Professional Documents
Culture Documents
RBC Membrane Deformability
RBC Membrane Deformability
Uploaded by
Myedelle Seacor0 ratings0% found this document useful (0 votes)
25 views16 pagesRBCs are biconcave discs that are highly deformable, allowing them to pass through narrow capillaries. This deformability depends on their membrane properties. The RBC membrane is a lipid bilayer containing cholesterol, phospholipids, proteins, and carbohydrates. Cholesterol provides strength while phospholipids and membrane proteins regulate permeability, transport, signaling and cytoskeletal structure. Together, the membrane components facilitate gas exchange and maintain osmotic balance through deformation, cation transport and water permeability.
Original Description:
Original Title
Rbc_membrane_deformability
Copyright
© © All Rights Reserved
Available Formats
PPTX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentRBCs are biconcave discs that are highly deformable, allowing them to pass through narrow capillaries. This deformability depends on their membrane properties. The RBC membrane is a lipid bilayer containing cholesterol, phospholipids, proteins, and carbohydrates. Cholesterol provides strength while phospholipids and membrane proteins regulate permeability, transport, signaling and cytoskeletal structure. Together, the membrane components facilitate gas exchange and maintain osmotic balance through deformation, cation transport and water permeability.
Copyright:
© All Rights Reserved
Available Formats
Download as PPTX, PDF, TXT or read online from Scribd
Download as pptx, pdf, or txt
0 ratings0% found this document useful (0 votes)
25 views16 pagesRBC Membrane Deformability
RBC Membrane Deformability
Uploaded by
Myedelle SeacorRBCs are biconcave discs that are highly deformable, allowing them to pass through narrow capillaries. This deformability depends on their membrane properties. The RBC membrane is a lipid bilayer containing cholesterol, phospholipids, proteins, and carbohydrates. Cholesterol provides strength while phospholipids and membrane proteins regulate permeability, transport, signaling and cytoskeletal structure. Together, the membrane components facilitate gas exchange and maintain osmotic balance through deformation, cation transport and water permeability.
Copyright:
© All Rights Reserved
Available Formats
Download as PPTX, PDF, TXT or read online from Scribd
Download as pptx, pdf, or txt
You are on page 1of 16
Rbc membrane deformability
RBCs are biconcave and average 90 fL in volume.
• Their average surface area is 140 μm2, a 40% excess of surface area
compared with a 90-fL sphere. This excess surface-to-volume ratio
enables RBCs to stretch undamaged up to 2.5 times their resting
diameter as they pass through narrow capillaries and through splenic
pores 2 μm in diameter; this property is called RBC deformability.
• The RBC plasma membrane, which is 5 μm thick, is 100 times more
elastic than a comparable latex membrane, yet it has tensile (lateral)
strength greater than that of steel.
• The deformable RBC membrane provides the broad surface area and
close tissue contact necessary to support the delivery of O2 from lungs
to body tissue and CO2 from body tissue to lungs.
• RBC deformability depends not only on RBC geometry but also on
relative cytoplasmic (hemoglobin) viscosity. The normal mean cell
hemoglobin concentration (MCHC) ranges from 32% to 36%, and as
MCHC rises, internal viscosity rises. MCHCs above 36% compromise
deformability and shorten the RBC life span because viscous cells
become damaged as they stretch to pass through narrow capillaries
or splenic pores. As RBCs age, they lose membrane surface area,
while retaining hemoglobin. As the MCHC rises, the RBC, unable to
pass through the splenic pores, is destroyed by splenic macrophages.
Rbc membrane lipids
Besides geometry and viscosity, membrane elasticity (pliancy) also contributes to
deformability.
The RBC membrane consists of approximately:
1. 8% carbohydrates
2. 52% proteins
3. 40% lipids.
