Professional Documents
Culture Documents
0 ratings0% found this document useful (0 votes)
67 viewsU3 2b
U3 2b
Uploaded by
yunohuEach electron in an atom is described by four quantum numbers: principal (n), angular momentum (λ), magnetic (m), and electron spin (s). The principal quantum number n determines the electron's energy level and shell (K, L, M, N etc.). The angular momentum quantum number λ indicates the orbital shape (s, p, d, f etc.) and allowed values are 0 to n-1. The magnetic quantum number m indicates the orbital orientation and allowed values are -λ to +λ. The electron spin quantum number s is either +1/2 or -1/2.
Copyright:
© All Rights Reserved
Available Formats
Download as PPT, PDF, TXT or read online from Scribd
You might also like
- Quantum Numbers 2. Azimuthal Quantum Number: (L)Document2 pagesQuantum Numbers 2. Azimuthal Quantum Number: (L)Selva ManiNo ratings yet
- Quantum NumbersDocument39 pagesQuantum NumbersTin SumangaNo ratings yet
- Atomic Structure: 2.2: Quantum MechanicsDocument48 pagesAtomic Structure: 2.2: Quantum MechanicsDaniel LimNo ratings yet
- 202003291623595323nkpandey Quantum NumbersDocument3 pages202003291623595323nkpandey Quantum NumbersBk Creative StudioNo ratings yet
- Orbitals and Quantum NumberDocument21 pagesOrbitals and Quantum NumberPan Kylie KateNo ratings yet
- Quantum Numbers and ECDocument3 pagesQuantum Numbers and ECShraddha Deshmukh-KelkarNo ratings yet
- CHEM 2101 Lecture 2 (Qunatum Number)Document5 pagesCHEM 2101 Lecture 2 (Qunatum Number)Asif UddinNo ratings yet
- Quantum Numbers Quantum NumbersDocument2 pagesQuantum Numbers Quantum Numbersaijaz100No ratings yet
- Quantum NumbersDocument3 pagesQuantum NumbersMia Pearl Tabios ValenzuelaNo ratings yet
- Advancechem Q1 LP4Document7 pagesAdvancechem Q1 LP4Francesca BuenoNo ratings yet
- AtomsFirst2e Day6 Sec3.7-3.8 CHMY171 Fall 2015Document17 pagesAtomsFirst2e Day6 Sec3.7-3.8 CHMY171 Fall 2015Xenia Mae FloresNo ratings yet
- Quantum NumbersDocument12 pagesQuantum NumbersSoub kuopNo ratings yet
- Atomic and Molecular Struc.Document25 pagesAtomic and Molecular Struc.lelouchvibritannia0502No ratings yet
- L3 Che101Document28 pagesL3 Che101Musa Ahammed MahinNo ratings yet
- AtomsFirst2e Day6 Sec3.7-3.8 CHMY171 Fall 2015Document17 pagesAtomsFirst2e Day6 Sec3.7-3.8 CHMY171 Fall 2015Claudius MorpheusNo ratings yet
- Quantum Numbers: Are Required To Describe The Distribution ofDocument17 pagesQuantum Numbers: Are Required To Describe The Distribution ofIrfan Sayeem SultanNo ratings yet
- q2 General Chemistry Rev (Complete)Document14 pagesq2 General Chemistry Rev (Complete)rosedancel52No ratings yet
- Atomic Orbitals: Quantum NumbersDocument16 pagesAtomic Orbitals: Quantum NumberslostgirlNo ratings yet
- 1A The Shapes and Structures of Molecules Student Handout Part Two 2022.23Document70 pages1A The Shapes and Structures of Molecules Student Handout Part Two 2022.23Music MaestroNo ratings yet
- Atomic OrbitalDocument25 pagesAtomic OrbitalCarolle PerezNo ratings yet
- Quantum Numbers and Electron ConfigurationsDocument57 pagesQuantum Numbers and Electron Configurationscath100% (1)
- How To Use Quantum Numbers To Describe AnDocument11 pagesHow To Use Quantum Numbers To Describe AnAntonio Exal ColladoNo ratings yet
- Atomic StructureDocument19 pagesAtomic StructureJhoanna Marie Manuel-AbelNo ratings yet
- Quantum Numbers and Electron Configuration.: ST ND RD THDocument2 pagesQuantum Numbers and Electron Configuration.: ST ND RD THKelly Gabriel Singuay OcceñoNo ratings yet
