Professional Documents
Culture Documents
Activity (A) : A F/ F Activity Coefficient
Activity (A) : A F/ F Activity Coefficient
Uploaded by
PRAVIN0 ratings0% found this document useful (0 votes)
5 views3 pagesThis document defines activity as the ratio of the fugacity of a component in a real solution to its fugacity in an ideal solution. It also discusses activity coefficients, which measure how much real solutions deviate from ideality, and lists several common equations used to model activity coefficients including Wohl's three suffix, Margules, and Wilson equations. Finally, it lists methods for consistency testing of vapor-liquid equilibrium data such as using the slope of lnγ curves or the Redlich-Kister method.
Original Description:
Original Title
Vapour.pptx
Copyright
© © All Rights Reserved
Available Formats
PPTX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document defines activity as the ratio of the fugacity of a component in a real solution to its fugacity in an ideal solution. It also discusses activity coefficients, which measure how much real solutions deviate from ideality, and lists several common equations used to model activity coefficients including Wohl's three suffix, Margules, and Wilson equations. Finally, it lists methods for consistency testing of vapor-liquid equilibrium data such as using the slope of lnγ curves or the Redlich-Kister method.
Copyright:
© All Rights Reserved
Available Formats
Download as PPTX, PDF, TXT or read online from Scribd
Download as pptx, pdf, or txt
0 ratings0% found this document useful (0 votes)
5 views3 pagesActivity (A) : A F/ F Activity Coefficient
Activity (A) : A F/ F Activity Coefficient
Uploaded by
PRAVINThis document defines activity as the ratio of the fugacity of a component in a real solution to its fugacity in an ideal solution. It also discusses activity coefficients, which measure how much real solutions deviate from ideality, and lists several common equations used to model activity coefficients including Wohl's three suffix, Margules, and Wilson equations. Finally, it lists methods for consistency testing of vapor-liquid equilibrium data such as using the slope of lnγ curves or the Redlich-Kister method.
Copyright:
© All Rights Reserved
Available Formats
Download as PPTX, PDF, TXT or read online from Scribd
Download as pptx, pdf, or txt
You are on page 1of 3
Activity(a):
It is defined as the fugacity of the existing condition
to the standard state fugacity
a=f/ fo
Activity coefficient :
It measure the extent to which real solution depart from
ideality.
ln=bx
ln=bx
Activity coefficient equations
Wohls three suffix equation
Margules equation
Van laar equation
Wilson equation
Non random two liquid (NRTL)equation
Universal quasi chemical (UNIQUAC)equation
Universal functional activity coefficient (UNIFAC)
method
Consistency test for VLE data
Using slope of ln curves
Using data at mid point
Redlich kister method
Using the coexistence equation
Using partial pressure data
You might also like
- UniquacDocument19 pagesUniquacAjeeth Prabhu100% (1)
- CE361 Lab - 5 Instructions+worksheetDocument4 pagesCE361 Lab - 5 Instructions+worksheetZainab FarooqNo ratings yet
- Differential DistillationDocument36 pagesDifferential DistillationShruti100% (1)
- Grade 9 - Chemistry - Ionic BondingDocument23 pagesGrade 9 - Chemistry - Ionic BondingPathmanathan NadesonNo ratings yet