The lipid portion, equal parts of cholesterol and phospholipids, forms a bilayer universal
to all animal cells. Phospholipids form an impenetrable fluid barrier as their hydrophilic
polar head groups are arrayed upon the membrane’s surfaces, oriented toward both the
aqueous plasma and the cytoplasm, respectively. Their hydrophobic nonpolar acyl tails
arrange themselves to form a central layer dynamically sequestered (hidden) from the
aqueous plasma and cytoplasm. The membrane maintains extreme differences in
osmotic pressure, cation concentrations, and gas concentrations between external
plasma and the cytoplasm. Phospholipids reseal rapidly when the membrane is torn.
• Cholesterol, esterified and largely hydrophobic, resides parallel to the
acyl tails of the phospholipids, equally distributed between the outer
and inner layers, and evenly dispersed within each layer,
approximately one cholesterol molecule per phospholipid molecule.
Cholesterol’s β-hydroxyl group, the only hydrophilic portion of the
molecule, anchors within the polar head groups, while the rest of the
molecule becomes intercalated among and parallel to the acyl tails.
Cholesterol confers tensile strength to the lipid bilayer.
https://images.app.goo.gl/X3NAuKoGraLRQ2LH7
The ratio of cholesterol to
phospholipids remains
relatively constant and balances
the need for deformability and
strength. Membrane enzymes
maintain the cholesterol
concentration by regularly
exchanging membrane and
plasma cholesterol. Deficiencies
in these enzymes are
associated with membrane
abnormalities such as
acanthocytosis, as the https://images.app.goo.gl/yNxgTxjizz4ZHa398
membrane loses tensile
strength. Conversely, as
cholesterol concentration rises,
the membrane gains strength
but loses elasticity.
• The phospholipids are asymmetrically distributed.
Phosphatidylcholine and sphingomyelin predominate in the outer
layer; phosphatidylserine (PS) and phosphatidylethanolamine form
most of the inner layer. Distribution of these four phospholipids is
energy dependent, relying on a number of membrane-associated
enzymes, whimsically termed flippases, floppases, and scramblases,
for their positions.
• When phospholipid distribution is disrupted, as in sickle cell anemia
and thalassemia or in RBCs that have reached their 120-day life
span, PS, the only negatively charged phospholipid, redistributes
(flips) to the outer layer. Splenic macrophages possess receptors that
bind PS and destroy senescent and damaged RBCs.
Membrane phospholipids and
cholesterol may also redistribute
laterally so that the RBC membrane may
respond to stresses and deform within
100 milliseconds of being challenged by
the presence of a narrow passage, such
as when arriving at a capillary.
Redistribution becomes limited as the
proportion of cholesterol increases.
Plasma bile salt concentration also
affects cholesterol exchange. In liver
disease with low bile salt concentration,
membrane cholesterol concentration
becomes reduced. As a result, the more
elastic cell membrane shows a “target
https://images.app.goo.gl/3ueu5YDwhKtQFCiE6
cell” appearance when the RBCs are
layered on a glass slide.
• Glycolipids (sugar-bearing lipids) make up 5% of the external half of
the RBC membrane. They associate in clumps or rafts and support
carbohydrate side chains that extend into the aqueous plasma to
anchor the glycocalyx. The glycocalyx is a layer of carbohydrates
whose net negative charge prevents microbial attack and protects the
RBC from mechanical damage caused by adhesion to neighboring
RBCs or to the endothelium. Glycolipids may bear copies of
carbohydrate-based blood group antigens, for example, antigens of
the ABH and the Lewis blood group systems.