- Quantum NumbersDocument8 pagesQuantum NumbersMoises espinozaNo ratings yet
- 1 Quantum Numbers-1Document34 pages1 Quantum Numbers-1Carlo James SablanNo ratings yet
- Electronic Structure of MatterDocument3 pagesElectronic Structure of Matterilikegay2dmensNo ratings yet
- Interpretation of Quantum Numbers: TU Bergakademie FreibergDocument2 pagesInterpretation of Quantum Numbers: TU Bergakademie FreibergPraveenNo ratings yet
- Quantum Numbers and Atomic OrbitalsDocument16 pagesQuantum Numbers and Atomic OrbitalsR-jayVenturilloNo ratings yet
- Chemistry - WEEK 3 - LAS (Quantum Numbers)Document5 pagesChemistry - WEEK 3 - LAS (Quantum Numbers)EZRA THERESE DE JESUSNo ratings yet
- Rhael Denielle P. Sunio 12 - Archimedes General Chemistry 1 - Module 1Document6 pagesRhael Denielle P. Sunio 12 - Archimedes General Chemistry 1 - Module 1Leah SunioNo ratings yet
- Quantum Numbers and Electron ConfigurationsDocument16 pagesQuantum Numbers and Electron ConfigurationsMARVIN OTHELLO MELCHOR DAUISNo ratings yet
- AD SirDocument23 pagesAD Sirlost dharNo ratings yet
- De Broglie Relation: EinsteinDocument19 pagesDe Broglie Relation: EinsteinMaisha MaimunaNo ratings yet
- Chemistry Notes Part 1Document59 pagesChemistry Notes Part 1SharleneNo ratings yet
- P11 ADocument9 pagesP11 ADana CapbunNo ratings yet
- DME I Year CHEDocument91 pagesDME I Year CHERadha KrishnaNo ratings yet
- Structure of AtomDocument29 pagesStructure of AtomAbhisek DehuriNo ratings yet
- Quantum Numbers Structure 2Document22 pagesQuantum Numbers Structure 2vishalsingh1997No ratings yet
- Chemistry Quantum NumbersDocument9 pagesChemistry Quantum NumbersSamiksha SainiNo ratings yet
- Unit - IV - Uv VisibleDocument27 pagesUnit - IV - Uv VisibleM. BalakrishnanNo ratings yet
- General Chemistry 1 M2W2Document7 pagesGeneral Chemistry 1 M2W2Warley JabelNo ratings yet
- Lecture 2 Part 2Document40 pagesLecture 2 Part 2ariannemariebaronNo ratings yet
- Atomic StructureDocument2 pagesAtomic StructureNarjis FatimaNo ratings yet
- Lecture 4 Wavefunction NewDocument53 pagesLecture 4 Wavefunction NewkedirNo ratings yet
- Quantum NumbersDocument15 pagesQuantum NumbersMohamed KilanyNo ratings yet
- Chapter 2 - Structure of Atom PeridiocityDocument97 pagesChapter 2 - Structure of Atom PeridiocityAina AthirahNo ratings yet
- CEM1008F - 3. The Periodic Table and Atomic Structure 2024 - Lecture - NotesDocument41 pagesCEM1008F - 3. The Periodic Table and Atomic Structure 2024 - Lecture - Notesparlypalesa90No ratings yet
- Exercises No. 2Document6 pagesExercises No. 2John Miguel BarbaNo ratings yet
- The Electronic Structure of AtomsDocument54 pagesThe Electronic Structure of AtomsElijah PunzalanNo ratings yet
- 2.2 Quantum Mechanis ModelDocument53 pages2.2 Quantum Mechanis ModelAbdullah AhmadNo ratings yet
- (L-10) - (JEE 3.0) - Atomic Structure - 11th May.Document57 pages(L-10) - (JEE 3.0) - Atomic Structure - 11th May.Thomas ShelbyNo ratings yet
- Quantum Numbers and Their Physical InterpetationDocument15 pagesQuantum Numbers and Their Physical InterpetationMomina LaraibNo ratings yet
- Schroedinger Equation Atomic Wave Functions Atomic Orbitals Quantum NumbersDocument45 pagesSchroedinger Equation Atomic Wave Functions Atomic Orbitals Quantum NumbersRishit JakhariaNo ratings yet
- Quantum Numbers For AtomsDocument6 pagesQuantum Numbers For AtomsMaricae NathanielNo ratings yet
- CHM131 - Chapter 2 - Structure of Atom - PeriodicityDocument97 pagesCHM131 - Chapter 2 - Structure of Atom - PeriodicityLeo PietroNo ratings yet
