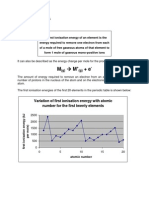
Professional Documents
Culture Documents
Ionisation Energies
Ionisation Energies
Uploaded by
stupid gurlzOriginal Description:
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Ionisation Energies
Ionisation Energies
Uploaded by
stupid gurlzCopyright:
Available Formats
1.
1 Exercise 4 ionisation energies
1. Why does the first ionisation energy of atoms generally increase across a period?
2. Why is the first ionisation energy of boron less than that of beryllium?
3. Why is the first ionisation energy of oxygen less than that of nitrogen?
4. Why do first ionisation energies decrease down a group?
5. Why does helium have the highest first ionisation energy of all the elements?
6. Why is the second ionisation energy of an atom always greater than the first?
7. Why is the second ionisation energy of sodium much greater than the first?
8. Why does atomic size decrease across a period?
9. Why does atomic size increase down a group?
10. Why are cations always smaller than the corresponding atoms?
11. Why are anions always larger than the corresponding atoms?
1.1 Exercise 4
1.
Number of protons increases, shielding stays the same, so attraction of outer electrons to nucleus
increases
2.
Outermost electron in B is 2p, outermost electron in Be is 2s, 2p electron in B better shielded than 2s
electron in Be, so it is less attracted to nucleus
3.
2p electron is paired in O but unpaired in N, so in O there is more repulsion in the orbital which
makes the electron easier to remove
4.
More shells, so more shielding, so attraction of outer electrons to the nucleus decreases
5.
No shielding in 1st period so electrons closely held than in other periods, and more protons than
hydrogen so greater attraction to nucleus
6.
Less electrons, so less electron repulsion
7.
1st electron removed from 3s, second electron removed from 2p so much less shielding
8.
Number of protons increases, shielding stays the same, so attraction of outer electrons to nucleus
increases and they move closer
9.
More shells, so more shielding, so attraction of outer electrons to the nucleus decreases and they are
pushed further away
10.
Less electrons, so less repulsion, so electrons can get closer to the nucleus
11.
More electrons, so more repulsion, so electrons are pushed further away
Mill Hill High School
6.
The values of the first ionisation energies of neon, sodium and magnesium are 2080,
494 and 736 kJ mol1, respectively.
(a)
Explain the meaning of the term first ionisation of an atom.
......................
......................
......................
(2)
(b)
Write an equation to illustrate the process occurring when the second ionisation
energy of magnesium is measured.
......................
......................
(2)
(c)
Explain why the value of the first ionisation energy of magnesium is higher than
that of sodium.
......................
......................
......................
(2)
(d)
Explain why the value of the first ionisation energy of neon is higher than that of
sodium.
......................
......................
......................
(2)
(Total 8 marks)
6.
(a)
(b)
(c)
(d)
Enthalpy change/required when an electron is removed/knocked
out/displaced (Ignore minimum energy)
From a gaseous atom
(could get this mark from equation)
Mg+(g) Mg2+(g) + e
Equation
+
2+
Or Mg (g) + e Mg (g) + 2e State symbols (Tied to M1)
1
1
Increased/stronger nuclear charge or more protons
1
Smaller atom or electrons enter the same shell or same/similar shielding 1
Electron removed from a shell of lower energy or smaller atom or e nearer
nucleus or e removed from 2p rather than from 3s
Less shielding
(Do not accept e from inner shell)
[8]
Mill Hill High School
4.
(a)
Write equations to show the chemical processes which occur when the first and the second
ionisation energies of lithium are measured.
First ionisation energy equation .........................................................................
Second ionisation energy equation .....................................................................
(3)
(b)
(i)
Explain why helium has a much higher first ionisation energy than lithium.
...................................................................................................................
...................................................................................................................
...................................................................................................................
(ii)
Explain why beryllium has a higher first ionisation energy than boron.
...................................................................................................................
...................................................................................................................
...................................................................................................................
(iii)
Explain why the first ionisation energy of krypton is greater than the first ionisation
energy of bromine.
..............................................................................................................................
..............................................................................................................................
(iv)
Explain why the second ionisation energy of beryllium is greater than the first
ionisation energy.
...................................................................................................................
...................................................................................................................
...................................................................................................................
(8)
(Total 11 marks)
4.