- GPA 2145-16 Table of Physical Properties of HC and Other Compounds of Interest To The NGA and NGLDocument64 pagesGPA 2145-16 Table of Physical Properties of HC and Other Compounds of Interest To The NGA and NGLAplena Sukan100% (2)
- Vapour LiquidDocument5 pagesVapour LiquidPRAVINNo ratings yet
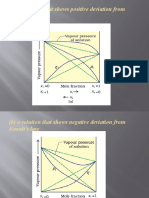
- (A) A Solution That Shows Positive Deviation From Raoult's LawDocument7 pages(A) A Solution That Shows Positive Deviation From Raoult's LawPRAVINNo ratings yet
- Effect of Pressure On VLE The High Pressure Diagrams Are Above Low PressureDocument7 pagesEffect of Pressure On VLE The High Pressure Diagrams Are Above Low PressurePRAVINNo ratings yet
- PokjhgvcDocument14 pagesPokjhgvcPRAVINNo ratings yet
- Composition in Gaseous Phase:: y P X P (I 1,2,3 ..N)Document8 pagesComposition in Gaseous Phase:: y P X P (I 1,2,3 ..N)PRAVINNo ratings yet
- Yip=Xiγi P (I= 1,2,…..,N) : Where Γi = Activity CoefficientDocument7 pagesYip=Xiγi P (I= 1,2,…..,N) : Where Γi = Activity CoefficientPRAVINNo ratings yet
- UygfcDocument10 pagesUygfcPRAVINNo ratings yet
- Where, P - Total Pressure P, P - Partial Pressure of ComponentDocument9 pagesWhere, P - Total Pressure P, P - Partial Pressure of ComponentPRAVINNo ratings yet
- Vap EquilibriumDocument14 pagesVap EquilibriumPRAVINNo ratings yet
- Vapour Liquid EquilibriumDocument15 pagesVapour Liquid EquilibriumPRAVINNo ratings yet
- TFDXZDocument14 pagesTFDXZPRAVINNo ratings yet
- Vle 4Document15 pagesVle 4PRAVINNo ratings yet
- Calculation of Vapor-Liquid Equilibria From Infinite-Dilution Excess Enthalpy Data Using The Wilson or NRTL EquationDocument12 pagesCalculation of Vapor-Liquid Equilibria From Infinite-Dilution Excess Enthalpy Data Using The Wilson or NRTL EquationneftNo ratings yet
- Liquid Liquid Column Drop: Spray Holdup, and Continuous Phase Mass TransferDocument6 pagesLiquid Liquid Column Drop: Spray Holdup, and Continuous Phase Mass TransferBastien DautricheNo ratings yet
- Activity CoefficientsDocument25 pagesActivity CoefficientsRajeev ReddyNo ratings yet
- Chemical Engineering Thermodynamics Project-I: TopicDocument11 pagesChemical Engineering Thermodynamics Project-I: TopicRohit GuptaNo ratings yet
- 49aiaa 51 AmjadDocument16 pages49aiaa 51 AmjadMuhammad IshaqNo ratings yet
- Controlling Turbulence in The Complex Ginzburg-Landau Equation - 2Document12 pagesControlling Turbulence in The Complex Ginzburg-Landau Equation - 2chenyeyuge94No ratings yet
- 7 Chapter 7: Non-Linear Thermodynamics of Irreversible ProcessesDocument13 pages7 Chapter 7: Non-Linear Thermodynamics of Irreversible ProcessesBARBOSA RAFFAELLINo ratings yet
- Accepted Manuscript: Chemical Engineering ScienceDocument36 pagesAccepted Manuscript: Chemical Engineering ScienceDiego JesusNo ratings yet
- The System Carbon Dioxide-Water and The Krichevsky-Kasarnovsky EquationDocument15 pagesThe System Carbon Dioxide-Water and The Krichevsky-Kasarnovsky EquationTiên PhạmNo ratings yet
- 1971 - Mass Transfer With Chemical Reaction From Spherical One or Two Component Bubbles or DropsDocument22 pages1971 - Mass Transfer With Chemical Reaction From Spherical One or Two Component Bubbles or DropsCristiane MesquitaNo ratings yet
- Fe Chemical EngineeringDocument5 pagesFe Chemical EngineeringJudith LugoNo ratings yet
- Lyapunov Function - Dynamical SystemsDocument4 pagesLyapunov Function - Dynamical SystemsmCmAlNo ratings yet