- Grade 9 SCIENCE (JAN. 18-22)Document3 pagesGrade 9 SCIENCE (JAN. 18-22)Jhev LeopandoNo ratings yet
- Lecture 2Document18 pagesLecture 2Ayan PatelNo ratings yet
- Quantum Numbers PowerpointDocument12 pagesQuantum Numbers Powerpointapi-201447595No ratings yet
- Electron Beam-Specimen Interactions and Simulation Methods in MicroscopyFrom EverandElectron Beam-Specimen Interactions and Simulation Methods in MicroscopyNo ratings yet
- Shape of Atomic OrbitalsDocument5 pagesShape of Atomic OrbitalsyunohuNo ratings yet
- The Anomalous Electronic Configurations of CR and CuDocument6 pagesThe Anomalous Electronic Configurations of CR and CuyunohuNo ratings yet
- Representing Electronic Configuration: Orbital DiagramDocument1 pageRepresenting Electronic Configuration: Orbital DiagramyunohuNo ratings yet
- Atomic OrbitalDocument1 pageAtomic OrbitalyunohuNo ratings yet
- MATTER (1.2 Mole Concept)Document101 pagesMATTER (1.2 Mole Concept)yunohuNo ratings yet
- C5.population GeneticsDocument28 pagesC5.population GeneticsyunohuNo ratings yet
- Sambungan P-N Dan Dioda Sambungan P-NDocument27 pagesSambungan P-N Dan Dioda Sambungan P-NUlfie Kusuma Wardhani100% (1)
- MAMALUBA - Assignment For Chem Lab 401Document2 pagesMAMALUBA - Assignment For Chem Lab 401Sophia Indirah MamalubaNo ratings yet
- Quantum Mechanics Course ZeemansplittingDocument29 pagesQuantum Mechanics Course ZeemansplittingjlbalbNo ratings yet
- Balancing Equations: Practice ProblemsDocument10 pagesBalancing Equations: Practice ProblemsMoriel Barrogo BequilloNo ratings yet
- Balancing Chemical EquationsDocument18 pagesBalancing Chemical EquationsWendye YimamNo ratings yet
- Daftar Bahan Kimia PaDocument4 pagesDaftar Bahan Kimia Papanda_oonNo ratings yet
- Salt AnalysisDocument15 pagesSalt AnalysisPrabhanshu Pandey (Chotu)No ratings yet
- Properties of Silver (I), Mercury (I) and Lead (II) SaltsDocument7 pagesProperties of Silver (I), Mercury (I) and Lead (II) SaltsnothaboNo ratings yet
- Born-Haber Cycle Born-Haber Cycle: Why Not Nacl ? Why MGCL ?Document1 pageBorn-Haber Cycle Born-Haber Cycle: Why Not Nacl ? Why MGCL ?Fara FransiskaNo ratings yet
- DFT PresentationDocument63 pagesDFT PresentationhammmoudaNo ratings yet
- Chemistry: Form 4-Chapter 8Document7 pagesChemistry: Form 4-Chapter 8Azrel YusoffNo ratings yet
- 2-Synthetic Methods Chap 2Document81 pages2-Synthetic Methods Chap 2MahsaNo ratings yet
- Intrinsic & Extrinsic SemiconductorsDocument20 pagesIntrinsic & Extrinsic SemiconductorsmaharjansumirNo ratings yet
- MCQ SemiconductorDocument12 pagesMCQ Semiconductormuktadir hosenNo ratings yet
- Introduction To Material Chem.Document23 pagesIntroduction To Material Chem.Gaurav MauryaNo ratings yet
- Assignment - EEE 103Document4 pagesAssignment - EEE 103Abu BasharNo ratings yet
- Section 18B Metric Salt TablesDocument22 pagesSection 18B Metric Salt TablesLazharNo ratings yet
- Transport in Graphene: Klaus RichterDocument50 pagesTransport in Graphene: Klaus RichterAnonymous 9rJe2lOskxNo ratings yet
- P Block Reactions PDFDocument3 pagesP Block Reactions PDFTarundeepNo ratings yet
- Microwave Mediated Green Synthesis of Copper Nanoparticles Using Aqueous Extract of Seeds and Particles CharacterisationDocument12 pagesMicrowave Mediated Green Synthesis of Copper Nanoparticles Using Aqueous Extract of Seeds and Particles Characterisationmiguel salasNo ratings yet
- Brine CalculatorDocument2 pagesBrine CalculatorzachNo ratings yet