Rbc membrane proteins
Although cholesterol and phospholipids constitute the principal RBC
membrane structure, transmembrane (integral) and cytoskeletal
(skeletal, peripheral) proteins make up 52% of the membrane structure
by mass. A proteomic study reveals there are at least 300 RBC
membrane proteins, including 105 transmembrane proteins. Of the
purported 300 membrane proteins, about 50 have been characterized
and named, some with a few hundred copies per cell, and others with
over a million copies per cell
Transmembrane proteins
• The transmembrane proteins serve a number of RBC functions. Through
glycosylation they support surface carbohydrates, which join with glycolipids
to make up the protective glycocalyx. They serve as transport and adhesion
sites and signaling receptors. Any disruption in transport protein function
changes the osmotic tension of the cytoplasm, which leads to a rise in
viscosity and loss of deformability. Any change affecting adhesion proteins
permits RBCs to adhere to one another and to the vessel walls, promoting
fragmentation (vesiculation), reducing membrane flexibility, and shortening
the RBC life span. Signaling receptors bind plasma ligands and trigger
activation of intracellular signaling proteins, which then initiate various
energy-dependent cellular activities, a process called signal transduction.
Skeletal Proteins Functions
α-spectrin Filamentous antiparallel heterodimer, primary
β-spectrin cytoskeletal proteins
Adducin Caps actin filament
Ankyrin Anchors band 3 and protein 4.2
Dematin Actin bundling protein
F-actin Binds β-spectrin
G3PD
Protein 4.1 Anchors 4.1 complex
Protein 4.2 (protein kinase) Anchors ankyrin complex
Tropomodulin Caps actin filament
Tropomyosin Regulates actin polymerization
Osmotic balance and permeability
• The RBC membrane is impermeable to cations Na+, K+, and Ca2+. It is
permeable to water and the anions bicarbonate (HCO3−) and chloride
(Cl−), which freely exchange between plasma and RBC cytoplasm.
Aquaporin 1 is a transmembrane protein that forms pores or channels
whose surface charges create inward water flow in response to internal
osmotic changes.
• The ATP–dependent cation pumps Na+ -ATPase and K+ -ATPase regulate
the concentrations of Na+ and K+, maintaining intracellular-to-
extracellular ratios of 1:12 and 25:1, respectively. Ca2+ -ATPase extrudes
calcium, maintaining low intracellular levels of 5 to 10 μmol/L.
Calmodulin, a cytoplasmic Ca2+-binding protein, controls the function of
Ca2+-ATPase. These enzymes, in addition to aquaporin, maintain
osmotic balance.
• The cation pumps consume 15% of RBC ATP production. ATP loss or
pump damage permits Ca2+ and Na+ influx, with water following
osmotically. The cell swells, becomes spheroid, and eventually
ruptures. This phenomenon is called colloid osmotic hemolysis.
• Sickle cell disease provides an example of increased cation
permeability. When crystallized sickle hemoglobin deforms the cell
membranes, internal levels of Na+, K+, and especially Ca2+ rise, which
results in hemolysis
https://images.app.goo.gl/QbmTLJyQb74yTgWM7
You might also like
