Professional Documents
Culture Documents
Edta +ZN (Zn-Edta) : Endpoint Initial
Edta +ZN (Zn-Edta) : Endpoint Initial
Uploaded by
jayakumar0 ratings0% found this document useful (0 votes)
16 views3 pagesEDTA forms a complex with zinc ions, where zinc bonds to EDTA through six coordination sites to form [Zn-EDTA]2-. The reaction uses an ammonia buffer at pH ~10.5 as the indicator Eriochrome Black T only changes color in the pH range of 7-11.
Original Description:
Estimation of Zinc
Original Title
Estimation of Zn
Copyright
© © All Rights Reserved
Available Formats
PPTX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentEDTA forms a complex with zinc ions, where zinc bonds to EDTA through six coordination sites to form [Zn-EDTA]2-. The reaction uses an ammonia buffer at pH ~10.5 as the indicator Eriochrome Black T only changes color in the pH range of 7-11.
Copyright:
© All Rights Reserved
Available Formats
Download as PPTX, PDF, TXT or read online from Scribd
Download as pptx, pdf, or txt
0 ratings0% found this document useful (0 votes)
16 views3 pagesEdta +ZN (Zn-Edta) : Endpoint Initial
Edta +ZN (Zn-Edta) : Endpoint Initial
Uploaded by
jayakumarEDTA forms a complex with zinc ions, where zinc bonds to EDTA through six coordination sites to form [Zn-EDTA]2-. The reaction uses an ammonia buffer at pH ~10.5 as the indicator Eriochrome Black T only changes color in the pH range of 7-11.
Copyright:
© All Rights Reserved
Available Formats
Download as PPTX, PDF, TXT or read online from Scribd
Download as pptx, pdf, or txt
You are on page 1of 3
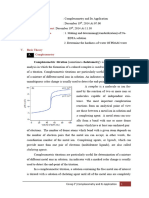
EDTA 4- +Zn [Zn-EDTA]2-
Endpoint Initial
EBT
Ethylene diaminetetraacetic acid, EDTA is a large molecule
which creates a complex with a metal ion, bonding through
six coordination sites.
The ammonia buffer (pH ~ 10.5) used here is needed as
Eriochrome Black T only changes colour in the pH range
7 – 11
You might also like
- Determination of Aluminium by Back TitrationDocument5 pagesDetermination of Aluminium by Back TitrationKojo Eghan75% (12)
- EdtaDocument13 pagesEdtaChongZY100% (1)
- Complexometric TitrationDocument30 pagesComplexometric TitrationSarthakAggarwalNo ratings yet
- Quantitative Determination of Total Hardness in Drinking Water by Complexometric EDTA TitrationDocument14 pagesQuantitative Determination of Total Hardness in Drinking Water by Complexometric EDTA Titrationabcd efgNo ratings yet
- Complexometric Titration 1Document14 pagesComplexometric Titration 1Girma Selale0% (1)
- Chapter 12Document35 pagesChapter 12Jolina PagulayanNo ratings yet
- Complexed Formation TitrationDocument4 pagesComplexed Formation TitrationQUENNIE BRIONESNo ratings yet
- EDTA Titrations: Metal Chelate ComplexesDocument35 pagesEDTA Titrations: Metal Chelate ComplexesJenny LlanesNo ratings yet
- 05 - Chapter 1 PDFDocument57 pages05 - Chapter 1 PDFravisankarNo ratings yet
- 20CH3050 Expt No 1EDTA TitrationsDocument28 pages20CH3050 Expt No 1EDTA TitrationsParameswari ArunNo ratings yet
- EDTA 3 pdf-4Document3 pagesEDTA 3 pdf-4Karan RavalNo ratings yet
- Complexometric TitrationDocument12 pagesComplexometric TitrationBwhzad HameedNo ratings yet
- EDTA-A Versatile Chelating AgentDocument14 pagesEDTA-A Versatile Chelating AgentMirza IjazNo ratings yet
- Complexometry Determination Hardness ofDocument30 pagesComplexometry Determination Hardness ofMichael CarloNo ratings yet
- Practicum 8 Complexometric Titration Experiment Summary: ObjectivesDocument4 pagesPracticum 8 Complexometric Titration Experiment Summary: ObjectivesMia RismaliaNo ratings yet
- Complexation Titration Department of Physical Sciences & Technology University of Sabaragamuwa Srilanka (Document33 pagesComplexation Titration Department of Physical Sciences & Technology University of Sabaragamuwa Srilanka (eswarNo ratings yet
- CompleximetryDocument29 pagesCompleximetrychamp delacruzNo ratings yet
- EDTADocument29 pagesEDTA5P4RT4NII7No ratings yet
- EdtaDocument2 pagesEdtaAnas IlhamNo ratings yet
- Experiment 2: Complexometric Titration 1.0 ObjectivesDocument4 pagesExperiment 2: Complexometric Titration 1.0 ObjectivesSangetha ChelladoraiNo ratings yet