- Experiment No. 1: Aim: To Identify The Cation and Anion in Copper Sulphate PentahydrateDocument3 pagesExperiment No. 1: Aim: To Identify The Cation and Anion in Copper Sulphate PentahydrateElite GamingNo ratings yet
- Chem 373 - Lecture 27: Homonuclear Diatomic Molecules-IIDocument22 pagesChem 373 - Lecture 27: Homonuclear Diatomic Molecules-IINuansak3No ratings yet
- Chapter 18: Electrical Properties II: Intrinsic & Extrinsic SemiconductorsDocument20 pagesChapter 18: Electrical Properties II: Intrinsic & Extrinsic SemiconductorsManish KumarNo ratings yet
- Epitaxy GrowthDocument24 pagesEpitaxy GrowthMD. MEHEDI HASAN100% (3)
- DatasheetDocument87 pagesDatasheetmdelcos100% (1)
- DiodeDocument19 pagesDiodeRamu kakaNo ratings yet
- 3.3 IV Characteristics of PN Junction Diode-ModifiedDocument18 pages3.3 IV Characteristics of PN Junction Diode-Modifiedraunak guptaNo ratings yet
- Chem 373 - Lecture 17: Radial Wavefunction and Orbital Levels of The Hydrogen AtomDocument21 pagesChem 373 - Lecture 17: Radial Wavefunction and Orbital Levels of The Hydrogen AtomNuansak3100% (1)
U3 2b
U3 2b
Uploaded by
yunohu0 ratings0% found this document useful (0 votes)
67 views10 pagesEach electron in an atom is described by four quantum numbers: principal (n), angular momentum (λ), magnetic (m), and electron spin (s). The principal quantum number n determines the electron's energy level and shell (K, L, M, N etc.). The angular momentum quantum number λ indicates the orbital shape (s, p, d, f etc.) and allowed values are 0 to n-1. The magnetic quantum number m indicates the orbital orientation and allowed values are -λ to +λ. The electron spin quantum number s is either +1/2 or -1/2.
Original Description:
yes my dear...
Original Title
U3.2b
Copyright
© © All Rights Reserved
Available Formats
PPT, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentEach electron in an atom is described by four quantum numbers: principal (n), angular momentum (λ), magnetic (m), and electron spin (s). The principal quantum number n determines the electron's energy level and shell (K, L, M, N etc.). The angular momentum quantum number λ indicates the orbital shape (s, p, d, f etc.) and allowed values are 0 to n-1. The magnetic quantum number m indicates the orbital orientation and allowed values are -λ to +λ. The electron spin quantum number s is either +1/2 or -1/2.
Copyright:
© All Rights Reserved
Available Formats
Download as PPT, PDF, TXT or read online from Scribd
Download as ppt, pdf, or txt
0 ratings0% found this document useful (0 votes)
67 views10 pagesU3 2b
U3 2b
Uploaded by
yunohuEach electron in an atom is described by four quantum numbers: principal (n), angular momentum (λ), magnetic (m), and electron spin (s). The principal quantum number n determines the electron's energy level and shell (K, L, M, N etc.). The angular momentum quantum number λ indicates the orbital shape (s, p, d, f etc.) and allowed values are 0 to n-1. The magnetic quantum number m indicates the orbital orientation and allowed values are -λ to +λ. The electron spin quantum number s is either +1/2 or -1/2.
Copyright:
© All Rights Reserved
Available Formats
Download as PPT, PDF, TXT or read online from Scribd
Download as ppt, pdf, or txt
You are on page 1of 10
Each of the electrons in an atom is described and
characterised by a set of four quantum numbers, namely
a) principal quantum number, n
b) angular momentum quantum number,
c) magnetic quantum number, m
d) electron spin quantum number, s.