- Preclinical Physiology Review 2023: For USMLE Step 1 and COMLEX-USA Level 1From EverandPreclinical Physiology Review 2023: For USMLE Step 1 and COMLEX-USA Level 1Rating: 1 out of 5 stars1/5 (1)
- The Ammonicarb Operation Book Format 4 PDF FreeDocument149 pagesThe Ammonicarb Operation Book Format 4 PDF FreePantelis Krintz100% (1)
- Biochem ABO IncompatibilityDocument62 pagesBiochem ABO IncompatibilityChi Samaniego100% (4)
- Membrane DeformabilityDocument2 pagesMembrane DeformabilityKenneth Jake BatiduanNo ratings yet
- RBC MembraneDocument41 pagesRBC MembranemwambungupNo ratings yet
- Erythrocyte Metabolism AND Membrane Structure and FunctionDocument29 pagesErythrocyte Metabolism AND Membrane Structure and FunctionJohn Marie IdavaNo ratings yet
- Red Blood Cell BiochemistryDocument19 pagesRed Blood Cell BiochemistryPrincewill SeiyefaNo ratings yet
- Plasma Membrane: The Lipid Bilayer Membrane ProteinsDocument79 pagesPlasma Membrane: The Lipid Bilayer Membrane ProteinsEarfanNo ratings yet
- Lipid Movement BiochemDocument9 pagesLipid Movement BiochemCrowNo ratings yet
- G Rojos BlancosDocument7 pagesG Rojos BlancosAndrea Valenzuela AedoNo ratings yet
- Module 3 - Lecture 3 - LectureDocument8 pagesModule 3 - Lecture 3 - LectureNi CaoNo ratings yet
- RBC MembraneDocument37 pagesRBC MembraneStanley ChikoveNo ratings yet
- 01 Cell MembraneDocument22 pages01 Cell MembraneMian SahibNo ratings yet
- Chapter 9 HemaDocument4 pagesChapter 9 HemaJoannah MarieNo ratings yet
- Study Exercise Biology 10-12 97.03Document7 pagesStudy Exercise Biology 10-12 97.03Fabiola Vania FeliciaNo ratings yet
- Research Paper On Plasma MembraneDocument4 pagesResearch Paper On Plasma Membranevvgnzdbkf100% (1)
- Erythrocyte Metabolism and Membrane Structure and FunctionDocument38 pagesErythrocyte Metabolism and Membrane Structure and FunctionRej MaeNo ratings yet
- Chapter 4-Jnu Biomembranes-1 - 複本Document48 pagesChapter 4-Jnu Biomembranes-1 - 複本Wai Kwong ChiuNo ratings yet
- Components of Plasma Membranes: Learning ObjectivesDocument14 pagesComponents of Plasma Membranes: Learning ObjectivesAlthea Junsan BernabeNo ratings yet
- Biochemistry & Genetics: II SHS 109: Resource Person: DR Tanveer Akbar Reference TextDocument60 pagesBiochemistry & Genetics: II SHS 109: Resource Person: DR Tanveer Akbar Reference TexttNo ratings yet
- The Cell MembraneDocument36 pagesThe Cell Membraneanulorance98No ratings yet
- Cell MembraneDocument2 pagesCell Membranehello123No ratings yet
- Lect 1 Membrane Structure and FunctionDocument67 pagesLect 1 Membrane Structure and FunctionIzukanji kayoraNo ratings yet
- Topic 3Document12 pagesTopic 3Ria AccadNo ratings yet
- Life at The Edge Concept 7.1: Structure! Cellular Membranes Are Fluid Mosaics of Lipids and ProteinsDocument6 pagesLife at The Edge Concept 7.1: Structure! Cellular Membranes Are Fluid Mosaics of Lipids and ProteinsAisha100% (1)
- Plasma Membrane Research PapersDocument6 pagesPlasma Membrane Research Papersaflbasnka100% (1)
- Lipid Traffic Floppy Drives and A SuperhighwayDocument12 pagesLipid Traffic Floppy Drives and A SuperhighwayJohan AlarcónNo ratings yet
- Red Blood Cell MembraneDocument28 pagesRed Blood Cell MembraneAisyah Atikah100% (1)
