Professional Documents
Culture Documents
Sbco e Poster
Sbco e Poster
Uploaded by
Jenifer KaterineCopyright:
Available Formats
You might also like
- Giuliano 1997Document6 pagesGiuliano 1997angelica cuevasNo ratings yet
- Evaluation of Axillary Lymphadenopathy With Carcinoma BreastDocument42 pagesEvaluation of Axillary Lymphadenopathy With Carcinoma BreastMashrufNo ratings yet
- Sentinel Lymph Node Methods in Breast CancerDocument10 pagesSentinel Lymph Node Methods in Breast CancerZuriNo ratings yet
- Wimmer2020 Article ImpactOfSurgicalMarginsInBreasDocument8 pagesWimmer2020 Article ImpactOfSurgicalMarginsInBreasNelly ChacónNo ratings yet
- Fertility Conserving Management of Early Cervical Cancer Our Experience of LLETZ and Pelvic Lymph Node DissectionDocument6 pagesFertility Conserving Management of Early Cervical Cancer Our Experience of LLETZ and Pelvic Lymph Node DissectionKaleb Rudy HartawanNo ratings yet
- Cryosurgery of Breast Cancer: Lizhi Niu, Liang Zhou, Kecheng XuDocument8 pagesCryosurgery of Breast Cancer: Lizhi Niu, Liang Zhou, Kecheng XuKaton Adi NugrohoNo ratings yet
- Nipple Discharge 3Document3 pagesNipple Discharge 3Dima PathNo ratings yet
- Ajr 10 4865Document6 pagesAjr 10 4865ChavdarNo ratings yet
- Lessismore:Minimally Invasiveandquality Surgicalmanagementof GynecologiccancerDocument12 pagesLessismore:Minimally Invasiveandquality Surgicalmanagementof GynecologiccancerNita AgarwalNo ratings yet
- Linfonodo CentinelaDocument8 pagesLinfonodo CentinelaKaren Palma CarrascoNo ratings yet
- Importance of Intraoperative-Touch Imprint Cytology in The Diagnosis of Neoplastic Lesions of Breast.Document8 pagesImportance of Intraoperative-Touch Imprint Cytology in The Diagnosis of Neoplastic Lesions of Breast.IOSRjournalNo ratings yet
- Overview of Sentinel Lymph Node Biopsy in Breast Cancer - UpToDateDocument37 pagesOverview of Sentinel Lymph Node Biopsy in Breast Cancer - UpToDatemayteveronica1000No ratings yet
- Accepted Manuscript: 10.1016/j.ejso.2016.12.011Document26 pagesAccepted Manuscript: 10.1016/j.ejso.2016.12.011akayaNo ratings yet
- Breast CancerDocument8 pagesBreast CancerRobert RobertsNo ratings yet
- Phyllodes TumorsDocument4 pagesPhyllodes TumorsNexi anessaNo ratings yet
- An Observation Trial Without Surgical Treatment in Patients With Papillary Microcarcinoma of The ThyroidDocument7 pagesAn Observation Trial Without Surgical Treatment in Patients With Papillary Microcarcinoma of The Thyroiddavid villamilNo ratings yet
- Salivary Gland MalignanciesDocument13 pagesSalivary Gland MalignanciesestantevirtualdosmeuslivrosNo ratings yet
- SMJ 60 247Document6 pagesSMJ 60 247Chee Yung NgNo ratings yet
- CIN SeminarDocument53 pagesCIN SeminarA PoudelNo ratings yet
- ST Gallen 1995Document6 pagesST Gallen 1995Nicola CrestiNo ratings yet
- nejm乳腺癌Document8 pagesnejm乳腺癌shentujin1997No ratings yet
- Baek 2017Document10 pagesBaek 2017witaNo ratings yet
- Laparoscopic-Assisted Radical Vaginal Hysterectomy (LARVH) : Prospective Evaluation of 200 Patients With Cervical CancerDocument7 pagesLaparoscopic-Assisted Radical Vaginal Hysterectomy (LARVH) : Prospective Evaluation of 200 Patients With Cervical CancerHari NugrohoNo ratings yet
- Es Necesario BGC Impronta 2019 OriginalDocument9 pagesEs Necesario BGC Impronta 2019 OriginalGlenda Marina Falcon PachecoNo ratings yet
- Sentinel Lymph Node Biopsy in Breast Cancer - A Technical and Clinical AppraisalDocument7 pagesSentinel Lymph Node Biopsy in Breast Cancer - A Technical and Clinical AppraisalIris LanNo ratings yet