Quantum Numbers
The value of n determines the energy of an orbital and thereby
the energy of the electron in that particular orbital.
The principal quantum number may have only integral values: n
=1, 2, 3, , .
Principal Quantum Number, n
n 1 2 3 4
shell K L M N
Orbital size
Energy
increases
Angular Momentum Quantum Number,
- Alternative name: Subsidiary / Azimuthal / Orbital
Quantum Number
- The value of indicates the shape of the atomic orbital (AO), the
types of orbitals, and the angular momentum of the electron.
- The allowed values of are 0, 1, 2,, (n1)
Letters are assigned to different numerical values of .
Numerical value of Symbol
0
1
2
3
Orbital shape
s
p
d
f
spherical
dumbbell
cloverleaf
Angular Momentum Quantum Number,
- is dependant on n. (i.e., 0 < n).
If n = 1, can only be 0 (s-orbital)
If n = 2, can be 0 or 1 giving rise to two subshells
(s and p-orbitals) of slightly different energy.
If n = 3, can be 0, 1, or 2 (there are three subshells:
(s, p, and d-orbitals)
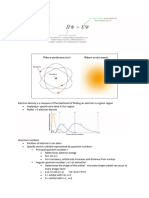
Magnetic Quantum Number, m
The direction or orientation of the magnetic field is determined
by the value of m.
Possible values of m depend on the value of . For a given , m
can be : , , 0, , +
( m +)
If = 0, m can only be 0 one orbital in s-subshell
If = 1, m can be 1, 0, +1 three orbitals in p-subshell
If = 2, m can be 2, 1, 0, +1, +2 five orbitals in d-subshell
The number of m values indicates the number of orbitals in
a subshell with a particular value.
The values of n = 2 and = 1 indicate that we have a 2p-
subshell, and in this subshell we have three 2p-orbitals
(because there are three values of m, given by -1, 0 and
+1)
Magnetic Quantum Number, m
Electron Spin Quantum Number, s
The value of s determines the direction of spinning motions of an
electron (either clockwise or counter clockwise) which is spinning
on its own axes, as Earth does.
The electron spin quantum number has a value of
+
1
2
-
1
2
or
Atomic orbitals with the same energy (i.e., the same value
of n and ) are said to be degenerated. Therefore, there are
(2+1) degenerate orbitals for each value of .
The maximum number of electrons in a particular energy
level, n is given by the expression as follows:
max no. of e
= 2n
2
Points to Remember
Exercise
Shell n
(<n)
Orbital
notation
m
(- m +)
No. of
degenerated
orbitals
K
L
M
0
0
0
1
1
2
1s
2s
3s
2p
3p
3d
0
0
0
-1, 0, +1
-1, 0, +1
-2, -1, 0, +1, +2
1
1
1
3
3
5
2
1
3
Exercise
State whether or not each of the following symbols is an
acceptable designation for an atomic orbital. Explain what
is wrong with the unacceptable symbols.
b) 6g
a) 2d
c) 7s
d) 5i
n=2 =2
( < n)
n=6
n=7
n=5
=4
=0
=6
unacceptable
acceptable
unacceptable
acceptable
You might also like
- Quantum Numbers 2. Azimuthal Quantum Number: (L)Document2 pagesQuantum Numbers 2. Azimuthal Quantum Number: (L)Selva ManiNo ratings yet
- Quantum NumbersDocument39 pagesQuantum NumbersTin SumangaNo ratings yet
- Atomic Structure: 2.2: Quantum MechanicsDocument48 pagesAtomic Structure: 2.2: Quantum MechanicsDaniel LimNo ratings yet
- 202003291623595323nkpandey Quantum NumbersDocument3 pages202003291623595323nkpandey Quantum NumbersBk Creative StudioNo ratings yet
- Orbitals and Quantum NumberDocument21 pagesOrbitals and Quantum NumberPan Kylie KateNo ratings yet
- Quantum Numbers and ECDocument3 pagesQuantum Numbers and ECShraddha Deshmukh-KelkarNo ratings yet
