Alcantarilladodaniel 93No ratings yet
- Washability Test Kel 1Document4 pagesWashability Test Kel 1siti maghfiraNo ratings yet
- Pressure DropDocument6 pagesPressure DropamrNo ratings yet
- Pressure DropDocument6 pagesPressure Dropdept.processnamoNo ratings yet
- Planilha para Simulação de Tratamento AcústicoDocument4 pagesPlanilha para Simulação de Tratamento AcústicoRodrigo FilhoNo ratings yet
- Simulación Reacción EDocument67 pagesSimulación Reacción EMilagros Morales VergarayNo ratings yet
- Wit MND NL STD Alg 20200101Document45 pagesWit MND NL STD Alg 20200101sharath_278No ratings yet
- Espectro de RespuestaDocument4 pagesEspectro de RespuestaCarlos RodriguezNo ratings yet
- 6058 Many2Document6 pages6058 Many2api-3699305No ratings yet
- EstadisticaDocument2 pagesEstadisticaJuan BonifazNo ratings yet
- Espectro De Diseño Sismico Gbds: Espectro Zona 3-Suelo S4 (1.5 Kg/cm2>σadm ≥ 0.5Kg/cm2)Document4 pagesEspectro De Diseño Sismico Gbds: Espectro Zona 3-Suelo S4 (1.5 Kg/cm2>σadm ≥ 0.5Kg/cm2)OscarQuirogaNo ratings yet
- Viga L (XX) - Pasto: 4% Vertical (CM)Document6 pagesViga L (XX) - Pasto: 4% Vertical (CM)Sebastian BernalNo ratings yet
- Nro Identificación Parcial 1 Parcial 2 Taller 1 Taller 2 Tarea 1 Tarea 2 Tarea 3 Tarea 4 TAREAS (2.0) E. PARCIALES (1.0) TALLERES (1.5)Document1 pageNro Identificación Parcial 1 Parcial 2 Taller 1 Taller 2 Tarea 1 Tarea 2 Tarea 3 Tarea 4 TAREAS (2.0) E. PARCIALES (1.0) TALLERES (1.5)Diego RoldanNo ratings yet
- PH V PH V: TitranDocument4 pagesPH V PH V: TitranStephen BagunaNo ratings yet
- Consum Cherestea Decembrie 2014 Tabel de Calcul Paleti Din Lemn PTR Tabla Palet Din LemnDocument8 pagesConsum Cherestea Decembrie 2014 Tabel de Calcul Paleti Din Lemn PTR Tabla Palet Din LemnGheorghita FanelNo ratings yet
- Sample Size EstimatorDocument8 pagesSample Size EstimatorRiyan MardiyantaNo ratings yet
- Copia de Flujo - JC LucasDocument37 pagesCopia de Flujo - JC LucasALEJANDRA GUTIERREZ SAAVEDRANo ratings yet
- Concentración de Equilibrio de Adsorbato Cantidad Específica de Equilibrio de AdsorbatoDocument5 pagesConcentración de Equilibrio de Adsorbato Cantidad Específica de Equilibrio de AdsorbatoDayNo ratings yet
- Intensidades Maximas IDFDocument12 pagesIntensidades Maximas IDFEricEspinozaAcostaNo ratings yet
- Llogaritja e KG Te LamarinesDocument17 pagesLlogaritja e KG Te LamarinesArmond KurtiNo ratings yet
- Math Practice Simplified: Decimals & Percents (Book H): Practicing the Concepts of Decimals and PercentagesFrom EverandMath Practice Simplified: Decimals & Percents (Book H): Practicing the Concepts of Decimals and PercentagesRating: 5 out of 5 stars5/5 (3)
- Vehicle ChecklistDocument1 pageVehicle ChecklistAbdus SamadNo ratings yet
- Optimization of Methanol ProductionDocument14 pagesOptimization of Methanol ProductionMuhammad AliNo ratings yet
- Portfolio in My Immersion Work 2.0Document41 pagesPortfolio in My Immersion Work 2.0floreson 16No ratings yet
- RESEARCHDocument70 pagesRESEARCHRizza Manabat PacheoNo ratings yet
- Komatsu Views 2014 No.31 (Ing)Document3 pagesKomatsu Views 2014 No.31 (Ing)Carlos Alfredo LauraNo ratings yet