- Surgery For Cervical Cancer Consensus & ControversiesDocument9 pagesSurgery For Cervical Cancer Consensus & ControversiesmilenahugueninNo ratings yet
- Elective Neck Irradiation N0 Neck Jia KouDocument13 pagesElective Neck Irradiation N0 Neck Jia KouSanjog SinghNo ratings yet
- JR Onkologi Bedah Kepala LeherDocument21 pagesJR Onkologi Bedah Kepala LeherRina DesdwiNo ratings yet
- AxilaDocument3 pagesAxilaEmilio SanchezNo ratings yet
- REVIEW. Keywords - Sentinel Lymph Node, Lymphatic Mapping, Breast Cancer, Esophageal Cancer, Less Invasive SurgeryDocument18 pagesREVIEW. Keywords - Sentinel Lymph Node, Lymphatic Mapping, Breast Cancer, Esophageal Cancer, Less Invasive SurgeryMDNo ratings yet
- Axillary ClearenceDocument9 pagesAxillary ClearencelakanthNo ratings yet
- Amivantamab plus lazertinib versus osimertinib in first-line EGFR-mutant advanced non-small-cell lung cancer with biomarkers of high-risk diseaseDocument12 pagesAmivantamab plus lazertinib versus osimertinib in first-line EGFR-mutant advanced non-small-cell lung cancer with biomarkers of high-risk diseaseIvan PerezNo ratings yet
- Abstracts 991: ST STDocument1 pageAbstracts 991: ST STAli Sibra MulluziNo ratings yet
- Preservation of Ejaculatory Function After PostcheDocument8 pagesPreservation of Ejaculatory Function After PostcheSeb TckNo ratings yet
- Clinical Feasibility of Imaging With Indocyanine G PDFDocument6 pagesClinical Feasibility of Imaging With Indocyanine G PDFMarisol MorenoNo ratings yet
- Cancer - 2005 - Kirova - Radiation Induced Sarcomas After Radiotherapy For Breast CarcinomaDocument8 pagesCancer - 2005 - Kirova - Radiation Induced Sarcomas After Radiotherapy For Breast CarcinomaRizki Amalia JuwitaNo ratings yet
- Rosendahl 2017Document7 pagesRosendahl 2017Prince VallejosNo ratings yet
- Evolution of Axillary Nodal Staging in Breast Cancer: Clinical Implications of The ACOSOG Z0011 TrialDocument10 pagesEvolution of Axillary Nodal Staging in Breast Cancer: Clinical Implications of The ACOSOG Z0011 Trialkarl abiaadNo ratings yet
- Important Surgical Concepts and Techniques in Inguinal Lymph Node DissectionDocument8 pagesImportant Surgical Concepts and Techniques in Inguinal Lymph Node DissectionVlad GrigoreNo ratings yet
- Axillary Clearance in MRMDocument58 pagesAxillary Clearance in MRMGanesh VlNo ratings yet
- Annsurg00055 0187 PDFDocument8 pagesAnnsurg00055 0187 PDFÁngel AranaNo ratings yet
- Breast Carcinoma in Axillary Tail of Spence: A Rare Case ReportDocument4 pagesBreast Carcinoma in Axillary Tail of Spence: A Rare Case Reportrajesh domakuntiNo ratings yet
- Cancers 16 01623Document19 pagesCancers 16 01623chfang410512No ratings yet
- Endoscopic Excision of Antrochoanal PolypDocument3 pagesEndoscopic Excision of Antrochoanal PolypreginalisaNo ratings yet
- Pure Laparoscopic Management of A Giant Ovarian Cyst in An AdolescentDocument3 pagesPure Laparoscopic Management of A Giant Ovarian Cyst in An AdolescentValian IndrianyNo ratings yet
- 64-Texto Del Artículo-720-1-10-20201209Document7 pages64-Texto Del Artículo-720-1-10-20201209carolinaNo ratings yet
- 2024 blue dye alone for sentinel lymph node biopsy after neoadjuvant chemotherapy in patients with initially node-positive breast cancerDocument7 pages2024 blue dye alone for sentinel lymph node biopsy after neoadjuvant chemotherapy in patients with initially node-positive breast cancerMohamed Mahmoud EzzatNo ratings yet
- Kodama 2021Document7 pagesKodama 2021Sanath NairNo ratings yet