(a)
(b)
First ionisation energy equation
Li(g) Li+(g) + e (1)
Second ionisation energy equation
Li+(g) Li2+(g) e (1)
tate symbols (1)
(i)
Hes electron in 1s (1)
closer to nucleus (or no shielding) (1)
(or converse argument for Li)
(ii)
Bes outer electron in 2s (1)
lower in energy than 2p (1)
(iii)
more protons (1)
or increased nuclear charge
attracting electrons in the same {shell/orbital/subshell/energy level (1)
or similar shielding
(iv)
Electron removed from positive ion (1)
which attracts the electron more (1)
(allow for 2nd electron nearer to the nucleus)
8
[11]
Mill Hill High School
You might also like
- Problem-Oriented Medical Diagnosis 7thDocument228 pagesProblem-Oriented Medical Diagnosis 7thnhtoan3286% (7)
- IOM NetProDual Daikin Rev 4 PDFDocument15 pagesIOM NetProDual Daikin Rev 4 PDFVin Ken0% (1)
- Markowitz WhitepaperDocument10 pagesMarkowitz WhitepaperWen BinNo ratings yet
- Electrons in Atoms.Document62 pagesElectrons in Atoms.Md.Tanjim reza TurjoNo ratings yet
- Chem 111 Week Three Notes BDocument7 pagesChem 111 Week Three Notes BnyachiosirNo ratings yet
- Ionisation Energy Q& A SampleDocument2 pagesIonisation Energy Q& A SampleShalu Princess DikshNo ratings yet
- 2 13 Ionisation EnergiesDocument6 pages2 13 Ionisation EnergiesRobertLiu100% (2)
- Che Chapter 3Document7 pagesChe Chapter 3lisaNo ratings yet
- Periodic Table and PeriodicityDocument11 pagesPeriodic Table and Periodicityakeemoluwadamilare623No ratings yet
- S2 - 3 (By: Feroz Akhtar)Document4 pagesS2 - 3 (By: Feroz Akhtar)Feroz AkhtarNo ratings yet
- Worksheet BenzenaDocument5 pagesWorksheet BenzenaRizky KurniawatiNo ratings yet
- Ionisation EnergyDocument6 pagesIonisation EnergyAathifa ThowfeekNo ratings yet
- Patterns of First Ionisation Energies in The Periodic TableDocument7 pagesPatterns of First Ionisation Energies in The Periodic TableRugen RajNo ratings yet
- Ionisation EnergyDocument12 pagesIonisation Energyhahajing1980No ratings yet
- Engineering Chemistry Notes UNIT 4Document9 pagesEngineering Chemistry Notes UNIT 4Nivetha ENo ratings yet
- Chemistry Note Ionic and Molecular Compounds'Document13 pagesChemistry Note Ionic and Molecular Compounds'zekarias wondafrashNo ratings yet
- Unit 1 Commonly Asked QuestionsDocument28 pagesUnit 1 Commonly Asked Questionssofia begumNo ratings yet
- Topic 1 - Atomic StructureDocument4 pagesTopic 1 - Atomic Structurejulian maltoNo ratings yet
- 2 - Periodicity in Atomic Properties (Structure 3.1.3)Document27 pages2 - Periodicity in Atomic Properties (Structure 3.1.3)JouliesNo ratings yet
- AS Level Summary-Unit 1Document11 pagesAS Level Summary-Unit 1Filiz Kocayazgan ShanablehNo ratings yet
- 11c Classification, Periodicity (Class Notes)Document20 pages11c Classification, Periodicity (Class Notes)RAJEEV KARAKOTINo ratings yet
- A-Level Chemistry NotesDocument10 pagesA-Level Chemistry NotesHannah Bryson-JonesNo ratings yet
- HL Electron Configuration and The Periodic TableDocument12 pagesHL Electron Configuration and The Periodic TableMarilee HuntNo ratings yet
- Ionization Energy and Electron AffinityDocument9 pagesIonization Energy and Electron AffinityKhan AaghaNo ratings yet
- Chemistry Unit 1 Edexcel Notes (AS Level)Document1 pageChemistry Unit 1 Edexcel Notes (AS Level)--------100% (1)