- Mandate Hub User GuideDocument53 pagesMandate Hub User GuideMuruganand RamalingamNo ratings yet
- A Study On Problem and Prospects of Women Entrepreneur in Coimbatore and Tirupur QuestionnaireDocument6 pagesA Study On Problem and Prospects of Women Entrepreneur in Coimbatore and Tirupur QuestionnaireAsheek ShahNo ratings yet
- Thorn Outdoor CatalogueDocument46 pagesThorn Outdoor CatalogueEmeka Patrick OgbuNo ratings yet
- FCE Exam 3 ListeningDocument6 pagesFCE Exam 3 ListeningSaul MendozaNo ratings yet
- Ch. 9 Sampling Distributions and Confidence Intervals For ProportionsDocument22 pagesCh. 9 Sampling Distributions and Confidence Intervals For ProportionsThanh PhamNo ratings yet
- Einstein - Remarks On Bertrand RussellDocument15 pagesEinstein - Remarks On Bertrand RussellGabriel Rogé Such100% (1)
- Jawapan Topikal Kertas 1 Tingkatan 4Document16 pagesJawapan Topikal Kertas 1 Tingkatan 4qistinaunie07No ratings yet
- (Appendix 4A) Teacher Reflection Form For T I-III For RPMS SY 2021-2022Document10 pages(Appendix 4A) Teacher Reflection Form For T I-III For RPMS SY 2021-2022Quia Ferraren UmayaoNo ratings yet
- The Changing Politics of Foreign PolicyDocument3 pagesThe Changing Politics of Foreign PolicyLuís PatriotaNo ratings yet
- RESMETH Instructors ManualDocument32 pagesRESMETH Instructors ManualportucasbasNo ratings yet
- Valkokari TIMReview August2015Document8 pagesValkokari TIMReview August2015Héctor BallesterosNo ratings yet
- Thesis Statement About Business AdministrationDocument4 pagesThesis Statement About Business Administrationokxyghxff100% (2)
- Specification For Marker PostsDocument5 pagesSpecification For Marker PostsTai ChoonNo ratings yet
- MKT Marunda Center ProfileDocument47 pagesMKT Marunda Center ProfileMuhammad AbidinNo ratings yet
- Corrigendum 18.10.2011Document4 pagesCorrigendum 18.10.2011Ravi KalesNo ratings yet
- Lab Assignment 11313Document2 pagesLab Assignment 11313abhaybaranwalNo ratings yet
- CJR FILSAFAT Sem 1Document17 pagesCJR FILSAFAT Sem 1Jesica RaivitaNo ratings yet
- Self-Efficacy Mediated Spiritual Leadership On Citizenship Behavior Towards The Environment of Employees at Harapan Keluarga HospitalDocument7 pagesSelf-Efficacy Mediated Spiritual Leadership On Citizenship Behavior Towards The Environment of Employees at Harapan Keluarga HospitalInternational Journal of Innovative Science and Research TechnologyNo ratings yet
- A. B. 0.99 M /KG 0.79 M /KG 0.89 M /KG 0.69m /KGDocument143 pagesA. B. 0.99 M /KG 0.79 M /KG 0.89 M /KG 0.69m /KGRyan Togonon100% (1)
- Internal Combustion Engines - R. K. RajputDocument352 pagesInternal Combustion Engines - R. K. RajputmeetbalakumarNo ratings yet
- Java Atm PtojectDocument10 pagesJava Atm PtojectKrinal kathiriyaNo ratings yet
- 5S Implementation Manual Part 2Document63 pages5S Implementation Manual Part 2jgprasadNo ratings yet
- General Chemistry 1 Module 1 Answer Sheet (Page 1 of 3) : Examples of Physical Properties IncludeDocument6 pagesGeneral Chemistry 1 Module 1 Answer Sheet (Page 1 of 3) : Examples of Physical Properties IncludeJeremiah Se-engNo ratings yet
- Literature Review of The Job SatisfactionDocument3 pagesLiterature Review of The Job SatisfactionsuryaNo ratings yet