- Suprasellar and Recurrent Pediatric Craniopharyngiomas: Expanding Indications For The Extended Endoscopic Transsphenoidal ApproachDocument9 pagesSuprasellar and Recurrent Pediatric Craniopharyngiomas: Expanding Indications For The Extended Endoscopic Transsphenoidal Approachbenjamin6macindewar6No ratings yet
- AsocogDocument7 pagesAsocogakayaNo ratings yet
- Clear Cell Adenocarcinoma of Ovary About A Rare Case and Review of The LiteratureDocument3 pagesClear Cell Adenocarcinoma of Ovary About A Rare Case and Review of The LiteratureInternational Journal of Innovative Science and Research TechnologyNo ratings yet
- Afp 20100601 P 1339Document8 pagesAfp 20100601 P 1339xiaojun yangNo ratings yet
- Su H. Et Al.,2023Document9 pagesSu H. Et Al.,2023Mai M. AlshalNo ratings yet
- 007 - Clinical-Trials-That-Have-Informed-the-Modern - 2023 - Surgical-Oncology-ClinicsDocument20 pages007 - Clinical-Trials-That-Have-Informed-the-Modern - 2023 - Surgical-Oncology-ClinicsDr-Mohammad Ali-Fayiz Al TamimiNo ratings yet
- Dam C2015Document6 pagesDam C2015Gordana PuzovicNo ratings yet
- Will Surgery Be A Part of Breast Cancer Treatment in The FutureDocument5 pagesWill Surgery Be A Part of Breast Cancer Treatment in The FutureValentina MihaelaNo ratings yet
- Transoral Laser Surgery For Supraglottic CancerDocument9 pagesTransoral Laser Surgery For Supraglottic CancerJose ManuelNo ratings yet
- FNAC Axillary 2012bDocument7 pagesFNAC Axillary 2012bDima PathNo ratings yet
- Fabs and Onco Post TestDocument20 pagesFabs and Onco Post TestJe KirsteneNo ratings yet
- 18 Cellular AberrationsDocument70 pages18 Cellular AberrationsBea Bianca CruzNo ratings yet
- UNIT PLAN Clinical Sp. IIDocument5 pagesUNIT PLAN Clinical Sp. IIedrinsneNo ratings yet
- MELANOMADocument3 pagesMELANOMAKathlyn PactorananNo ratings yet
- Koagulasi Intravaskuler Diseminata Pada Kanker: January 2015Document13 pagesKoagulasi Intravaskuler Diseminata Pada Kanker: January 2015Sri IriantiNo ratings yet
- Silverberg Grading SystemDocument7 pagesSilverberg Grading SystemFlaviu Ionuț FaurNo ratings yet
- Articulo Endometrio y Menopáusisa 2021Document5 pagesArticulo Endometrio y Menopáusisa 2021Fabiola Milagros Castillo LlicaNo ratings yet
- 8WCC New DelhiDocument17 pages8WCC New DelhicaaaNo ratings yet
- Magnetic Resonance Imaging of The Breast: Recommendations From The EUSOMA Working GroupDocument21 pagesMagnetic Resonance Imaging of The Breast: Recommendations From The EUSOMA Working GroupNelly ChacónNo ratings yet
- Henrietta Lacks Web Quest Bio 2015Document4 pagesHenrietta Lacks Web Quest Bio 2015TAKAYAH COUCHCOXNo ratings yet
- Neoplasma 2018-2021Document1 pageNeoplasma 2018-2021Al FatihNo ratings yet
- 2 5458691794407524041Document7 pages2 5458691794407524041Ali ALansariNo ratings yet
- Emerging Treatment Options For Prostate CancerDocument8 pagesEmerging Treatment Options For Prostate CancerMax Carrasco SanchezNo ratings yet
- Urological Cancer Clinical ScenariosDocument27 pagesUrological Cancer Clinical ScenariosMohammed FathyNo ratings yet
- Alisa Sokoloff, MD Joins New York Cancer & Blood SpecialistsDocument3 pagesAlisa Sokoloff, MD Joins New York Cancer & Blood SpecialistsPR.comNo ratings yet
- Urology Sample Osce Exam e PDFDocument8 pagesUrology Sample Osce Exam e PDFLana LocoNo ratings yet
- Nihms 737785Document78 pagesNihms 737785afg_19No ratings yet
- KASUS 3 Early Staged Breast CancerDocument27 pagesKASUS 3 Early Staged Breast CancerWadhe Omara TobingNo ratings yet
- Syllabus Oncology 2024Document6 pagesSyllabus Oncology 2024Kunal BhamareNo ratings yet
