Professional Documents
Culture Documents
Flavonoid Bio Synthesis in Bilberry
Flavonoid Bio Synthesis in Bilberry
Uploaded by
Maris KurniawatiCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Flavonoid Bio Synthesis in Bilberry
Flavonoid Bio Synthesis in Bilberry
Uploaded by
Maris KurniawatiCopyright:
Available Formats
Prev
Flavonoid biosynthesis in bilberry (Vaccinium myrtillus L.) Chapter 1. Introduction Next
1.3. Flavonoid biosynthetic pathway
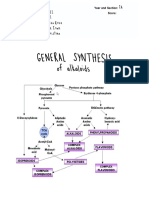
Flavonoids are synthesized via the phenylpropanoid pathway. Phenylalanine ammonia lyase (PAL) catalyzes the conversion of phenylalanine to cinnamate. PAL also shows activity with converting tyrosine to p-coumarate, albeit to a lower efficiency. The cinnamate 4-hydroxylase (C4H) catalyzes the synthesis of p-hydroxycinnamate from cinnamate and 4-coumarate:CoA ligase (4CL) converts p-coumarate to its coenzyme-A ester, activating it for reaction with malonyl CoA. The flavonoid biosynthetic pathway starts with the condensation of one molecule of 4-coumaroyl-CoA and three molecules of malonyl-CoA, yielding naringenin chalcone. This reaction is carried out by the enzyme chalcone synthase (CHS). Chalcone is isomerised to a flavanone by the enzyme chalcone flavanone isomerase (CHI). From these central intermediates, the pathway diverges into several side branches, each resulting in a different class of flavonoids. Flavanone 3-hydroxylase (F3H) catalyzes the stereospecific 3hydroxylation of (2S)-flavanones to dihydroflavonols. For the biosynthesis of anthocyanins, dihydroflavonol reductase (DFR) catalyzes the reduction of dihydroflavonols to flavan-3,4diols (leucoanthocyanins), which are converted to anthocyanidins by anthocyanidin synthase (ANS). The formation of glucosides is catalyzed by UDP glucose-flavonoid 3-o-glucosyl transferase (UFGT), which stabilize the anthocyanidins by 3-O-glucosylation (Harborne 1994, Bohm 1998). The overview of the flavonoid pathway is presented in Fig 1B. There is evidence that the enzymes involved in flavonoid metabolism might be acting as membraneassociated multienzyme complexes, which has implications on the overall efficiency, specificity, and regulation of the pathway (Stafford 1991, Winkel-Shirley 1999, 2001). Studies of the flavonoid pathway range from classical genetic analysis of flower color inheritance patterns by Mendel, through the establishment of their chemical structures, to efforts to understand the factors involved in their biochemical synthesis (Bohm 1998). Basic knowledge of the flavonoid biosynthesis was gained from experimental studies using radiolabeled precursors at the end of 1950s. The development of more sophisticated methods in analytical chemistry and enzymology, and later in gene technology, has produced a vast number of studies and detailed information of the flavonoid biosynthesis in several plant species. The flavonoid biosynthetic pathway has been comprehensively reviewed (e.g. by Dooner & Robbins 1991, Koes et al. 1994, Holton & Cornish 1995, Mol et al. 1998, Weisshaar & Jenkins 1998, Winkel-Shirley 2001). The first gene isolated from the flavonoid biosynthetic pathway was a CHS gene from parsley (Petroselinum hortense) (Kreuzaler et al. 1983). Transcriptional control of the structural genes of the flavonoid biosynthetic pathway has been most intensively studied in relation to the biosynthesis of anthocyanins. Groundbreaking research concerning the expression of the structural and regulatory genes of the flavonoid pathway has been done with maize (Zea mays) (Goff et al. 1990, Taylor et al. 1990, Tonelli et al. 1991), arabidopsis (Arabidopsis thaliana) (Shirley et al. 1992) and with ornamental plants like snapdragon (Antirrhinum majus) (Martin et al.1991), petunia (van der Krol et al. 1988) and gerbera (Elomaa et al. 1993, Helariutta et al. 1993, 1995). Naturally occurring flavonoid mutants and variants, or genetically transformed mutant plants have been important tools in several investigations
clarifying the functions of the flavonoid pathway genes (Shirley et al. 1995, Tanaka et al. 1998). The expression of flavonoid pathway genes in fruit tissues has been studied on grape (Vitis vinifera) (Boss et al. 1996, Kobayashi et al. 2001), citrus (Citrus unshiu Marc.) (Moriguchi et al. 2001), and strawberry plants (Fragaria spp.) (Manning 1998, Aharoni et al. 2001). The scarcity of studies in this area may be due to a difficulty caused by the special features of the fruit tissues, e.g. the richness of different secondary metabolites and RNases, which may hinder the easy application of the molecular biological research methods.