- Plasma Membrane in DetailsDocument24 pagesPlasma Membrane in DetailsSyeda Kaneez Mohammadi MohammadiNo ratings yet
- CellDocument48 pagesCellSoumik SahaNo ratings yet
- Kuliah MembranDocument61 pagesKuliah Membrannikmah nuur rochmahNo ratings yet
- Solution BiotechDocument54 pagesSolution Biotechkavita sahaiNo ratings yet
- Biological Cell MembranesDocument44 pagesBiological Cell MembranesMohammed ShaikNo ratings yet
- Chemistry of Lipid Domains: Advanced Article Richard M. EpandDocument5 pagesChemistry of Lipid Domains: Advanced Article Richard M. EpandazzaassNo ratings yet
- Chapter 5 Lipids Membrane LectureslidesDocument65 pagesChapter 5 Lipids Membrane Lectureslidesduy trầnNo ratings yet
- Chapter Blood: RBC Platelet HemostasisDocument89 pagesChapter Blood: RBC Platelet Hemostasisapi-19916399100% (1)
- Plasma MembraneDocument35 pagesPlasma MembraneMini RůžičkaNo ratings yet
- Cell Membrane (Structure and Function)Document150 pagesCell Membrane (Structure and Function)Dr. Tapan Kr. Dutta100% (1)
- CellMembrane Structure and Functional PropertiesDocument5 pagesCellMembrane Structure and Functional PropertiesAnastasiafynnNo ratings yet
- K - 8 Blood Brain Barrier (Biokimia)Document31 pagesK - 8 Blood Brain Barrier (Biokimia)Firman YunusNo ratings yet
- Biochemistry ProblemDocument5 pagesBiochemistry ProblemJnanendra SenguptaNo ratings yet
- Red Cell Membrane Structure 2Document16 pagesRed Cell Membrane Structure 2MONo ratings yet
- Fisiologi Cairan Tubuh & Darah: Dr. Nindya Aryanty, M.Med - EdDocument43 pagesFisiologi Cairan Tubuh & Darah: Dr. Nindya Aryanty, M.Med - EdIman TaufiqNo ratings yet
- Introductory One Written Answers: 1) Mention 4 Sultar Containing GAGS and The Function of Two of ThemDocument8 pagesIntroductory One Written Answers: 1) Mention 4 Sultar Containing GAGS and The Function of Two of ThemEbrahim RagabNo ratings yet
- Plasma MembraneDocument18 pagesPlasma MembraneJemuel Bucud LagartoNo ratings yet
- Lesson 3 Red Cell Memebrane Developing-Metabolic PathwaysDocument19 pagesLesson 3 Red Cell Memebrane Developing-Metabolic Pathwaysتجربة أولىNo ratings yet
- Membrane Structure and Function: Lecture OutlineDocument11 pagesMembrane Structure and Function: Lecture Outlinehaha_le12No ratings yet
- Normal Erythropoiesis: A. Inner and Outer LayersDocument9 pagesNormal Erythropoiesis: A. Inner and Outer LayersTina MultazamiNo ratings yet
- Biochem ReportDocument10 pagesBiochem ReportLahari PylaNo ratings yet
- Unit 2 ReviewerDocument4 pagesUnit 2 Revieweraaleah moscaNo ratings yet
- Composition of Blood DR - Fayaz MemonDocument29 pagesComposition of Blood DR - Fayaz Memonhifzaaijaz366No ratings yet
- Lecture 4 - MembraneDocument30 pagesLecture 4 - MembranePhạm ĐứcNo ratings yet
- Lecture 4 - MembraneDocument30 pagesLecture 4 - MembranePhạm ĐứcNo ratings yet
- 03lect Cell2 Summary2017Document4 pages03lect Cell2 Summary2017Dan ChoiNo ratings yet
- Plasma Membrane and Transport MechanismsDocument63 pagesPlasma Membrane and Transport MechanismsVaccedNo ratings yet
- Chemical Composition of Blood Cells: MLS 302: Basic HaematologyDocument13 pagesChemical Composition of Blood Cells: MLS 302: Basic HaematologyBarakat IsmailNo ratings yet
