Professional Documents
Culture Documents
Mycorrhizae: Plant Roots and Fungi
Mycorrhizae: Plant Roots and Fungi
Uploaded by
Muhammad Faiq0 ratings0% found this document useful (0 votes)
14 views29 pagesThis document discusses mycorrhizae, which are symbiotic relationships between fungi and plant roots. It describes the two main types - ectomycorrhizae and endomycorrhizae. Ectomycorrhizae involve a fungal sheath and Hartig net surrounding root cells. These relationships benefit both the plant and fungus by increasing nutrient and water absorption for the plant and providing carbohydrates for the fungus.
Original Description:
Original Title
Ectomycorrhizae 2019
Copyright
© © All Rights Reserved
Available Formats
PPTX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document discusses mycorrhizae, which are symbiotic relationships between fungi and plant roots. It describes the two main types - ectomycorrhizae and endomycorrhizae. Ectomycorrhizae involve a fungal sheath and Hartig net surrounding root cells. These relationships benefit both the plant and fungus by increasing nutrient and water absorption for the plant and providing carbohydrates for the fungus.
Copyright:
© All Rights Reserved
Available Formats
Download as PPTX, PDF, TXT or read online from Scribd
Download as pptx, pdf, or txt
0 ratings0% found this document useful (0 votes)
14 views29 pagesMycorrhizae: Plant Roots and Fungi
Mycorrhizae: Plant Roots and Fungi
Uploaded by
Muhammad FaiqThis document discusses mycorrhizae, which are symbiotic relationships between fungi and plant roots. It describes the two main types - ectomycorrhizae and endomycorrhizae. Ectomycorrhizae involve a fungal sheath and Hartig net surrounding root cells. These relationships benefit both the plant and fungus by increasing nutrient and water absorption for the plant and providing carbohydrates for the fungus.
Copyright:
© All Rights Reserved
Available Formats
Download as PPTX, PDF, TXT or read online from Scribd
Download as pptx, pdf, or txt
You are on page 1of 29
Mycorrhizae
Plant roots and fungi
Mycorrhizae
• Widespread interactions between fungi and plant
(primarily vascular plants) roots
• For angiosperms, gymnosperms, ferns and some
mosses – mycorrhizal association appears to be the
norm
• Range over broad spectrum of interactions
• Fungus parasitizes plant
• Plant parasitizes fungus
• Most cases – mutualistic – both benefit
Types of mycorrhizae
• Ectomycorrhizae (ectotrophic, sheathing) – hyphae of
fungus do not penetrate cells of plant root
• Endomycorrhizae – hyphae penetrate cells of plant
• Arbuscular mycorrhizae (AM) – aseptate hyphae, most
widespread
• Septate hyphae
• Ericoid, Arbutoid & Monotropoid – plants are Ericales
• Orchid – plants are orchids
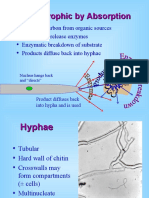
Ectomycorrhizae
• Most conspicuous and easily recognized
• Best characterized
• Plant roots are enclosed by a sheath of fungal hyphae –
fungal mycelium penetrates between cells in cortex of
the root
• Fungal tissue may account for up to 40% mass of root
• Hyphae also extend out into the soil – extramatrical
hyphae
In ectomycorrhizal (ECM)
roots, the fungus forms
extraradical hyphae and a
fungal sheath outside the
root (A) and the Hartig
net surrounding root cells
(B) hiding epidermal cells
and cortical (cc) cells (C).
(A) In fungal cells, the uptake of Pi occurs mostly
through Pht1 phosphate transporters.
To date, only HcPT1.1, HcPT2 (Tatry et al., 2009), and
BePT (Wang et al., 2014) genes have been characterized
by heterologous expression in yeasts.
Genomics and transcriptomic data suggest that other
transporters may play a role in phosphate uptake (e.g.,
HcPT1.2, LbPTs, AmPTs, TvPTs, TmPT3 (Pht2; Casieri
et al., 2013).
(B) In the Hartig net, fungal and plant cells have a common
apoplastic space with no direct symplastic communication.
It is hypothesized that the hydrolysis (a) of polyphosphate
(PolyP) increases Pi concentration in the cytosol of the fungus.