- CHEM 2101 Lecture 2 (Qunatum Number)Document5 pagesCHEM 2101 Lecture 2 (Qunatum Number)Asif UddinNo ratings yet
- Quantum Numbers Quantum NumbersDocument2 pagesQuantum Numbers Quantum Numbersaijaz100No ratings yet
- Quantum NumbersDocument3 pagesQuantum NumbersMia Pearl Tabios ValenzuelaNo ratings yet
- Advancechem Q1 LP4Document7 pagesAdvancechem Q1 LP4Francesca BuenoNo ratings yet
- AtomsFirst2e Day6 Sec3.7-3.8 CHMY171 Fall 2015Document17 pagesAtomsFirst2e Day6 Sec3.7-3.8 CHMY171 Fall 2015Xenia Mae FloresNo ratings yet
- Quantum NumbersDocument12 pagesQuantum NumbersSoub kuopNo ratings yet
- Atomic and Molecular Struc.Document25 pagesAtomic and Molecular Struc.lelouchvibritannia0502No ratings yet
- L3 Che101Document28 pagesL3 Che101Musa Ahammed MahinNo ratings yet
- AtomsFirst2e Day6 Sec3.7-3.8 CHMY171 Fall 2015Document17 pagesAtomsFirst2e Day6 Sec3.7-3.8 CHMY171 Fall 2015Claudius MorpheusNo ratings yet
- Quantum Numbers: Are Required To Describe The Distribution ofDocument17 pagesQuantum Numbers: Are Required To Describe The Distribution ofIrfan Sayeem SultanNo ratings yet
- q2 General Chemistry Rev (Complete)Document14 pagesq2 General Chemistry Rev (Complete)rosedancel52No ratings yet
- Atomic Orbitals: Quantum NumbersDocument16 pagesAtomic Orbitals: Quantum NumberslostgirlNo ratings yet
- 1A The Shapes and Structures of Molecules Student Handout Part Two 2022.23Document70 pages1A The Shapes and Structures of Molecules Student Handout Part Two 2022.23Music MaestroNo ratings yet
- Atomic OrbitalDocument25 pagesAtomic OrbitalCarolle PerezNo ratings yet
- Quantum Numbers and Electron ConfigurationsDocument57 pagesQuantum Numbers and Electron Configurationscath100% (1)
- How To Use Quantum Numbers To Describe AnDocument11 pagesHow To Use Quantum Numbers To Describe AnAntonio Exal ColladoNo ratings yet
- Atomic StructureDocument19 pagesAtomic StructureJhoanna Marie Manuel-AbelNo ratings yet
- Quantum Numbers and Electron Configuration.: ST ND RD THDocument2 pagesQuantum Numbers and Electron Configuration.: ST ND RD THKelly Gabriel Singuay OcceñoNo ratings yet
- Quantum NumbersDocument8 pagesQuantum NumbersMoises espinozaNo ratings yet
- 1 Quantum Numbers-1Document34 pages1 Quantum Numbers-1Carlo James SablanNo ratings yet
- Electronic Structure of MatterDocument3 pagesElectronic Structure of Matterilikegay2dmensNo ratings yet
- Interpretation of Quantum Numbers: TU Bergakademie FreibergDocument2 pagesInterpretation of Quantum Numbers: TU Bergakademie FreibergPraveenNo ratings yet
- Quantum Numbers and Atomic OrbitalsDocument16 pagesQuantum Numbers and Atomic OrbitalsR-jayVenturilloNo ratings yet
- Chemistry - WEEK 3 - LAS (Quantum Numbers)Document5 pagesChemistry - WEEK 3 - LAS (Quantum Numbers)EZRA THERESE DE JESUSNo ratings yet
- Rhael Denielle P. Sunio 12 - Archimedes General Chemistry 1 - Module 1Document6 pagesRhael Denielle P. Sunio 12 - Archimedes General Chemistry 1 - Module 1Leah SunioNo ratings yet
- Quantum Numbers and Electron ConfigurationsDocument16 pagesQuantum Numbers and Electron ConfigurationsMARVIN OTHELLO MELCHOR DAUISNo ratings yet
- AD SirDocument23 pagesAD Sirlost dharNo ratings yet
- De Broglie Relation: EinsteinDocument19 pagesDe Broglie Relation: EinsteinMaisha MaimunaNo ratings yet
- Chemistry Notes Part 1Document59 pagesChemistry Notes Part 1SharleneNo ratings yet