- Scribd G GGGGG GGGGG GGGGGDocument8 pagesScribd G GGGGG GGGGG GGGGGR Carlos HCNo ratings yet
- Metodos AnaliticosDocument136 pagesMetodos AnaliticosCarlos Velando CabañasNo ratings yet
- Logistic Regression: Logistic Regression and The New: Residual Logistic RegressionDocument31 pagesLogistic Regression: Logistic Regression and The New: Residual Logistic RegressionAfrian Addlu MatinNo ratings yet
- Integration of The Synthetic Dynamics Power Flow Equations With The Optimal MultiplierDocument5 pagesIntegration of The Synthetic Dynamics Power Flow Equations With The Optimal MultiplierMarchena Segil ErikNo ratings yet
- Spreadsheet Distillation PDFDocument8 pagesSpreadsheet Distillation PDFArunkumarNo ratings yet
- Definitions Topic 5.1.1 How Fast OCR A Chemistry A LevelDocument2 pagesDefinitions Topic 5.1.1 How Fast OCR A Chemistry A LevelCHRONIKNo ratings yet
- Smith - 8th CH 13 Thermodynamic Formulations For Vapor Liquid EquilibriumDocument59 pagesSmith - 8th CH 13 Thermodynamic Formulations For Vapor Liquid Equilibriumjeongyun271No ratings yet
- CH 6 The 2 K Factorial DesignDocument56 pagesCH 6 The 2 K Factorial Designdaimon_pNo ratings yet
- Intro To Computational Fluid Dynamics: Brandon Lloyd COMP 259 April 16, 2003Document32 pagesIntro To Computational Fluid Dynamics: Brandon Lloyd COMP 259 April 16, 2003Vedant DubeyNo ratings yet
- Gloss PDFDocument8 pagesGloss PDFSaadat Ullah KhanNo ratings yet
- Rayleigh PDFDocument36 pagesRayleigh PDFJomhel CalluengNo ratings yet
- Simoni Fpe2007Document37 pagesSimoni Fpe2007Hassan BadriNo ratings yet
- Kumaraswamy Ponnambalam and Ravi M. Phatarfod: Journal of Water Management ModelingDocument8 pagesKumaraswamy Ponnambalam and Ravi M. Phatarfod: Journal of Water Management ModelingJosimar AlvesNo ratings yet
- 07-Absorption For HAP and VOCcontrolDocument118 pages07-Absorption For HAP and VOCcontrolTakeshi Tanohuye TanohuyeNo ratings yet
- Chapter 4Document23 pagesChapter 4syedmuhammadtariqueNo ratings yet
- Basics of Probability TheoryDocument22 pagesBasics of Probability TheoryersukhmeetkaurNo ratings yet
- Extension of Peng-Robinson For Complex MixturesDocument18 pagesExtension of Peng-Robinson For Complex MixturesMandy NelsonNo ratings yet
- Hudon - STABILIZATION - OF - CHEMICAL - REACTORS - USING - APPROXIMADocument7 pagesHudon - STABILIZATION - OF - CHEMICAL - REACTORS - USING - APPROXIMAWarrenNo ratings yet
- Application of Simple Fixed PointDocument11 pagesApplication of Simple Fixed PointRhea MambaNo ratings yet
- Cmpe107 NotesDocument14 pagesCmpe107 NotesjerendarNo ratings yet
- Lecture 2: Thermodynamics and Equilibrium: D RTDLNFDocument2 pagesLecture 2: Thermodynamics and Equilibrium: D RTDLNFRinaldi SaputraNo ratings yet
- Fourth Chapter - Part 1-1Document25 pagesFourth Chapter - Part 1-1toslim jahidNo ratings yet
- 3G4 Flash CalculationsDocument9 pages3G4 Flash CalculationsRafael Reyes0% (1)
- Delta Excited Nuclear Matter in DerivatiDocument10 pagesDelta Excited Nuclear Matter in Derivatimfgobbi4515No ratings yet
- Analysis and Design of Pharmacokinetic ModelsDocument77 pagesAnalysis and Design of Pharmacokinetic ModelsYuppie RajNo ratings yet
- Lo2001 - Testing The Number of Components in A Normal MixtureDocument12 pagesLo2001 - Testing The Number of Components in A Normal MixtureYingChun PatNo ratings yet
- Fulltext 7056Document36 pagesFulltext 7056gabesh3192No ratings yet
- How To Solve A ProblemDocument20 pagesHow To Solve A ProblemnacachimbandiNo ratings yet