- Fat-Containing Liver Lesions On Imaging: Detection and Differential DiagnosisDocument10 pagesFat-Containing Liver Lesions On Imaging: Detection and Differential DiagnosisOana AndreeaNo ratings yet
- Lung CancerDocument3 pagesLung CancerApril Ann HortilanoNo ratings yet
- Breast CalcificationsDocument2 pagesBreast CalcificationsAndreea MorgovanNo ratings yet
- Case Study Cancer of The Female BreastDocument6 pagesCase Study Cancer of The Female BreastKath Dela CruzNo ratings yet
- Capecitabine As Adjuvant Treatment For Stage III Colon Cancer - NEJMDocument6 pagesCapecitabine As Adjuvant Treatment For Stage III Colon Cancer - NEJMAkhil SharmaNo ratings yet
- Artificial Intelligence For Clinical OncologyDocument23 pagesArtificial Intelligence For Clinical OncologyJuan Fernando Méndez RamírezNo ratings yet
- Atividade de Interpretação em PDFDocument4 pagesAtividade de Interpretação em PDFgeovanavilela.sampaioNo ratings yet
- Contouring Inguinal and Femoral Nodes How Much Margin Is Needed Around The Vessels?Document5 pagesContouring Inguinal and Femoral Nodes How Much Margin Is Needed Around The Vessels?Rainey AhmadNo ratings yet
- Blood Group Change in Pediatric Leukemia: A Rare Phenomena: Scientific LetterDocument1 pageBlood Group Change in Pediatric Leukemia: A Rare Phenomena: Scientific LetterparamitaekadevaNo ratings yet
- Depth of Invasion in Oral Squamous Cell CarcinomaDocument63 pagesDepth of Invasion in Oral Squamous Cell CarcinomaDR. ISHITA SINGHALNo ratings yet
- Primary Bone Tumor Due Tue Osteosarcoma: Nurul Ishla Ardy A C014182071Document34 pagesPrimary Bone Tumor Due Tue Osteosarcoma: Nurul Ishla Ardy A C014182071MYMANo ratings yet
Sbco e Poster
Sbco e Poster
Uploaded by
Jenifer KaterineOriginal Description:
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Sbco e Poster
Sbco e Poster
Uploaded by
Jenifer KaterineCopyright:
Available Formats
ID 123730
USE OF RADIOACTIVE IODINE (I125) SEED FOR
THE LOCALIZATION OF AXILLARY LYMPH
NODES IN BREAST CANCER PATIENTS
Anschau JKP¹; Krumenauerr KM¹; Pizaia R¹; Lourenço RAMC¹; Costa MM2;
Oliveira MVFT3; Fonseca LMB4; Júnior RRM4; Franca CAS4; Penna ABRC5.
¹Acadêmico IDOMED - Angra dos Reis/RJ, ² Hospital Universitário
Clementino Fraga Filho – Rio de Janeiro, 3LBG Celular Diagnóstico,
4
Docente IDOMED – Angra dos Reis/RJ, 5 A.B.R.C. - Radioterapia Ingá
E-mail para contato: jeniferkpa@gmail.com
INTRODUCTION: Therapies against breast cancer are diverse. Neoadjuvant
chemotherapy is an example used as a tool to decrease the tumor stage, improve
breast conservation therapy, and reduce the stage of axillary lymph nodes. Routine
axillary lymph node dissection is increasingly being considered overtreatment in women
where a complete pathological axillary response is obtained. As an alternative, axillary
lymph node marking with low-dose I125 seeds is being used, known as a minimally
invasive method to assess the pathological response of lymph node metastases after
neoadjuvant systemic treatment in patients with breast cancer. The seeds help with the
precision of the lesion territory, where they are located by a probe capable of
highlighting the emitted gamma radiation. Therefore, it is possible to reduce the amount
of dissected normal tissue, minimizing risks, and avoiding unwanted aesthetic
alterations.
OBJECTIVE: The present study assessed the feasibility of marking clinically suspected
enlarged axillary nodes with I125 seeds before neoadjuvant chemotherapy, facilitating
their identification and surgical excision after neoadjuvant treatment.