Up to now, the molecular mechanisms sustaining P efflux from
the fungus (b) to the apoplast and P influx (c) from the apoplast
to the plant cell have not been identified.
It is also hypothesized that fungal P transporters may not be
functioning (d)
(C) In plant cells, phosphate ions enter through plant P
transporters. Little is known about plant transporters
responsible for Pi acquisition in ECM roots. Only phosphate
transporters from Populus trichocarpa (PtPTs; Loth-Pereda et
al., 2011) and Eucalyptus marginata (EmPhts; Kariman et al.,
2014) have so far been identified.
Transcriptomic data for Pinus pinaster (Canales et al., 2013)
showed putative encoding sequences for phosphate
transporters (PpPTs).
Full lines indicate transport systems whose
capability in phosphate transport has been
verified by heterologous expression in
yeast.
Dotted lines indicate transport systems
whose involvement in phosphate
transport during mycorrhizal symbioses is
suggested by genomic or transcriptomic
data.
Ed: endodermal cells. Hc: Hebeloma
cylindrosporum, Be: Boletus edulis, Tm:
Tuber melanosporum, Am: Amanita
muscaria, Lb: Laccaria bicolor, Tv:
Tricholoma vaccinum, Pt: Populus
trichocarpa, Em: Eucalyptus marginata,
Pp: Pinus pinaster.
Ectomycorrhizae
• Contains a fungal sheath
• Parenchyma of root
cortex is surrounded by
hyphae – Hartig net
Ectomycorrhizal root
Ectomycorrhizae
• Absorbing roots are those that are affected
• Become thicker and repeatedly branched after
infection
Ectomycorrhizae
Ectomycorrhizae Symbionts
• 2000 plant species – primarily temperate trees and eucalyptus
• Major species of coniferous and deciduous trees
• Rare to find uninfected trees
• In some trees, the association is obligate, in others facultative
• Mycorrhizal association important in forestry
Ectomycorrhizae Symbionts
• Basidiomycetes – Agaricales (many mushroom
species), Lycoperdales, Sclerodermatales, few
Aphyllophorales
• Pisolithus tinctorus – used to form commercial inoculum
for nursery trees, common in southern pine
• Ascomycota – Pezizales – cup fungi and truffles
• Over 5000 species of fungi have been shown to form
ectomycorrhizae
Specificity of association
• Great deal of variability
• Most tree species form mycorrhizal associations with a
number of different fungal species
• May have different mycorrhizal fungi on roots of one plant
• Some fungi are fairly specific and will form associations
with only one plant species – these mushrooms are
common in stands of that tree
• Others are not specific
Specificity
• Douglas fir has been
extensively studied and ca
2000 species of fungi have
been identified from its
roots
• In forests, a high
percentage of fruiting
bodies are mycorrhizal
fungi
Occurence
Methods for detection
• Census of fruiting bodies produced by different
species
• Soil cores – separate and identify mycorrhizal roots by
morphology, Hartig net
• Recently molecular methods have been used to
identify the fungi present in mycorrhizal roots – e.g.
Restriction Fragment Length Polymorphism (RFLP)
Ectomycorrhizal fungi
• Can also grow saprotrophically
• Many have been cultured
• Most that have been studied do not have the
capability to degrade complex plant polymers (e.g.