- Rbcs Metabolism: D.Hythem SaeedDocument19 pagesRbcs Metabolism: D.Hythem Saeedsamar yousif mohamedNo ratings yet
- Zoology Lecture 4Document18 pagesZoology Lecture 4Stephen OcheaNo ratings yet
- HUBS2206 - Human Biochemistry and CellDocument87 pagesHUBS2206 - Human Biochemistry and CellLuke ThomsonNo ratings yet
- X Membrane and TransportDocument35 pagesX Membrane and TransportHitesh MoreNo ratings yet
- Red Blood Cells, Functions, Diseases A Simple Guide To The Condition, Diagnosis, Treatment, And Related ConditionsFrom EverandRed Blood Cells, Functions, Diseases A Simple Guide To The Condition, Diagnosis, Treatment, And Related ConditionsNo ratings yet
- Activity 4-Smearing and Simple StainingDocument7 pagesActivity 4-Smearing and Simple StainingMyedelle SeacorNo ratings yet
- Governmental Health AgenciesDocument4 pagesGovernmental Health AgenciesMyedelle SeacorNo ratings yet
- 6 ETS VenipunctureDocument7 pages6 ETS VenipunctureMyedelle SeacorNo ratings yet
- Phlebotomy TechniqueDocument2 pagesPhlebotomy TechniqueMyedelle SeacorNo ratings yet
- The Philippines During Martial Law: "Suko Na! Talo Ka Na! Marcos, LAYAS NA!" - Sigaw NG BayanDocument23 pagesThe Philippines During Martial Law: "Suko Na! Talo Ka Na! Marcos, LAYAS NA!" - Sigaw NG BayanMyedelle SeacorNo ratings yet
- MLS109Lab LabApp&MicroscopeDocument4 pagesMLS109Lab LabApp&MicroscopeMyedelle SeacorNo ratings yet
- Phase of MatterDocument12 pagesPhase of MatterMyedelle SeacorNo ratings yet
- Physical Characteristics: Functions of BloodDocument5 pagesPhysical Characteristics: Functions of BloodMyedelle SeacorNo ratings yet
- Normoblastic Mat Urat IonDocument15 pagesNormoblastic Mat Urat IonMyedelle SeacorNo ratings yet
- Medical Examiner Qualifications: Please PRINT ClearlyDocument2 pagesMedical Examiner Qualifications: Please PRINT ClearlyMyedelle SeacorNo ratings yet
- Transwomen Are Women: Writing PromptDocument2 pagesTranswomen Are Women: Writing PromptMyedelle SeacorNo ratings yet
- The Phil Blood Bank Laws-PART1Document5 pagesThe Phil Blood Bank Laws-PART1Myedelle SeacorNo ratings yet
- REFERENCE NUMBER: 000014120: and Mathematics (Stem)Document2 pagesREFERENCE NUMBER: 000014120: and Mathematics (Stem)Myedelle SeacorNo ratings yet
- How To Write A Position PaperDocument10 pagesHow To Write A Position PaperMyedelle SeacorNo ratings yet
- The Phil Blood Bank Laws-PART1Document6 pagesThe Phil Blood Bank Laws-PART1Myedelle SeacorNo ratings yet
- Empowerment Technologies Midterms ReviewerDocument11 pagesEmpowerment Technologies Midterms ReviewerMyedelle SeacorNo ratings yet
- 12-Stem-C Capstone Research Project: Name Field Capstone TitleDocument1 page12-Stem-C Capstone Research Project: Name Field Capstone TitleMyedelle SeacorNo ratings yet
- Sts History ReviewerDocument3 pagesSts History ReviewerMyedelle SeacorNo ratings yet
- Introduction To The LessonDocument4 pagesIntroduction To The LessonMyedelle SeacorNo ratings yet
- Chemistry Investigatory ProjectDocument16 pagesChemistry Investigatory ProjectDibyanshu NathNo ratings yet
- Asm1 21200Document6 pagesAsm1 21200nooneNo ratings yet
- Human Anatomy and Physiology 2nd Edition Amerman Test BankDocument32 pagesHuman Anatomy and Physiology 2nd Edition Amerman Test BankallisontaylorfnrqzamgksNo ratings yet