METHODS: A retrospective analysis was conducted on breast cancer patients who
underwent marking of clinically positive axillary lymph nodes with I125 before receiving
neoadjuvant chemotherapy. The gamma-probe was used to guide the excision of the
marked lymph node, ensuring the removal of the correct lymph node by detecting the
I125 source within it and confirming the absence of radiation in the rest of the axilla.
The study was approved by the research ethics committee (CAEE
47149315.4.0000.5533).
RESULTS: The study included a total of nine patients with clinically node-positive
disease. The mean age was 60 years (95% CI 57.4 - 62.7 / SD ± 10.0), and all patients
had invasive ductal carcinoma. The axillary lymph node marking procedure using I125
seeds was successfully performed in all cases, with no major complications reported.
The marked axillary lymph nodes facilitated accurate identification and dissection
during subsequent surgery, as the I125 seeds were found within the lymph node in all
patients.
CONCLUSION: This single-institution experience demonstrates the successful use of
I125 for axillary lymph node marking in breast cancer patients, facilitating its surgical
removal.
REFERENCES: 1- Axillary Staging After Neoadjuvant Chemotherapy for Breast Cancer: A Pilot Study
Combining Sentinel Lymph Node Biopsywith Radioactive Seed Localization of Pre-treatment Positive Axillary
Lymph Nodes.
2- A Novel Less-invasive Approach for Axillary Staging After Neoadjuvant Chemotherapy in Patients With
Axillary Node-positive Breast Cancer by Combining Radioactive Iodine Seed Localization in the Axilla With the
Sentinel Node Procedure (RISAS): A Dutch Prospective Multicenter Validation Study
3- Marking Axillary Lymph Nodes With Radioactive Iodine Seeds for Axillary Staging After Neoadjuvant
Systemic Treatment in Breast Cancer Patient.
4- Radioactive Iodine-125 in Tumor Therapy: Advances and Future Directions
5- Radioactive Seed Localization for Conservative Surgery of Nonpalpable Breast Cancer: Recommendations for
Technology Implantation Program
You might also like
- Giuliano 1997Document6 pagesGiuliano 1997angelica cuevasNo ratings yet
- Evaluation of Axillary Lymphadenopathy With Carcinoma BreastDocument42 pagesEvaluation of Axillary Lymphadenopathy With Carcinoma BreastMashrufNo ratings yet
- Sentinel Lymph Node Methods in Breast CancerDocument10 pagesSentinel Lymph Node Methods in Breast CancerZuriNo ratings yet
- Wimmer2020 Article ImpactOfSurgicalMarginsInBreasDocument8 pagesWimmer2020 Article ImpactOfSurgicalMarginsInBreasNelly ChacónNo ratings yet
- Fertility Conserving Management of Early Cervical Cancer Our Experience of LLETZ and Pelvic Lymph Node DissectionDocument6 pagesFertility Conserving Management of Early Cervical Cancer Our Experience of LLETZ and Pelvic Lymph Node DissectionKaleb Rudy HartawanNo ratings yet
- Cryosurgery of Breast Cancer: Lizhi Niu, Liang Zhou, Kecheng XuDocument8 pagesCryosurgery of Breast Cancer: Lizhi Niu, Liang Zhou, Kecheng XuKaton Adi NugrohoNo ratings yet
- Nipple Discharge 3Document3 pagesNipple Discharge 3Dima PathNo ratings yet
- Ajr 10 4865Document6 pagesAjr 10 4865ChavdarNo ratings yet
- Lessismore:Minimally Invasiveandquality Surgicalmanagementof GynecologiccancerDocument12 pagesLessismore:Minimally Invasiveandquality Surgicalmanagementof GynecologiccancerNita AgarwalNo ratings yet
- Linfonodo CentinelaDocument8 pagesLinfonodo CentinelaKaren Palma CarrascoNo ratings yet