- Chemical Kinetics LECTUREDocument36 pagesChemical Kinetics LECTUREMaame DurowaaNo ratings yet
- Group-Contribution Estimation of Activity Coefficients in Nonideal Liquid Mixtures. Aiche 1975Document14 pagesGroup-Contribution Estimation of Activity Coefficients in Nonideal Liquid Mixtures. Aiche 1975Cristina ACNo ratings yet
- Schmid2011 Article ApplicationOfTheDynamicModeDecDocument8 pagesSchmid2011 Article ApplicationOfTheDynamicModeDecJules JoeNo ratings yet
- Where, P - Total Pressure P, P - Partial Pressure of ComponentDocument3 pagesWhere, P - Total Pressure P, P - Partial Pressure of ComponentPRAVINNo ratings yet
- Composition in Gaseous Phase:: y P X P (I 1,2,3 ..N)Document8 pagesComposition in Gaseous Phase:: y P X P (I 1,2,3 ..N)PRAVINNo ratings yet
- Yip=Xiγip (I= 1,2,…..,N) : Where Γi = Activity CoefficientDocument2 pagesYip=Xiγip (I= 1,2,…..,N) : Where Γi = Activity CoefficientPRAVINNo ratings yet
- OIUHGFDXZDocument5 pagesOIUHGFDXZPRAVINNo ratings yet
- UygfcDocument10 pagesUygfcPRAVINNo ratings yet
- Effect of Pressure On VLE The High Pressure Diagrams Are Above Low PressureDocument7 pagesEffect of Pressure On VLE The High Pressure Diagrams Are Above Low PressurePRAVINNo ratings yet
- Vapiquid IbriumDocument15 pagesVapiquid IbriumPRAVINNo ratings yet
- Where, P - Total Pressure P, P - Partial Pressure of ComponentDocument9 pagesWhere, P - Total Pressure P, P - Partial Pressure of ComponentPRAVINNo ratings yet
- Vapiquid IbriumDocument15 pagesVapiquid IbriumPRAVINNo ratings yet
- TFDXZDocument14 pagesTFDXZPRAVINNo ratings yet
- Activity (A) :: It Is Defined As The Fugacity of The Existing Condition To The Standard State FugacityDocument4 pagesActivity (A) :: It Is Defined As The Fugacity of The Existing Condition To The Standard State FugacityPRAVINNo ratings yet
- Activity (A) :: It Is Defined As The Fugacity of The Existing Condition To The Standard State FugacityDocument4 pagesActivity (A) :: It Is Defined As The Fugacity of The Existing Condition To The Standard State FugacityPRAVINNo ratings yet
- PokjhgvcDocument14 pagesPokjhgvcPRAVINNo ratings yet
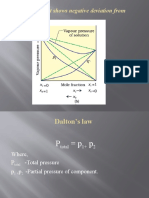
- Dalton's Law: Where, P - Total Pressure P, P - Partial Pressure of ComponentDocument12 pagesDalton's Law: Where, P - Total Pressure P, P - Partial Pressure of ComponentPRAVINNo ratings yet
- Vapour LiquidDocument5 pagesVapour LiquidPRAVINNo ratings yet
- Vap EquilibriumDocument14 pagesVap EquilibriumPRAVINNo ratings yet
- (A) A Solution That Shows Positive Deviation From Raoult's LawDocument7 pages(A) A Solution That Shows Positive Deviation From Raoult's LawPRAVINNo ratings yet
- Yip=Xiγi P (I= 1,2,…..,N) : Where Γi = Activity CoefficientDocument7 pagesYip=Xiγi P (I= 1,2,…..,N) : Where Γi = Activity CoefficientPRAVINNo ratings yet
- Vapour Liquid EquilibriumDocument11 pagesVapour Liquid EquilibriumPRAVINNo ratings yet
- Vle 9Document10 pagesVle 9PRAVINNo ratings yet
- What Is Equilibrium?Document12 pagesWhat Is Equilibrium?PRAVINNo ratings yet
- (B) A Solution That Shows Negative Deviation From Raoult's LawDocument9 pages(B) A Solution That Shows Negative Deviation From Raoult's LawPRAVINNo ratings yet
- Vapour Liquid EquilibriumDocument15 pagesVapour Liquid EquilibriumPRAVINNo ratings yet
- Vle 4Document15 pagesVle 4PRAVINNo ratings yet