cellulose and lignin)
• Depend on soluble carbohydrates
• Many have organic growth factor requirements –
vitamins, amino acids
• Not decomposers but depend on plant
Benefits to fungus
• Provided with source of C and energy
• Plants provided with 14CO2 demonstrated that 14C
appears in fungus
• Sucrose from plant converted into trehalose, mannitol
by fungus
• Estimates that up to 10% (or more) of photosynthate
produced by trees is passed to mycorrhizae and other
rhizosphere organisms
Benefits to trees
• Numerous studies have
shown that tree growth
is better when
mycorrhizae are present
Benefits to trees
Benefits to trees
• Fungi increase supply of inorganic nutrients to tree. P is insoluble in
most soils
• Extramatrical hyphae extend over a larger volume of soil than roots
can – increase ability to absorb insoluble nutrients such as P
Extramatrical
hyphae
Volume of soil explored
Benefits to trees
• Plant hormones produced by fungus changes the physiological state
of roots – physiologically active root area for nutrient and water
absorption is increased
• Increases tolerance of plant to drought, high temperatures, pH
extremes, heavy metals
• Increases resistance to infection by root pathogens – provides a
physical barrier
You might also like
- Fungal PharmacyDocument121 pagesFungal PharmacyAntoni Gandia100% (7)
- TIR-A15-14 Design Wind Load DeterminationDocument31 pagesTIR-A15-14 Design Wind Load Determinationjay kimNo ratings yet
- Feasibility StudyDocument5 pagesFeasibility StudyKath Garcia50% (4)
- Jharkhand General Knowledge (Objective Question)Document8 pagesJharkhand General Knowledge (Objective Question)Gunjan86% (28)
- Tesla Motors Expansion StrategyDocument26 pagesTesla Motors Expansion StrategyVu Nguyen75% (4)
- Research Methods For Architecture Ebook - Lucas, Ray - Kindle Store PDFDocument1 pageResearch Methods For Architecture Ebook - Lucas, Ray - Kindle Store PDFMohammed ShriamNo ratings yet
- Mycorrhizae: Plant Roots and FungiDocument29 pagesMycorrhizae: Plant Roots and FungigitaNo ratings yet
- MikorizaDocument29 pagesMikorizayasmine aurelliaNo ratings yet
- Mycorrhiza 180310082441Document58 pagesMycorrhiza 180310082441Khadija100% (1)
- Ectomy Corr Hi ZaeDocument15 pagesEctomy Corr Hi ZaeAnkit VermaNo ratings yet
- MYCORRHIZA Prep by Ali Irfan ILBASDocument41 pagesMYCORRHIZA Prep by Ali Irfan ILBASMurilo MorenoNo ratings yet
- MycorrhizaeDocument23 pagesMycorrhizaeHussain TaqiNo ratings yet
- Mycorrhiza in Agriculture: Unit 04: Plant-Microbe Interaction Lecture By: Rajiv PathakDocument35 pagesMycorrhiza in Agriculture: Unit 04: Plant-Microbe Interaction Lecture By: Rajiv Pathakrajiv pathakNo ratings yet
- GeethaDocument27 pagesGeethalatif khanNo ratings yet
- Diversity of PlantsDocument15 pagesDiversity of PlantsDavidNo ratings yet
- FUNGIDocument33 pagesFUNGIGabz Gabby57% (7)
- 1008458-Chapter 7 - Ecosystem DynamicDocument12 pages1008458-Chapter 7 - Ecosystem DynamicrhimalinyNo ratings yet
- Intro To Fungi Presentation - Fall 2008Document36 pagesIntro To Fungi Presentation - Fall 2008davidbraunsteinNo ratings yet
- Lecture 1 - Diversity and Evolution of Plants (Non-Vascular, Gymnosperm, and Angiosperm)Document30 pagesLecture 1 - Diversity and Evolution of Plants (Non-Vascular, Gymnosperm, and Angiosperm)Jan Leo QuinioNo ratings yet
- Mcat Self PrepDocument27 pagesMcat Self PrepAbdul Manan100% (1)
- Yeasts 2023Document82 pagesYeasts 2023am6443559No ratings yet
- Mycorrhiza 1Document2 pagesMycorrhiza 1KAJAL MAITYNo ratings yet
- Week 5 Lecture 20231013 FinalDocument54 pagesWeek 5 Lecture 20231013 FinalBrylle ChavezNo ratings yet