- Importance of Intraoperative-Touch Imprint Cytology in The Diagnosis of Neoplastic Lesions of Breast.Document8 pagesImportance of Intraoperative-Touch Imprint Cytology in The Diagnosis of Neoplastic Lesions of Breast.IOSRjournalNo ratings yet
- Overview of Sentinel Lymph Node Biopsy in Breast Cancer - UpToDateDocument37 pagesOverview of Sentinel Lymph Node Biopsy in Breast Cancer - UpToDatemayteveronica1000No ratings yet
- Accepted Manuscript: 10.1016/j.ejso.2016.12.011Document26 pagesAccepted Manuscript: 10.1016/j.ejso.2016.12.011akayaNo ratings yet
- Breast CancerDocument8 pagesBreast CancerRobert RobertsNo ratings yet
- Phyllodes TumorsDocument4 pagesPhyllodes TumorsNexi anessaNo ratings yet
- An Observation Trial Without Surgical Treatment in Patients With Papillary Microcarcinoma of The ThyroidDocument7 pagesAn Observation Trial Without Surgical Treatment in Patients With Papillary Microcarcinoma of The Thyroiddavid villamilNo ratings yet
- Salivary Gland MalignanciesDocument13 pagesSalivary Gland MalignanciesestantevirtualdosmeuslivrosNo ratings yet
- SMJ 60 247Document6 pagesSMJ 60 247Chee Yung NgNo ratings yet
- CIN SeminarDocument53 pagesCIN SeminarA PoudelNo ratings yet
- ST Gallen 1995Document6 pagesST Gallen 1995Nicola CrestiNo ratings yet
- nejm乳腺癌Document8 pagesnejm乳腺癌shentujin1997No ratings yet
- Baek 2017Document10 pagesBaek 2017witaNo ratings yet
- Laparoscopic-Assisted Radical Vaginal Hysterectomy (LARVH) : Prospective Evaluation of 200 Patients With Cervical CancerDocument7 pagesLaparoscopic-Assisted Radical Vaginal Hysterectomy (LARVH) : Prospective Evaluation of 200 Patients With Cervical CancerHari NugrohoNo ratings yet
- Es Necesario BGC Impronta 2019 OriginalDocument9 pagesEs Necesario BGC Impronta 2019 OriginalGlenda Marina Falcon PachecoNo ratings yet
- Sentinel Lymph Node Biopsy in Breast Cancer - A Technical and Clinical AppraisalDocument7 pagesSentinel Lymph Node Biopsy in Breast Cancer - A Technical and Clinical AppraisalIris LanNo ratings yet
- Surgery For Cervical Cancer Consensus & ControversiesDocument9 pagesSurgery For Cervical Cancer Consensus & ControversiesmilenahugueninNo ratings yet
- Elective Neck Irradiation N0 Neck Jia KouDocument13 pagesElective Neck Irradiation N0 Neck Jia KouSanjog SinghNo ratings yet
- JR Onkologi Bedah Kepala LeherDocument21 pagesJR Onkologi Bedah Kepala LeherRina DesdwiNo ratings yet
- AxilaDocument3 pagesAxilaEmilio SanchezNo ratings yet
- REVIEW. Keywords - Sentinel Lymph Node, Lymphatic Mapping, Breast Cancer, Esophageal Cancer, Less Invasive SurgeryDocument18 pagesREVIEW. Keywords - Sentinel Lymph Node, Lymphatic Mapping, Breast Cancer, Esophageal Cancer, Less Invasive SurgeryMDNo ratings yet
- Axillary ClearenceDocument9 pagesAxillary ClearencelakanthNo ratings yet
- Amivantamab plus lazertinib versus osimertinib in first-line EGFR-mutant advanced non-small-cell lung cancer with biomarkers of high-risk diseaseDocument12 pagesAmivantamab plus lazertinib versus osimertinib in first-line EGFR-mutant advanced non-small-cell lung cancer with biomarkers of high-risk diseaseIvan PerezNo ratings yet
- Abstracts 991: ST STDocument1 pageAbstracts 991: ST STAli Sibra MulluziNo ratings yet
- Preservation of Ejaculatory Function After PostcheDocument8 pagesPreservation of Ejaculatory Function After PostcheSeb TckNo ratings yet
- Clinical Feasibility of Imaging With Indocyanine G PDFDocument6 pagesClinical Feasibility of Imaging With Indocyanine G PDFMarisol MorenoNo ratings yet
- Cancer - 2005 - Kirova - Radiation Induced Sarcomas After Radiotherapy For Breast CarcinomaDocument8 pagesCancer - 2005 - Kirova - Radiation Induced Sarcomas After Radiotherapy For Breast CarcinomaRizki Amalia JuwitaNo ratings yet