- P11 ADocument9 pagesP11 ADana CapbunNo ratings yet
- DME I Year CHEDocument91 pagesDME I Year CHERadha KrishnaNo ratings yet
- Structure of AtomDocument29 pagesStructure of AtomAbhisek DehuriNo ratings yet
- Quantum Numbers Structure 2Document22 pagesQuantum Numbers Structure 2vishalsingh1997No ratings yet
- Chemistry Quantum NumbersDocument9 pagesChemistry Quantum NumbersSamiksha SainiNo ratings yet
- Unit - IV - Uv VisibleDocument27 pagesUnit - IV - Uv VisibleM. BalakrishnanNo ratings yet
- General Chemistry 1 M2W2Document7 pagesGeneral Chemistry 1 M2W2Warley JabelNo ratings yet
- Lecture 2 Part 2Document40 pagesLecture 2 Part 2ariannemariebaronNo ratings yet
- Atomic StructureDocument2 pagesAtomic StructureNarjis FatimaNo ratings yet
- Lecture 4 Wavefunction NewDocument53 pagesLecture 4 Wavefunction NewkedirNo ratings yet
- Quantum NumbersDocument15 pagesQuantum NumbersMohamed KilanyNo ratings yet
- Chapter 2 - Structure of Atom PeridiocityDocument97 pagesChapter 2 - Structure of Atom PeridiocityAina AthirahNo ratings yet
- CEM1008F - 3. The Periodic Table and Atomic Structure 2024 - Lecture - NotesDocument41 pagesCEM1008F - 3. The Periodic Table and Atomic Structure 2024 - Lecture - Notesparlypalesa90No ratings yet
- Exercises No. 2Document6 pagesExercises No. 2John Miguel BarbaNo ratings yet
- The Electronic Structure of AtomsDocument54 pagesThe Electronic Structure of AtomsElijah PunzalanNo ratings yet
- 2.2 Quantum Mechanis ModelDocument53 pages2.2 Quantum Mechanis ModelAbdullah AhmadNo ratings yet
- (L-10) - (JEE 3.0) - Atomic Structure - 11th May.Document57 pages(L-10) - (JEE 3.0) - Atomic Structure - 11th May.Thomas ShelbyNo ratings yet
- Quantum Numbers and Their Physical InterpetationDocument15 pagesQuantum Numbers and Their Physical InterpetationMomina LaraibNo ratings yet
- Schroedinger Equation Atomic Wave Functions Atomic Orbitals Quantum NumbersDocument45 pagesSchroedinger Equation Atomic Wave Functions Atomic Orbitals Quantum NumbersRishit JakhariaNo ratings yet
- Quantum Numbers For AtomsDocument6 pagesQuantum Numbers For AtomsMaricae NathanielNo ratings yet
- CHM131 - Chapter 2 - Structure of Atom - PeriodicityDocument97 pagesCHM131 - Chapter 2 - Structure of Atom - PeriodicityLeo PietroNo ratings yet
- Grade 9 SCIENCE (JAN. 18-22)Document3 pagesGrade 9 SCIENCE (JAN. 18-22)Jhev LeopandoNo ratings yet
- Lecture 2Document18 pagesLecture 2Ayan PatelNo ratings yet
- Quantum Numbers PowerpointDocument12 pagesQuantum Numbers Powerpointapi-201447595No ratings yet
- Electron Beam-Specimen Interactions and Simulation Methods in MicroscopyFrom EverandElectron Beam-Specimen Interactions and Simulation Methods in MicroscopyNo ratings yet
- Shape of Atomic OrbitalsDocument5 pagesShape of Atomic OrbitalsyunohuNo ratings yet
- The Anomalous Electronic Configurations of CR and CuDocument6 pagesThe Anomalous Electronic Configurations of CR and CuyunohuNo ratings yet
- Representing Electronic Configuration: Orbital DiagramDocument1 pageRepresenting Electronic Configuration: Orbital DiagramyunohuNo ratings yet
- Atomic OrbitalDocument1 pageAtomic OrbitalyunohuNo ratings yet
- MATTER (1.2 Mole Concept)Document101 pagesMATTER (1.2 Mole Concept)yunohuNo ratings yet
- C5.population GeneticsDocument28 pagesC5.population GeneticsyunohuNo ratings yet
- Sambungan P-N Dan Dioda Sambungan P-NDocument27 pagesSambungan P-N Dan Dioda Sambungan P-NUlfie Kusuma Wardhani100% (1)