- D3E801B9571-Air Conditioning System With Refrigerant R134aDocument115 pagesD3E801B9571-Air Conditioning System With Refrigerant R134aDaniel PelteaNo ratings yet
- B.SC New - 20192110073946 PDFDocument106 pagesB.SC New - 20192110073946 PDFDivyanshu ShekharNo ratings yet
- Exothermic and Endothermic Reactions #1111Document5 pagesExothermic and Endothermic Reactions #1111SubatomoNo ratings yet
- Electron Configuration and Chemical Periodicity: Dr. Wolf's CHM 101Document42 pagesElectron Configuration and Chemical Periodicity: Dr. Wolf's CHM 101natebarrett11No ratings yet
- Acid Base and Salt: 1. Objective QuestionsDocument8 pagesAcid Base and Salt: 1. Objective QuestionsKabir MaheshwariNo ratings yet
- Photoenolization-Induced Oxirane Ring Opening in 2,5-Dimethylbenzoyl Oxiranes To Form Pharmaceutically Promising Indanone DerivativesDocument10 pagesPhotoenolization-Induced Oxirane Ring Opening in 2,5-Dimethylbenzoyl Oxiranes To Form Pharmaceutically Promising Indanone DerivativesDiogomussumNo ratings yet
- Salt PreparationDocument4 pagesSalt PreparationAmna Yousaf100% (1)
- Atoms and Elements: ObjectivesDocument5 pagesAtoms and Elements: ObjectivesAngel RingorNo ratings yet
- The Circulation Water and Steam in Water-Tube Boilers, and The Rational Simplification of Boiler DesignDocument35 pagesThe Circulation Water and Steam in Water-Tube Boilers, and The Rational Simplification of Boiler DesignAbdul Ghafoor BhattiNo ratings yet
- MANUAL OPERACIÓN InRoomDocument84 pagesMANUAL OPERACIÓN InRoomtecnofresh.serviciosNo ratings yet
- Plasma Physics HomeworkDocument4 pagesPlasma Physics Homeworkafnadyebqjzazl100% (1)
- Medical Gases SupplyDocument6 pagesMedical Gases SupplyManuela PascalNo ratings yet
- Cambridge O Level: CHEMISTRY 5070/21Document20 pagesCambridge O Level: CHEMISTRY 5070/21Fasih RazaNo ratings yet
- Electrochemistry (Galvanic Cells)Document1 pageElectrochemistry (Galvanic Cells)julianalelis1234No ratings yet
- Icse Class X Chemical BondingDocument3 pagesIcse Class X Chemical BondingKush kankariya KankariyaNo ratings yet
- Cambridge Secondary Checkpoint - Science (1113) October 2018 Paper 2 Mark SchemeDocument11 pagesCambridge Secondary Checkpoint - Science (1113) October 2018 Paper 2 Mark SchemeMilcah Tesfaye100% (2)
- Test 1 PhysChem - May 2022 - W - AnsDocument5 pagesTest 1 PhysChem - May 2022 - W - Ansmarc jacobs davisNo ratings yet
- Nuclear ModelsDocument55 pagesNuclear Modelsdevika VNo ratings yet
- Modeling and Simulation of A Hydrocracking UnitDocument18 pagesModeling and Simulation of A Hydrocracking UnitAnnu RawatNo ratings yet
- Properties of Matter Key Term ReviewDocument1 pageProperties of Matter Key Term ReviewRishikesh HooliNo ratings yet
- Charles LawDocument3 pagesCharles LawJohn Eliandrei Bañaga IINo ratings yet
- REPORTDocument31 pagesREPORTUnique Boss50% (2)
- RAC AssignmentDocument4 pagesRAC Assignmentreghurahul88No ratings yet
- Spontaneously Not Vice Versa: Second-Law - PPT Modified 10/9/02Document37 pagesSpontaneously Not Vice Versa: Second-Law - PPT Modified 10/9/02T Hari PrasadNo ratings yet
- 11.reheat CycleDocument12 pages11.reheat CycleAarvin GeorgeNo ratings yet
- Physics of SuperconductivityDocument106 pagesPhysics of SuperconductivityRaj JanaNo ratings yet
- Assignment I SolubilityDocument24 pagesAssignment I Solubilityabthapa100% (1)
- Structural Survey of Carbonate-Containing Antacids: J. Serna LDocument4 pagesStructural Survey of Carbonate-Containing Antacids: J. Serna LJcNo ratings yet