Figure 1. A) The structures of selected flavonoid classes. B) A schematic presentation of the flavonoid biosynthetic pathway. Enzyme abbreviations: PAL, phenylalanine ammonia-lyase; C4H, cinnamate 4-hydroxylase; 4CL, 4-coumaroyl:CoA ligase; CHS,
chalcone synthase; CHI, chalcone isomerase; F3H, flavanone 3-hydroxylase; DFR, dihydroflavonol 4-reductase; ANS, anthocyanidin synthase; UFGT, UDP glucoseflavonoid 3-o-glucosyl transferase.
Prev Flavonoids
Home Up
Next Flavonoids in fruits
Homepage of this publication Library Units | Collections | Databases | Library News | Library Services | Electronic Collection | Links elsewhere | Alphabetical Index 2003 Oulu University Library Loading next page... - Page 2 -
You might also like
- Full Ebook of Pizzo Poplack S Pediatric Oncology Eighth Edition Susan M Blaney Lee J Helman Peter C Adamson Online PDF All ChapterDocument69 pagesFull Ebook of Pizzo Poplack S Pediatric Oncology Eighth Edition Susan M Blaney Lee J Helman Peter C Adamson Online PDF All Chapterffancinetefn100% (5)
- NSTM Chapter 074 Volume 2 - Non-Destructive Testing of Metals - Qualification and Certification Requirements For Naval Personnel Nuclear)Document28 pagesNSTM Chapter 074 Volume 2 - Non-Destructive Testing of Metals - Qualification and Certification Requirements For Naval Personnel Nuclear)sailboy21100% (1)
- Lab 2 - Report - FO1Document23 pagesLab 2 - Report - FO1V TH100% (1)
- LoreSmyth's Ultimate Guide To Remarkable Inns and Their DrinksDocument89 pagesLoreSmyth's Ultimate Guide To Remarkable Inns and Their DrinksTom Kakanowski100% (2)
- Fenolne Kiseline I DerivatiDocument47 pagesFenolne Kiseline I DerivatiCica Zlica100% (3)
- Genetics and Biochemistry of Ant Hocyanin Biosynthesis: Timothy A. Holton' and Edwina C. CornishDocument13 pagesGenetics and Biochemistry of Ant Hocyanin Biosynthesis: Timothy A. Holton' and Edwina C. CornishPatrisia HallaNo ratings yet
- 09 Can A BisDocument8 pages09 Can A BispentaanNo ratings yet
- Distinct Metabolic Pathways Drive Monoterpenoid Biosynthesis in A Natural Population of PelargoniumDocument14 pagesDistinct Metabolic Pathways Drive Monoterpenoid Biosynthesis in A Natural Population of PelargoniumЕгяна КазимомаNo ratings yet
- Dramatic Increase in Content of Diverse FlavonoidsDocument20 pagesDramatic Increase in Content of Diverse FlavonoidsYou Jin LimNo ratings yet
- Weijn 2013Document12 pagesWeijn 2013Oscar OrtizNo ratings yet
- Via FenilpropanoideDocument20 pagesVia FenilpropanoideCLaudia Julieta Robles PLanellsNo ratings yet
- CHS PDFDocument16 pagesCHS PDFOtniel Dita NugrohoNo ratings yet
- 2009 Fowler KoffasDocument11 pages2009 Fowler KoffasJenny MarfàNo ratings yet
- Chloroplast Import of Four Carotenoid Biosynthetic EnzymesDocument9 pagesChloroplast Import of Four Carotenoid Biosynthetic EnzymesNithin RNo ratings yet
- Anthocyanin Production in Transgenic Tomato Plants: Arabidopsis Thaliana MYB75/PAP1 Transcription Factor InducesDocument13 pagesAnthocyanin Production in Transgenic Tomato Plants: Arabidopsis Thaliana MYB75/PAP1 Transcription Factor InducesJeffrison William SaribuNo ratings yet
- Jmbfs 106 AmerDocument12 pagesJmbfs 106 AmerLuluk MaysarohNo ratings yet