- MAMALUBA - Assignment For Chem Lab 401Document2 pagesMAMALUBA - Assignment For Chem Lab 401Sophia Indirah MamalubaNo ratings yet
- Quantum Mechanics Course ZeemansplittingDocument29 pagesQuantum Mechanics Course ZeemansplittingjlbalbNo ratings yet
- Balancing Equations: Practice ProblemsDocument10 pagesBalancing Equations: Practice ProblemsMoriel Barrogo BequilloNo ratings yet
- Balancing Chemical EquationsDocument18 pagesBalancing Chemical EquationsWendye YimamNo ratings yet
- Daftar Bahan Kimia PaDocument4 pagesDaftar Bahan Kimia Papanda_oonNo ratings yet
- Salt AnalysisDocument15 pagesSalt AnalysisPrabhanshu Pandey (Chotu)No ratings yet
- Properties of Silver (I), Mercury (I) and Lead (II) SaltsDocument7 pagesProperties of Silver (I), Mercury (I) and Lead (II) SaltsnothaboNo ratings yet
- Born-Haber Cycle Born-Haber Cycle: Why Not Nacl ? Why MGCL ?Document1 pageBorn-Haber Cycle Born-Haber Cycle: Why Not Nacl ? Why MGCL ?Fara FransiskaNo ratings yet
- DFT PresentationDocument63 pagesDFT PresentationhammmoudaNo ratings yet
- Chemistry: Form 4-Chapter 8Document7 pagesChemistry: Form 4-Chapter 8Azrel YusoffNo ratings yet
- 2-Synthetic Methods Chap 2Document81 pages2-Synthetic Methods Chap 2MahsaNo ratings yet
- Intrinsic & Extrinsic SemiconductorsDocument20 pagesIntrinsic & Extrinsic SemiconductorsmaharjansumirNo ratings yet
- MCQ SemiconductorDocument12 pagesMCQ Semiconductormuktadir hosenNo ratings yet
- Introduction To Material Chem.Document23 pagesIntroduction To Material Chem.Gaurav MauryaNo ratings yet
- Assignment - EEE 103Document4 pagesAssignment - EEE 103Abu BasharNo ratings yet
- Section 18B Metric Salt TablesDocument22 pagesSection 18B Metric Salt TablesLazharNo ratings yet
- Transport in Graphene: Klaus RichterDocument50 pagesTransport in Graphene: Klaus RichterAnonymous 9rJe2lOskxNo ratings yet
- P Block Reactions PDFDocument3 pagesP Block Reactions PDFTarundeepNo ratings yet
- Microwave Mediated Green Synthesis of Copper Nanoparticles Using Aqueous Extract of Seeds and Particles CharacterisationDocument12 pagesMicrowave Mediated Green Synthesis of Copper Nanoparticles Using Aqueous Extract of Seeds and Particles Characterisationmiguel salasNo ratings yet
- Brine CalculatorDocument2 pagesBrine CalculatorzachNo ratings yet
- Experiment No. 1: Aim: To Identify The Cation and Anion in Copper Sulphate PentahydrateDocument3 pagesExperiment No. 1: Aim: To Identify The Cation and Anion in Copper Sulphate PentahydrateElite GamingNo ratings yet
- Chem 373 - Lecture 27: Homonuclear Diatomic Molecules-IIDocument22 pagesChem 373 - Lecture 27: Homonuclear Diatomic Molecules-IINuansak3No ratings yet
- Chapter 18: Electrical Properties II: Intrinsic & Extrinsic SemiconductorsDocument20 pagesChapter 18: Electrical Properties II: Intrinsic & Extrinsic SemiconductorsManish KumarNo ratings yet
- Epitaxy GrowthDocument24 pagesEpitaxy GrowthMD. MEHEDI HASAN100% (3)
- DatasheetDocument87 pagesDatasheetmdelcos100% (1)
- DiodeDocument19 pagesDiodeRamu kakaNo ratings yet
- 3.3 IV Characteristics of PN Junction Diode-ModifiedDocument18 pages3.3 IV Characteristics of PN Junction Diode-Modifiedraunak guptaNo ratings yet
- Chem 373 - Lecture 17: Radial Wavefunction and Orbital Levels of The Hydrogen AtomDocument21 pagesChem 373 - Lecture 17: Radial Wavefunction and Orbital Levels of The Hydrogen AtomNuansak3100% (1)