- Ionisation Energy EdexcelDocument5 pagesIonisation Energy EdexcelKevin The Chemistry Tutor100% (1)
- 08 Physics 11se Ch08Document42 pages08 Physics 11se Ch08pecan_lisa38No ratings yet
- 3 2 Physical PropertiesDocument4 pages3 2 Physical PropertiesNguyenHoangMinhDucNo ratings yet
- A - Level Inorganic Chemistry 2-1Document171 pagesA - Level Inorganic Chemistry 2-1kitderoger_391648570No ratings yet
- Ion EnergiesDocument4 pagesIon EnergiesNamratha NairNo ratings yet
- A'Level Inorganic ChemistryDocument84 pagesA'Level Inorganic Chemistrymukisajob586No ratings yet
- Unit Objectives: Periodic TrendsDocument9 pagesUnit Objectives: Periodic Trendsmartin mulengaNo ratings yet
- Chapter 1 - Atomic StructureDocument9 pagesChapter 1 - Atomic Structureleonide357No ratings yet
- Ionization EnergyDocument69 pagesIonization EnergyVisalakshi Venkat100% (2)
- Chemistry Semester 1 Final Study Guide KeyDocument7 pagesChemistry Semester 1 Final Study Guide Keyalexanderhdinh50% (2)
- Ionization EnergyDocument8 pagesIonization EnergyHafiza Sikder AnishaNo ratings yet
- AQA A Level Chemistry Unit 1 NotesDocument17 pagesAQA A Level Chemistry Unit 1 NotesMuadh Chati100% (1)
- PeriodicityDocument6 pagesPeriodicityNetkoNo ratings yet
- Electron Configuration and The Periodic PropertiesDocument4 pagesElectron Configuration and The Periodic Propertiesapi-240883010No ratings yet
- Chapter 2 - Electrons in AtomsDocument32 pagesChapter 2 - Electrons in AtomsFandyNo ratings yet
- Chapter 2-Electrons in AtomsDocument41 pagesChapter 2-Electrons in AtomsFelix LiandaNo ratings yet
- Amit Bikram Mishra - BSCH201Document10 pagesAmit Bikram Mishra - BSCH201Amit Bikram MishraNo ratings yet
- Ebenezer Matric .Hr. Sec. School: 1.what Is Ionization Energy?Document3 pagesEbenezer Matric .Hr. Sec. School: 1.what Is Ionization Energy?r karthickNo ratings yet
- Inorganic Chemistry NotesDocument105 pagesInorganic Chemistry NotesOdongo TonnyNo ratings yet
- Classification of Elements & Periodic Table: Iind PartDocument31 pagesClassification of Elements & Periodic Table: Iind PartKeshan PaudelNo ratings yet
- Chem Topic 2 and 3Document4 pagesChem Topic 2 and 3aliyaahsnahNo ratings yet
- Ionisation Energy ClassDocument13 pagesIonisation Energy ClassKordell McShineNo ratings yet
- PeriodicTable and Trends DPDocument109 pagesPeriodicTable and Trends DPSurya NairNo ratings yet
- 5 Trends in The Periodic TableDocument4 pages5 Trends in The Periodic TableCris CorsinoNo ratings yet
- Ionisation EnergyDocument6 pagesIonisation EnergySiya ChiniahNo ratings yet
- Grade 8 Atomic Structure - Notes...Document7 pagesGrade 8 Atomic Structure - Notes...Antonita100% (1)
- A - Level Inorganic ChemistryDocument543 pagesA - Level Inorganic Chemistrystephenshada9No ratings yet
- Ionization Energy Trend in The Periodic TableDocument3 pagesIonization Energy Trend in The Periodic TableOluwabamire OreoluwaNo ratings yet
- As 1 PerDocument4 pagesAs 1 PerzoeakatNo ratings yet
- Chapter 3 Periodic Table (SUEC) AnswerDocument12 pagesChapter 3 Periodic Table (SUEC) AnswerLee AustinNo ratings yet
- AtomsDocument5 pagesAtomsjijigox479No ratings yet
- Ionization Energy Cape Unit 1Document21 pagesIonization Energy Cape Unit 1Shanice JohnsonNo ratings yet
- Theperiodictable 23Document17 pagesTheperiodictable 23Muneer KayaniNo ratings yet
- UntitledDocument27 pagesUntitledFatimah AgboolaNo ratings yet