- Effects of ABA On Primary Terpenoids and THC in Cannabis Sativa at Flowering StageDocument9 pagesEffects of ABA On Primary Terpenoids and THC in Cannabis Sativa at Flowering StageJaviercm10No ratings yet
- Keum 2008Document10 pagesKeum 2008Lucero HernándezNo ratings yet
- PHENOLSDocument4 pagesPHENOLSRaian D. QuelbioNo ratings yet
- Shikimate PathwayDocument13 pagesShikimate PathwayPavan GadamchettyNo ratings yet
- Ligan KuersetinDocument10 pagesLigan KuersetinrabiatulNo ratings yet
- Articulo Ref Licopeno 4Document9 pagesArticulo Ref Licopeno 4Eric Arturo Medina SuNo ratings yet
- AnthocyaninDocument5 pagesAnthocyaninNguyen HoaNo ratings yet
- Secondary Metabolyte - Organic Compound AnalysisDocument12 pagesSecondary Metabolyte - Organic Compound AnalysisSisilia Anabina TariganNo ratings yet
- Review Article Number 104 Molecular Organization of The Shikimate Pathway in Higher PlantsDocument13 pagesReview Article Number 104 Molecular Organization of The Shikimate Pathway in Higher PlantsJuan Valentin Delgadillo ColinNo ratings yet
- The Identification of Flavonoids and The Expression of Genes of Anthocyanin Biosynthesis in The Chrysanthemum FlowersDocument7 pagesThe Identification of Flavonoids and The Expression of Genes of Anthocyanin Biosynthesis in The Chrysanthemum FlowersYou Jin LimNo ratings yet
- Bennett2003 PDFDocument8 pagesBennett2003 PDFArief Adi NugrohoNo ratings yet
- 芸香CYP98A22Document13 pages芸香CYP98A22周正No ratings yet
- A Genomewide Admixture Mapping Study For Yield Factors and Morphological Traits in A Cultivated Cocoa (Theobroma Cacao L.) PopulationDocument9 pagesA Genomewide Admixture Mapping Study For Yield Factors and Morphological Traits in A Cultivated Cocoa (Theobroma Cacao L.) PopulationCamilo Ernesto Araujo BarabasNo ratings yet
- Organic Chemistry Experiment Report Phytochemical Test On The Extract of Wild Ginger (Curcuma Zanthorrhiza)Document18 pagesOrganic Chemistry Experiment Report Phytochemical Test On The Extract of Wild Ginger (Curcuma Zanthorrhiza)auraimanaNo ratings yet
- Initiation of The Exirubin Biosynthesis Inchitinophaga PinensisDocument10 pagesInitiation of The Exirubin Biosynthesis Inchitinophaga PinensisCarlos SilvaNo ratings yet
- Sphingomonas Aromaticivorans SP.: Nov., Sphingomonas Subterranea Sp. Nov., and Sphingomonas Stygia Sp. NovDocument11 pagesSphingomonas Aromaticivorans SP.: Nov., Sphingomonas Subterranea Sp. Nov., and Sphingomonas Stygia Sp. NovrahamathunnishaNo ratings yet
- Effect of PMT Gene Overexpression On Tropane Alkaloid Production in Transformed Root Cultures of Datura Metel and Hyoscyamus MuticusDocument9 pagesEffect of PMT Gene Overexpression On Tropane Alkaloid Production in Transformed Root Cultures of Datura Metel and Hyoscyamus MuticusHumiopNo ratings yet
- Invitro Quantification of Flavonoids and Phenolic Content of - SuranDocument5 pagesInvitro Quantification of Flavonoids and Phenolic Content of - SuranJimoh Daud SmartNo ratings yet
- Research Project: Prof. Dr. Rehab HamedDocument13 pagesResearch Project: Prof. Dr. Rehab HamedHassaninElaradyNo ratings yet
- Polyphenol Compounds and Their Benefits of Mangifera Indica L. (Var. Kottukonam) Grow in Varied Seasons and Altitude Shalaj Rasheed-125Document12 pagesPolyphenol Compounds and Their Benefits of Mangifera Indica L. (Var. Kottukonam) Grow in Varied Seasons and Altitude Shalaj Rasheed-12512th B 48 Akshay GadekarNo ratings yet