- Plant/Microbe Interactions - BeneficialDocument31 pagesPlant/Microbe Interactions - BeneficialAdi CandraNo ratings yet
- Fungal NutritionDocument32 pagesFungal NutritionHarits AmrullohNo ratings yet
- Seedless Vascular PlantsDocument33 pagesSeedless Vascular PlantsMnqobi ZuluNo ratings yet
- Kingdom FungiDocument31 pagesKingdom Fungiraul sinatoNo ratings yet
- Basic MycologyDocument30 pagesBasic Mycologymalikdanig40No ratings yet
- Overview of Green PlantsDocument49 pagesOverview of Green PlantsMary R. R. PanesNo ratings yet
- Plant Development - An OverviewDocument53 pagesPlant Development - An OverviewBinita SedhaiNo ratings yet
- Evolution of PlantsDocument29 pagesEvolution of Plantstapas kunduNo ratings yet
- Mycorrhiza AssignmentDocument4 pagesMycorrhiza AssignmentmohkristNo ratings yet
- Jim's Discussion Notes: How Plants Colonized LandDocument21 pagesJim's Discussion Notes: How Plants Colonized LandSalik Sarosh TehamiNo ratings yet
- Tracheoph YTA: Report By: Veronica BajeDocument22 pagesTracheoph YTA: Report By: Veronica BajejohnNo ratings yet
- MycorrhizaDocument11 pagesMycorrhizaJames Nicholas JohnnyNo ratings yet
- Animlas & FungiDocument39 pagesAnimlas & Fungihajrabatool27No ratings yet
- Biol 1011 Plants Lecture 1 BrightspaceDocument61 pagesBiol 1011 Plants Lecture 1 Brightspaceojh0218azatNo ratings yet
- Mycorrhizae: Dr. Siti Sunariyati, Msi Faculty of Teacher Training and Education of Palangkaraya UniversityDocument43 pagesMycorrhizae: Dr. Siti Sunariyati, Msi Faculty of Teacher Training and Education of Palangkaraya UniversityArrum Chyntia YuliyantiNo ratings yet
- Week 7 PP10 LecturesDocument34 pagesWeek 7 PP10 LecturesHannymie maurealNo ratings yet
- Biology Quiz (1st Year)Document26 pagesBiology Quiz (1st Year)Lisa PretoriusNo ratings yet
- Kingdom PlantaeDocument27 pagesKingdom PlantaeKinza AminNo ratings yet
- Oraganism and Its EnvironmentsDocument7 pagesOraganism and Its EnvironmentsKim AlvarezNo ratings yet
- Dr. Abhinandan R Patil Research ScientistDocument59 pagesDr. Abhinandan R Patil Research ScientistAbhinandan PatilNo ratings yet
- Nutrition (XI) in PlantsDocument56 pagesNutrition (XI) in PlantsMehak MuradaniNo ratings yet
- Lecture 2 - Survey of The Plant Kingdom Part 2 PDFDocument15 pagesLecture 2 - Survey of The Plant Kingdom Part 2 PDFpandorum0% (1)
- The Rhizosphere: A Playground and Battlefield For Soilborne Pathogens and Beneficial MicroorganismsDocument17 pagesThe Rhizosphere: A Playground and Battlefield For Soilborne Pathogens and Beneficial Microorganismswafiyyatunnufus100% (1)
- Basic Biology 2 Plant Diversity-1Document12 pagesBasic Biology 2 Plant Diversity-1TeeNo ratings yet
- Kingdom PlantaeDocument14 pagesKingdom Plantaeazileinra OhNo ratings yet
- Bon Fante 2010Document11 pagesBon Fante 2010Gabriel Leonardo Tacchi NascimentoNo ratings yet
- Characteristics of FungiDocument1 pageCharacteristics of FungiBeyond GodlikeNo ratings yet
- BioreportDocument8 pagesBioreportAbigail CostalesNo ratings yet
- Green Algea: Phylum Chlorophyta: - Most Aquatic (Freshwater - Also Found On SnowDocument29 pagesGreen Algea: Phylum Chlorophyta: - Most Aquatic (Freshwater - Also Found On SnowErry MaryaniNo ratings yet
- Unearthing The Roots of Ectomycorrhizal SymbiosesDocument14 pagesUnearthing The Roots of Ectomycorrhizal SymbiosesIan SaundersNo ratings yet
- BLG 1502 QuizDocument26 pagesBLG 1502 QuizLisa PretoriusNo ratings yet
- BIO 102 Lecture 3 Bryophytes UpdatedDocument55 pagesBIO 102 Lecture 3 Bryophytes UpdatedEsther OgelekaNo ratings yet
- Plant KingdomDocument8 pagesPlant Kingdomsaikatmajee727No ratings yet
- Biology 1 Species of OrganismsDocument9 pagesBiology 1 Species of OrganismsOrlan Defensor BalanoNo ratings yet