- Course Name: Volumetric and Gravimetric Analytical Chemistry Course Code: 4022133-3Document26 pagesCourse Name: Volumetric and Gravimetric Analytical Chemistry Course Code: 4022133-3faycalfaidiNo ratings yet
- Complexationtitration KDBDocument31 pagesComplexationtitration KDBKiranNo ratings yet
- Experiment: Determination of The Total Hardness (Permanent and Temporary)Document1 pageExperiment: Determination of The Total Hardness (Permanent and Temporary)Subhash DhungelNo ratings yet
- Coordination Assignment 2Document6 pagesCoordination Assignment 2sara noorNo ratings yet
- Complexometric Titrations - PPT 1Document26 pagesComplexometric Titrations - PPT 1Khairi Mustafa Salem67% (3)
- Titrari EDTADocument35 pagesTitrari EDTAAdrian BlidarNo ratings yet
- Complexometric TitrationDocument64 pagesComplexometric TitrationToyeba RahiNo ratings yet
- Complexiometric TitrationsDocument4 pagesComplexiometric Titrationskasonde.musonda8No ratings yet
- Complexmetric TitrationDocument17 pagesComplexmetric TitrationAnonymous oC3F7cxlLH100% (1)
- Complexometry 7199 1Document9 pagesComplexometry 7199 1Admiral General AmanNo ratings yet
- EDTA Titration CurveDocument14 pagesEDTA Titration CurveM'maay WrtyNo ratings yet
- Scott A. Sinex - EDTA, A Molecule With A Complex Story PDFDocument6 pagesScott A. Sinex - EDTA, A Molecule With A Complex Story PDFcountdankNo ratings yet
- Complexometric Titration With EDTADocument8 pagesComplexometric Titration With EDTAManP13100% (1)
- Complexometry TitartaionDocument50 pagesComplexometry TitartaionHaritNo ratings yet
- Complexometric EDTADocument36 pagesComplexometric EDTANqobile Nqoerh MnguniNo ratings yet
- CHM121 Module 8 Complexation-TitrimeteryDocument7 pagesCHM121 Module 8 Complexation-TitrimeteryEdcel ZabalaNo ratings yet
- Editable Experiment Record TemplateDocument5 pagesEditable Experiment Record TemplateNemalnath reddy KasarapuNo ratings yet
- Complexometry: Pharmaceutical Analysis For Liquid and Semisolid PreparationDocument32 pagesComplexometry: Pharmaceutical Analysis For Liquid and Semisolid PreparationaulianiNo ratings yet
- 4047683-complexometryDocument37 pages4047683-complexometryArnab ChakrabortyNo ratings yet
- Complexometric Titrations: StructureDocument13 pagesComplexometric Titrations: StructureAnujNo ratings yet
- Chap 3. Complexation ReactionTitrationDocument51 pagesChap 3. Complexation ReactionTitrationlesangg1406No ratings yet
- Complexometric Titration: Complex (Coordination Compound)Document13 pagesComplexometric Titration: Complex (Coordination Compound)Ben AbellaNo ratings yet
- Engineering Chemistry (CHY1701) : Schedule (A2+TA2)Document24 pagesEngineering Chemistry (CHY1701) : Schedule (A2+TA2)Sooraj DevNo ratings yet
- Lewis Acid-Base Concept: Chapter 13 EDTA TitrationsDocument23 pagesLewis Acid-Base Concept: Chapter 13 EDTA TitrationsEdin AbolenciaNo ratings yet
- EDTADocument2 pagesEDTARay AribatoNo ratings yet
- Tittrations Based On Complex FormationDocument5 pagesTittrations Based On Complex FormationNisaNo ratings yet
- KGB Complexometric TitrationDocument36 pagesKGB Complexometric TitrationpharmaprvNo ratings yet
- Complexation NotesDocument6 pagesComplexation NotesDeepak PradhanNo ratings yet
- Complexometric TitrationDocument8 pagesComplexometric TitrationYUNITA DWINo ratings yet
- Complexometric TitrationsDocument16 pagesComplexometric TitrationsRakesh Chawla0% (1)
- EdtaDocument6 pagesEdtasam310justNo ratings yet
- Complexometry-1Document11 pagesComplexometry-1dhagedaksheshNo ratings yet
- Experiment 2:: Complexometric TitrationsDocument6 pagesExperiment 2:: Complexometric TitrationsGhali El SamadNo ratings yet
- Complexometric TitrationDocument29 pagesComplexometric TitrationLive Happy100% (1)
- QC Lec NotesDocument20 pagesQC Lec NotesJerome Delos ReyesNo ratings yet
- Complexometric Titration PDFDocument11 pagesComplexometric Titration PDFDr. Gunjan SarkarNo ratings yet
- Electro-Fenton For Industrial Wastewater TreatmentDocument5 pagesElectro-Fenton For Industrial Wastewater TreatmentKhaled NaguibNo ratings yet