- Rosendahl 2017Document7 pagesRosendahl 2017Prince VallejosNo ratings yet
- Evolution of Axillary Nodal Staging in Breast Cancer: Clinical Implications of The ACOSOG Z0011 TrialDocument10 pagesEvolution of Axillary Nodal Staging in Breast Cancer: Clinical Implications of The ACOSOG Z0011 Trialkarl abiaadNo ratings yet
- Important Surgical Concepts and Techniques in Inguinal Lymph Node DissectionDocument8 pagesImportant Surgical Concepts and Techniques in Inguinal Lymph Node DissectionVlad GrigoreNo ratings yet
- Axillary Clearance in MRMDocument58 pagesAxillary Clearance in MRMGanesh VlNo ratings yet
- Annsurg00055 0187 PDFDocument8 pagesAnnsurg00055 0187 PDFÁngel AranaNo ratings yet
- Breast Carcinoma in Axillary Tail of Spence: A Rare Case ReportDocument4 pagesBreast Carcinoma in Axillary Tail of Spence: A Rare Case Reportrajesh domakuntiNo ratings yet
- Cancers 16 01623Document19 pagesCancers 16 01623chfang410512No ratings yet
- Endoscopic Excision of Antrochoanal PolypDocument3 pagesEndoscopic Excision of Antrochoanal PolypreginalisaNo ratings yet
- Pure Laparoscopic Management of A Giant Ovarian Cyst in An AdolescentDocument3 pagesPure Laparoscopic Management of A Giant Ovarian Cyst in An AdolescentValian IndrianyNo ratings yet
- 64-Texto Del Artículo-720-1-10-20201209Document7 pages64-Texto Del Artículo-720-1-10-20201209carolinaNo ratings yet
- 2024 blue dye alone for sentinel lymph node biopsy after neoadjuvant chemotherapy in patients with initially node-positive breast cancerDocument7 pages2024 blue dye alone for sentinel lymph node biopsy after neoadjuvant chemotherapy in patients with initially node-positive breast cancerMohamed Mahmoud EzzatNo ratings yet
- Kodama 2021Document7 pagesKodama 2021Sanath NairNo ratings yet
- Suprasellar and Recurrent Pediatric Craniopharyngiomas: Expanding Indications For The Extended Endoscopic Transsphenoidal ApproachDocument9 pagesSuprasellar and Recurrent Pediatric Craniopharyngiomas: Expanding Indications For The Extended Endoscopic Transsphenoidal Approachbenjamin6macindewar6No ratings yet
- AsocogDocument7 pagesAsocogakayaNo ratings yet
- Clear Cell Adenocarcinoma of Ovary About A Rare Case and Review of The LiteratureDocument3 pagesClear Cell Adenocarcinoma of Ovary About A Rare Case and Review of The LiteratureInternational Journal of Innovative Science and Research TechnologyNo ratings yet
- Afp 20100601 P 1339Document8 pagesAfp 20100601 P 1339xiaojun yangNo ratings yet
- Su H. Et Al.,2023Document9 pagesSu H. Et Al.,2023Mai M. AlshalNo ratings yet
- 007 - Clinical-Trials-That-Have-Informed-the-Modern - 2023 - Surgical-Oncology-ClinicsDocument20 pages007 - Clinical-Trials-That-Have-Informed-the-Modern - 2023 - Surgical-Oncology-ClinicsDr-Mohammad Ali-Fayiz Al TamimiNo ratings yet
- Dam C2015Document6 pagesDam C2015Gordana PuzovicNo ratings yet
- Will Surgery Be A Part of Breast Cancer Treatment in The FutureDocument5 pagesWill Surgery Be A Part of Breast Cancer Treatment in The FutureValentina MihaelaNo ratings yet
- Transoral Laser Surgery For Supraglottic CancerDocument9 pagesTransoral Laser Surgery For Supraglottic CancerJose ManuelNo ratings yet
- FNAC Axillary 2012bDocument7 pagesFNAC Axillary 2012bDima PathNo ratings yet
- Fabs and Onco Post TestDocument20 pagesFabs and Onco Post TestJe KirsteneNo ratings yet
- 18 Cellular AberrationsDocument70 pages18 Cellular AberrationsBea Bianca CruzNo ratings yet
- UNIT PLAN Clinical Sp. IIDocument5 pagesUNIT PLAN Clinical Sp. IIedrinsneNo ratings yet