- Bio Note9Document4 pagesBio Note9Debela mendaraNo ratings yet
- WHY Are They Important?: Food, Carbon Regulation, O Prod. Habitat, Timber, Medicine and Beautiful Ecology To Enjoy!Document68 pagesWHY Are They Important?: Food, Carbon Regulation, O Prod. Habitat, Timber, Medicine and Beautiful Ecology To Enjoy!BennyNo ratings yet
- DAHILOG WORKSHEET 1 ScieEd 210Document9 pagesDAHILOG WORKSHEET 1 ScieEd 210Ybur Clieve Olsen DahilogNo ratings yet
- Perbandingan Jumlah Eritrosit Pada Sampel Darah 3, 2 Dan 1 ML Dengan Antikoagulan K2EdtaDocument6 pagesPerbandingan Jumlah Eritrosit Pada Sampel Darah 3, 2 Dan 1 ML Dengan Antikoagulan K2EdtaNia AzNo ratings yet
- Essay EnvironmentDocument4 pagesEssay Environmenttuevptvhd100% (2)
- Employee Uniform Agreement Sample Template 092321 3Document3 pagesEmployee Uniform Agreement Sample Template 092321 3zenithcrew.financeNo ratings yet
- Eng/Npd Girish Comprehen Sive Charan Marketing: Internal Quality Audit Schedule No:Qms Ia-1Document1 pageEng/Npd Girish Comprehen Sive Charan Marketing: Internal Quality Audit Schedule No:Qms Ia-1DhinakaranNo ratings yet
- SAP HANA StudioDocument33 pagesSAP HANA StudioPrateekVermaNo ratings yet
- Emflon PFRW Filter Cartridges: For Sterile Filtration of GasesDocument3 pagesEmflon PFRW Filter Cartridges: For Sterile Filtration of GasesRichard BakerNo ratings yet
- Normalized Impedance and Admittance Coordinates: Name Title Dwg. NoDocument1 pageNormalized Impedance and Admittance Coordinates: Name Title Dwg. NoJayeng WidiatmokoNo ratings yet
- Ict Dos and DontsDocument5 pagesIct Dos and DontsLouwellaEredianoIbabaoNo ratings yet
- Experience With The Munster-Type Below-Elbow Prosthesis, A Preliminary ReportDocument5 pagesExperience With The Munster-Type Below-Elbow Prosthesis, A Preliminary ReportrehabeasyNo ratings yet
- ION-Evolution - ERA PANOS-RSDocument29 pagesION-Evolution - ERA PANOS-RSSokratesNo ratings yet
- Arsenio T. Mendiola V. CaDocument7 pagesArsenio T. Mendiola V. CaCharisa BelistaNo ratings yet
- Hdat2en 4 52Document156 pagesHdat2en 4 52blahh12345689No ratings yet
- 11thsamplebooklet PDFDocument200 pages11thsamplebooklet PDFSoumyodeep ChowdhuryNo ratings yet
- Debug Tacacs CiscoDocument5 pagesDebug Tacacs CiscodeztrocxeNo ratings yet
- Dwnload Full Intermediate Accounting 8th Edition Spiceland Solutions Manual PDFDocument12 pagesDwnload Full Intermediate Accounting 8th Edition Spiceland Solutions Manual PDFsorbite.bebloodu2hh0j100% (15)
- Digital NativesDocument10 pagesDigital NativesJonathan Blanco Sa RiyadhNo ratings yet
- Health Education & Behaviour Change CommunicationDocument50 pagesHealth Education & Behaviour Change CommunicationAarya MathewNo ratings yet
- C-18 Dme-I-SemDocument94 pagesC-18 Dme-I-SemBollu SatyanarayanaNo ratings yet
- Engleza Maritima 4: 2a-SMCP External ROUTINE Communication - READING Comprehension-Book1Document5 pagesEngleza Maritima 4: 2a-SMCP External ROUTINE Communication - READING Comprehension-Book1dark959595No ratings yet
- H11L1Document6 pagesH11L1Silvio QuerzoliNo ratings yet
- Arm RollDocument2 pagesArm Rollahmad elnggarNo ratings yet
- Human Resource Information System: Presented By: Jasleen Dutta Presented To: Prof. Afreen AliDocument22 pagesHuman Resource Information System: Presented By: Jasleen Dutta Presented To: Prof. Afreen AliAmanpreet DuttaNo ratings yet
- Bulletin Low Res 2Document19 pagesBulletin Low Res 2Аксана ГатауллинаNo ratings yet
- Police RRDocument17 pagesPolice RRindrajitdhadhal400No ratings yet
- Product Environmental Profile: Canalis KNA 40A To 160ADocument7 pagesProduct Environmental Profile: Canalis KNA 40A To 160AJavier LazoNo ratings yet