- 05-Terpenos y EsteroidesDocument23 pages05-Terpenos y EsteroidesAle VillaNo ratings yet
- pcx093 LaudanineDocument13 pagespcx093 LaudanineLulu SetiyabudiNo ratings yet
- Phenylpropanoid BiosynthesisDocument19 pagesPhenylpropanoid BiosynthesisAnn MayNo ratings yet
- Bergman and Phillips 2020 Phytochemistry Reviews OnlineDocument27 pagesBergman and Phillips 2020 Phytochemistry Reviews OnlineLeen OdehNo ratings yet
- Functional Expression of Horseradish Peroxidase in Saccharomyces Cerevisiae and Pichia PastorisDocument8 pagesFunctional Expression of Horseradish Peroxidase in Saccharomyces Cerevisiae and Pichia PastorisAlan SublimeNo ratings yet
- Metabolic Engineering of Yeast For The Production of Plant Secondary MetabolitesDocument4 pagesMetabolic Engineering of Yeast For The Production of Plant Secondary MetabolitesGung De RypNo ratings yet
- FengDocument12 pagesFengAJMRNo ratings yet
- Liu IIIDocument8 pagesLiu IIIaji subaktiNo ratings yet
- Neil C. Bruce - AlkaloidsDocument11 pagesNeil C. Bruce - AlkaloidsHumiopNo ratings yet
- Trantas 2009Document12 pagesTrantas 2009Cristofer Perez SeguraNo ratings yet
- Isolasi Dan Identifikasi Senyawa Steroid Fraksi Kloroform Dari FRAKSINASI EKSTRAK METANOL DAUN KEREHAU (Callicarpa Longifolia Lam.)Document6 pagesIsolasi Dan Identifikasi Senyawa Steroid Fraksi Kloroform Dari FRAKSINASI EKSTRAK METANOL DAUN KEREHAU (Callicarpa Longifolia Lam.)Wenti Adya SilvanyNo ratings yet
- 1 - Intro To Enzymes PDFDocument93 pages1 - Intro To Enzymes PDFAnonymous ojJ64xSOGNo ratings yet
- 罗勒3羟化酶Document9 pages罗勒3羟化酶周正No ratings yet
- Paper Seminario 03Document13 pagesPaper Seminario 03aznatsnoc hgNo ratings yet
- 5c0a PDFDocument9 pages5c0a PDFnelisaNo ratings yet
- Won 2003Document14 pagesWon 2003Roberto C. BárcenasNo ratings yet
- Group 03 (2A) 2Document8 pagesGroup 03 (2A) 2-No ratings yet
- Paper 7 PDFDocument6 pagesPaper 7 PDFFatinKhusmarinaNo ratings yet
- Petroleum PDFDocument11 pagesPetroleum PDFGabrielCamarenaNo ratings yet
- Ukr Biochem J 2019vol 91N3Document15 pagesUkr Biochem J 2019vol 91N3aisNo ratings yet
- tmp83F2 TMPDocument10 pagestmp83F2 TMPFrontiersNo ratings yet
- Pseudomonas Was Second Most Dominant: Pseudomonas and The Plant AlsoDocument5 pagesPseudomonas Was Second Most Dominant: Pseudomonas and The Plant AlsofupaNo ratings yet
- Plant Physiol.-2005-González-Ballester-522-33Document12 pagesPlant Physiol.-2005-González-Ballester-522-33Jaisson Miyosi OkaNo ratings yet
- Structural Implications On Color, Xuorescence, and Antiradical Activity in BetalainsDocument12 pagesStructural Implications On Color, Xuorescence, and Antiradical Activity in BetalainsNeyder PantojaNo ratings yet
- Anthocyanin Production by Over-Expression of GrapeDocument9 pagesAnthocyanin Production by Over-Expression of GrapeSudarshanee GeekiyanageNo ratings yet
- Labdanes of Natural Origin-Biological ActivitiesDocument24 pagesLabdanes of Natural Origin-Biological Activitiestheyuri@tlen.plNo ratings yet
- Phenylpropanoid Biosynthesis: Thomas VogtDocument19 pagesPhenylpropanoid Biosynthesis: Thomas VogtEster Setyaning Anjani PutriNo ratings yet