- Tanapp Tcm18-410hurDocument5 pagesTanapp Tcm18-410hurNguh FabriceNo ratings yet
- Classification of ElementsDocument74 pagesClassification of ElementsJimit Patel BankNo ratings yet
- CHM 111 Lecture Note FiveDocument5 pagesCHM 111 Lecture Note Fivealfreddanladi321No ratings yet
- Is Nuclear Energy Safe? -Nuclear Energy and Fission - Physics 7th Grade | Children's Physics BooksFrom EverandIs Nuclear Energy Safe? -Nuclear Energy and Fission - Physics 7th Grade | Children's Physics BooksNo ratings yet
- IEC 62040-2 Uninterruptible Power Systems (Ups) - Part 2: Electromagnetic Compatibility (EMC) Requirements Interpretation Sheet 1Document3 pagesIEC 62040-2 Uninterruptible Power Systems (Ups) - Part 2: Electromagnetic Compatibility (EMC) Requirements Interpretation Sheet 1Agfagf212No ratings yet
- Caterpillar Monitoring SystemDocument3 pagesCaterpillar Monitoring SystemFajarTaufiq100% (1)
- Psychometric Success Mechanical Reasoning Practice Test 1Document0 pagesPsychometric Success Mechanical Reasoning Practice Test 1felixdelaliNo ratings yet
- Gidaa Alamry: AbstractDocument15 pagesGidaa Alamry: AbstractJann Ernest R. FesalbonNo ratings yet
- GeographyDocument13 pagesGeographyRao RaasNo ratings yet
- ManualeHardware L15 enDocument129 pagesManualeHardware L15 enAndreaNo ratings yet
- CS 701 Research Paper (MS150200090)Document7 pagesCS 701 Research Paper (MS150200090)Mian Saeed RafiqueNo ratings yet
- 30MHZ AM ReceiverDocument1 page30MHZ AM ReceiverrujNo ratings yet
- Unit 4Document53 pagesUnit 4Ashish JoshiNo ratings yet
- MI20190007 Resolution and OrderDocument10 pagesMI20190007 Resolution and OrderLa Isla OesteNo ratings yet
- Chapter 4 PLANTDocument3 pagesChapter 4 PLANTSedsed QuematonNo ratings yet
- RTWP - 2Document62 pagesRTWP - 2AsmaeNo ratings yet
- SJ 46np46n49np49nDocument8 pagesSJ 46np46n49np49nSiti Khadijah Ab RahmanNo ratings yet
- Extrusion and Drawing PDFDocument16 pagesExtrusion and Drawing PDFAravindhan AnbalaganNo ratings yet
- Astm D2487 11Document21 pagesAstm D2487 11angie rodriguezNo ratings yet
- ATIC112Document1 pageATIC112lennon rNo ratings yet
- SAMPLE MCQS For REAL ESTATE APPRAISER LICENSURE EXAMSDocument4 pagesSAMPLE MCQS For REAL ESTATE APPRAISER LICENSURE EXAMSadobopinikpikan0% (1)
- Honda Accord Saloon 7th Gen FaceliftDocument43 pagesHonda Accord Saloon 7th Gen FaceliftAOCUK50% (2)
- Pre-Flight Inspection R22Document73 pagesPre-Flight Inspection R22Ann LechatNo ratings yet
- Q-A Ch-4 Tribals, DikusDocument3 pagesQ-A Ch-4 Tribals, Dikusyuvraj singhNo ratings yet
- Q-PANEL Standard Substrate Applications GuideDocument2 pagesQ-PANEL Standard Substrate Applications GuideGhulam HussainNo ratings yet
- Performance of Digital Communication LabDocument4 pagesPerformance of Digital Communication LabFrogie HuniebieNo ratings yet
- B02. Junior Engineer MechanicalDocument115 pagesB02. Junior Engineer MechanicalRafay MohiuddinNo ratings yet
- Reed Switch Appln PDFDocument15 pagesReed Switch Appln PDFJeeva BharathiNo ratings yet
- Kateofgaia Wordpress Com 2014-05-07 Babylon Is FallenDocument18 pagesKateofgaia Wordpress Com 2014-05-07 Babylon Is FallenJames WatsonNo ratings yet
- 10 Cum - DayDocument5 pages10 Cum - DayAbhijit MondalNo ratings yet
- Switzer - Genset PlanDocument1 pageSwitzer - Genset PlanArun MohanNo ratings yet