- Pgurl 2679036134142300Document4 pagesPgurl 2679036134142300Barbara VasquezNo ratings yet
- Major Classification of Indian SoilsDocument4 pagesMajor Classification of Indian SoilsALABANI MANGANo ratings yet
- Collagen StructureDocument42 pagesCollagen StructureAatmaanandaNo ratings yet
- Carbohydrates NotesDocument28 pagesCarbohydrates Noteschaitanyaa baliNo ratings yet
- Pig Iron Blast Furnace: Mcqs Preparation For Engineering Competitive ExamsDocument20 pagesPig Iron Blast Furnace: Mcqs Preparation For Engineering Competitive ExamschauhanNo ratings yet
- Lec 27Document18 pagesLec 27capcanNo ratings yet
- Hard Gelatin CapsulesDocument58 pagesHard Gelatin Capsulesas.jessy0608No ratings yet
- Ufi-String D PDFDocument4 pagesUfi-String D PDFNermeen ElmelegaeNo ratings yet
- 3 LAB ACTIVITY SHEET 3 Precipitation and Solubility 1Document3 pages3 LAB ACTIVITY SHEET 3 Precipitation and Solubility 1Jurel JohnNo ratings yet
- Chemistry Content Based ExpirimentsDocument22 pagesChemistry Content Based ExpirimentsHari VaarthanNo ratings yet
- Dowex™ Monosphere™ 88: Product Type Matrix Functional GroupDocument2 pagesDowex™ Monosphere™ 88: Product Type Matrix Functional GroupBrianNo ratings yet
- Class 12 Chemistry MCQ Worksheet-2Document7 pagesClass 12 Chemistry MCQ Worksheet-28D audio StationNo ratings yet
- Chemistry of BORON and SILICONDocument32 pagesChemistry of BORON and SILICONprashantNo ratings yet
- MSDSDocument25 pagesMSDShenry.anthirayasNo ratings yet
- HDRscholrarship AustraliaDocument2 pagesHDRscholrarship AustraliaMuhammad Sohail AshrafNo ratings yet
- Preparation of ColloidsDocument11 pagesPreparation of ColloidsNurhidayah Mat Sarit80% (5)
- Preparation of Cupramonium Rayon ThreadDocument22 pagesPreparation of Cupramonium Rayon ThreadRahul MNo ratings yet
- Textbook of CosmetologyDocument96 pagesTextbook of CosmetologyRicky SetiawanNo ratings yet
- CLASS VIII QUESTION BANK - 4. Metals and Non-MetalsDocument7 pagesCLASS VIII QUESTION BANK - 4. Metals and Non-MetalsSurbhi NayarNo ratings yet
- Advanced Concrete Technology Past Paper Questions 1Document11 pagesAdvanced Concrete Technology Past Paper Questions 1Asad QaziNo ratings yet
- 56 102 1 SMDocument6 pages56 102 1 SMAzka Hamam BaihaqiNo ratings yet
- Keerthi M.Nair, Vignesh Kumaravel, Suresh C Pillai Carbonaceous Cathode Materials For Electro-Fenton TechnologyDocument142 pagesKeerthi M.Nair, Vignesh Kumaravel, Suresh C Pillai Carbonaceous Cathode Materials For Electro-Fenton TechnologyVăn Thành MaiNo ratings yet
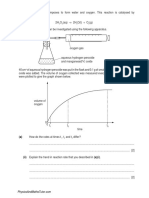
- 1 Hydrogen Peroxide Decomposes To Form Water and Oxygen. This Reaction Is Catalysed byDocument9 pages1 Hydrogen Peroxide Decomposes To Form Water and Oxygen. This Reaction Is Catalysed byKelvin DenhereNo ratings yet
- and 68070 S2012 Final v2 No AnswersDocument42 pagesand 68070 S2012 Final v2 No AnswersZadrin TuckerNo ratings yet
- 2016 Book TheZincBromineFlowBatteryDocument113 pages2016 Book TheZincBromineFlowBatteryraghu7862No ratings yet
- A Combined Spectroscopy and Computational MoleculaDocument13 pagesA Combined Spectroscopy and Computational MoleculadubasilioNo ratings yet