- The Total Synthesis of Natural ProductsFrom EverandThe Total Synthesis of Natural ProductsJohn ApSimonNo ratings yet
- Operating Limits of Steels in Hydrogen Service - From API RP 941Document1 pageOperating Limits of Steels in Hydrogen Service - From API RP 941Ajmi HmidaNo ratings yet
- General Epidemiology:: Historical Development, Definition, Scope and ApplicationDocument32 pagesGeneral Epidemiology:: Historical Development, Definition, Scope and ApplicationscholasticaNo ratings yet
- Purcom Speech 1Document1 pagePurcom Speech 1LUCYDHARYLL JOHN E. SEJALBONo ratings yet
- Pa Schools CurriculumDocument33 pagesPa Schools CurriculumNathalee WalkerNo ratings yet
- Unit 6: Energy Loss in PipelinesDocument25 pagesUnit 6: Energy Loss in PipelinesAnn RazonNo ratings yet
- Clv2050 Masterplan PDFDocument268 pagesClv2050 Masterplan PDFZNo ratings yet
- DM CHFDocument2 pagesDM CHFSafa Abdualrahaman Ali HamadNo ratings yet
- Qur'an and The BrainDocument7 pagesQur'an and The BrainFatin Farhana RahamanNo ratings yet
- PRM40-VUCA-Question PaperDocument11 pagesPRM40-VUCA-Question PapersrishtyshamNo ratings yet
- Handful of Leaves-An Anthology From The Anguttara Nikaya-Vol-3Document388 pagesHandful of Leaves-An Anthology From The Anguttara Nikaya-Vol-3actionistNo ratings yet
- Maintenance PKSDocument63 pagesMaintenance PKSBunga PanjaitanNo ratings yet
- Comparison of The Combustion Characteristics of Rice HuskDocument4 pagesComparison of The Combustion Characteristics of Rice HuskMicah Juguan SalgadoNo ratings yet
- Manuscript Revised 51419 TOCDocument10 pagesManuscript Revised 51419 TOCRex CamiroNo ratings yet
- Safety Data Sheet Stencil Cleaner Scs/13: 1 Identification of The Substance/Preparation and of The Company/UndertakingDocument3 pagesSafety Data Sheet Stencil Cleaner Scs/13: 1 Identification of The Substance/Preparation and of The Company/UndertakingSuriyachai NiamsornNo ratings yet
- AnalogyDocument24 pagesAnalogyTrung TranNo ratings yet
- Activated Charcoal For Acute OverdoseDocument6 pagesActivated Charcoal For Acute OverdoseTony ChuNo ratings yet
- Borderline Personality Disorder: An Interview With Psychiatrist Dr. Roy KrawitzDocument8 pagesBorderline Personality Disorder: An Interview With Psychiatrist Dr. Roy KrawitzClinton PowerNo ratings yet
- Catalan - Lbyme4a - Ee3 - Design of Experiment - Written ReportDocument5 pagesCatalan - Lbyme4a - Ee3 - Design of Experiment - Written Reportcatalan153709No ratings yet
- Sika PDS E Plastomix - 705Document2 pagesSika PDS E Plastomix - 705lwin_oo2435No ratings yet
- 5.0 Module Pahang For Chemical BondingDocument19 pages5.0 Module Pahang For Chemical Bondingkhayranizam0% (1)
- Guidance Notes On Use of Uterotonics During LaborDocument28 pagesGuidance Notes On Use of Uterotonics During LaborKinjal VasavaNo ratings yet
- Health Problems MyselfDocument54 pagesHealth Problems MyselfKrishnaveni Murugesh100% (2)
- RRL ReviseDocument3 pagesRRL ReviseKerby SabbalucaNo ratings yet
- Myx Menu (New)Document16 pagesMyx Menu (New)G Sathesh KumarNo ratings yet
- English Vocabulary For Mobile Phones / Cell PhonesDocument10 pagesEnglish Vocabulary For Mobile Phones / Cell PhoneskevinNo ratings yet
- Demeuse Teaching PaperDocument8 pagesDemeuse Teaching Paperapi-512525946No ratings yet