- MELANOMADocument3 pagesMELANOMAKathlyn PactorananNo ratings yet
- Koagulasi Intravaskuler Diseminata Pada Kanker: January 2015Document13 pagesKoagulasi Intravaskuler Diseminata Pada Kanker: January 2015Sri IriantiNo ratings yet
- Silverberg Grading SystemDocument7 pagesSilverberg Grading SystemFlaviu Ionuț FaurNo ratings yet
- Articulo Endometrio y Menopáusisa 2021Document5 pagesArticulo Endometrio y Menopáusisa 2021Fabiola Milagros Castillo LlicaNo ratings yet
- 8WCC New DelhiDocument17 pages8WCC New DelhicaaaNo ratings yet
- Magnetic Resonance Imaging of The Breast: Recommendations From The EUSOMA Working GroupDocument21 pagesMagnetic Resonance Imaging of The Breast: Recommendations From The EUSOMA Working GroupNelly ChacónNo ratings yet
- Henrietta Lacks Web Quest Bio 2015Document4 pagesHenrietta Lacks Web Quest Bio 2015TAKAYAH COUCHCOXNo ratings yet
- Neoplasma 2018-2021Document1 pageNeoplasma 2018-2021Al FatihNo ratings yet
- 2 5458691794407524041Document7 pages2 5458691794407524041Ali ALansariNo ratings yet
- Emerging Treatment Options For Prostate CancerDocument8 pagesEmerging Treatment Options For Prostate CancerMax Carrasco SanchezNo ratings yet
- Urological Cancer Clinical ScenariosDocument27 pagesUrological Cancer Clinical ScenariosMohammed FathyNo ratings yet
- Alisa Sokoloff, MD Joins New York Cancer & Blood SpecialistsDocument3 pagesAlisa Sokoloff, MD Joins New York Cancer & Blood SpecialistsPR.comNo ratings yet
- Urology Sample Osce Exam e PDFDocument8 pagesUrology Sample Osce Exam e PDFLana LocoNo ratings yet
- Nihms 737785Document78 pagesNihms 737785afg_19No ratings yet
- KASUS 3 Early Staged Breast CancerDocument27 pagesKASUS 3 Early Staged Breast CancerWadhe Omara TobingNo ratings yet
- Syllabus Oncology 2024Document6 pagesSyllabus Oncology 2024Kunal BhamareNo ratings yet
- Fat-Containing Liver Lesions On Imaging: Detection and Differential DiagnosisDocument10 pagesFat-Containing Liver Lesions On Imaging: Detection and Differential DiagnosisOana AndreeaNo ratings yet
- Lung CancerDocument3 pagesLung CancerApril Ann HortilanoNo ratings yet
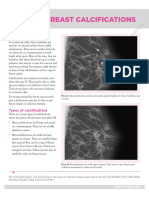
- Breast CalcificationsDocument2 pagesBreast CalcificationsAndreea MorgovanNo ratings yet
- Case Study Cancer of The Female BreastDocument6 pagesCase Study Cancer of The Female BreastKath Dela CruzNo ratings yet
- Capecitabine As Adjuvant Treatment For Stage III Colon Cancer - NEJMDocument6 pagesCapecitabine As Adjuvant Treatment For Stage III Colon Cancer - NEJMAkhil SharmaNo ratings yet
- Artificial Intelligence For Clinical OncologyDocument23 pagesArtificial Intelligence For Clinical OncologyJuan Fernando Méndez RamírezNo ratings yet
- Atividade de Interpretação em PDFDocument4 pagesAtividade de Interpretação em PDFgeovanavilela.sampaioNo ratings yet
- Contouring Inguinal and Femoral Nodes How Much Margin Is Needed Around The Vessels?Document5 pagesContouring Inguinal and Femoral Nodes How Much Margin Is Needed Around The Vessels?Rainey AhmadNo ratings yet
- Blood Group Change in Pediatric Leukemia: A Rare Phenomena: Scientific LetterDocument1 pageBlood Group Change in Pediatric Leukemia: A Rare Phenomena: Scientific LetterparamitaekadevaNo ratings yet
- Depth of Invasion in Oral Squamous Cell CarcinomaDocument63 pagesDepth of Invasion in Oral Squamous Cell CarcinomaDR. ISHITA SINGHALNo ratings yet
- Primary Bone Tumor Due Tue Osteosarcoma: Nurul Ishla Ardy A C014182071Document34 pagesPrimary Bone Tumor Due Tue Osteosarcoma: Nurul Ishla Ardy A C014182071MYMANo ratings yet